
Progress of Amlodipine and Enalapril in the Treatment of
Hypertension
Yifan Wang
College of Pharmaceutical Sciences, Zhejiang Chinese Medical University, Hangzhou, China
Keywords: Hypertension, Amlodipine, Enalapril, Pharmacology, Origin, Delivery, Pharmacoeconomic.
Abstract: BACKGROUND: Hypertension is a long-term chronic disease that has a tremendous negative impact on
people's lives, and its prevalence varies by gender, region, and ethnicity. Calcium channel blockers (CCBs)
and angiotensin-converting enzyme inhibitors (ACEIs) are widely used among the first-line antihypertensive
agents. CONTENT: The type, epidemiology, pathology, and therapeutic means of hypertension were
analysed. The origin of amlodipine, a representative drug of CCBs, and enalapril, a representative drug of
ACEIs for the treatment of hypertension was analysed, and their latest progress in pharmacological effects,
delivery, combined use, and pharmacoeconomics were summarized and compared. RESULTS: Amlodipine
is more suitable for patients who need myocardial protection, and enalapril is more suitable for patients with
ventricular hypertrophy. The combination of the two drugs provides better protection for the cardiovascular
system. Both drugs are primarily administered orally, but different effects can be achieved by using different
dosage forms. Amlodipine is more affordable than enalapril and has a larger market.
1 INTRODUCTION
Heart and cardiovascular and cerebrovascular
diseases are the number one cause of death for people
in developed countries and 44% of people in China
died because of heart and cardiovascular and
cerebrovascular diseases (Bai, et al. 2018). Therefore,
research on the cardiovascular system has become the
focus of attention of scientists in the fields of
medicine and pharmacy from all over the world.
Among them, hypertension, as an important factor in
increasing the risk of cardiovascular disease has
attracted much attention.
This article analyzed s the typology,
epidemiology, pathology, and treatment of
hypertension in the first place. Hypertension, also
known as systemic arterial hypertension, is
manifested by a long-term increase in systemic
arterial blood pressure. Blood pressure is usually
expressed as the ratio of the systolic pressure (that is,
the pressure of the arterial wall when the heart is
contracting) to the diastolic pressure (the pressure
when the heart is in diastole) (Oparil, et al. 2018). As
shown in Table 1, hypertension can be divided into
the following stages. Hypertension is a long-term
chronic disease that has a great negative impact on
people's lives. And because hypertension is difficult
to cure, the vast majority of hypertensive patients
need long-term medication or even lifelong
medication (Tsioufis, and Thomopoulos 2017).
Hypertension is still an unsolved medical problem,
and therefore hypertension research still needs to be
carried out in depth. The analyzation of hypertension
in this paper provides some material to support and
enlighten the subsequent related studies.
Table 1: Categories of BP in Adults
Blood
Pressure
Category
Systolic blood
pressure (SBP)
Diastolic blood
pressure (DBP)
Normal <120mmHg and <80mmHg
Elevated 120~129mmHg and <80mmHg
Hypertension
Stage 1 130~139mmHg or 80~89mmHg
Stage 2 ≥140mmHg or ≥90mmHg
The article analysed the origins of amlodipine, a
representative drug of the CCB class for the treatment
of hypertension, and enalapril, a representative drug of
the ACEI class, to sort out the history of drug
development and make it clearer and more definite.
The pharmacological effects, routes of administration,
co-administration, and the latest progress of
pharmacoeconomics of the two drugs are also
206
Wang, Y.
Progress of Amlodipine and Enalapril in the Treatment of Hypertension.
DOI: 10.5220/0011244300003438
In Proceedings of the 1st International Conference on Health Big Data and Intelligent Healthcare (ICHIH 2022), pages 206-214
ISBN: 978-989-758-596-8
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
summarized and compared. It provides a
comprehensive reference for the selection and
comparison of drugs for the treatment of hypertension,
and lays the foundation for the in-depth study of the
pharmacological effects and targets of action of these
two drugs.
2 HYPERTENSION
2.1 Types of the Hypertension
The cause of the vast majority of hypertension is
unknown, called essential hypertension(EH); a small
amount of hypertension has a cause that can be
checked, called secondary hypertension(SH).
The etiology and mechanism of essential
hypertension are still unclear, but with the continuous
deepening of research, some people believe that
essential hypertension is not a disease but a
syndrome. A major manifestation of this syndrome is
increased blood pressure (Manosroi, and Williams
2019). The emergence of essential hypertension is not
caused by a single factor. It is more caused by the
cumulative effect of multiple factors. Studies have
supported this view, such as genetic inheritance and
long-term high-salt diet. Habits, the influence of the
microbiota in the body on blood pressure
(Chakraborty, et al. 2020).
Common causes of secondary hypertension
include obstructive sleep apnea (OSA), renal
parenchymal disease, renal artery stenosis, and
primary hyperaldosteronism (PA) (Rimoldi, Scherrer,
and
Messerli
2014). It is worth mentioning that many
patients with secondary hypertension still have
hypertension symptoms after removing the secondary
causes. This indicates that many patients with
secondary hypertension may be accompanied by
essential hypertension or irreversible remodeling of
blood vessels.
2.2 Epidemiology of the Hypertension
In China, the symptoms of hypertension are SBP
140mmHg and DBP 90mmHg. According to the
report paper, the prevalence of hypertension in China
is increasing, and it is characterized by 1)higher rates
of hypertension in men than in women, 2)higher
prevalence in the north than in the south, 3)higher
prevalence in rural areas than in urban areas, 4)and
differences in prevalence between ethnic groups (J
Geriatr Cardiol 2019). As of 2015, the awareness rate
of patients with hypertension was only 55.7%, the
treatment rate was less than 50%, and only 20% of the
patients treated had their blood pressure under control
(Wei, et al. 2021).
Globally, about 31.1% of adults suffer from
hypertension, and the incidence in developed
countries is higher than that in developing countries.
There is an age difference in hypertension, that is, as
the age increases, the incidence increases, as shown
in Table 2.
Table 2: Age-specific and age-standardized prevalence estimates and absolute numbers of men and women with hypertension
in high-income and low- and middle-income countries in 2010 (Mills, et al., 2016)
High blood pressure is often not a fatal factor, but
high blood pressure can cause organ damage, increase
the risk of other cardiovascular diseases, and may
have complications. Some complications of
hypertension include renal failure, stroke, heart
failure, coronary heart disease, etc., and most of these
complications can cause death and disability (Flack,
et al. 2003).
Progress of Amlodipine and Enalapril in the Treatment of Hypertension
207

2.3 Pathology of the Hypertension
The basic factors that form arterial blood pressure are
cardiac output and peripheral vascular resistance. The
former is affected by heart function, return blood
volume, and blood volume and the latter is mainly
affected by the tension of the arterioles. The
sympathetic nervous system and the renin-
angiotensin system (RAS) regulate the above two
factors to control blood pressure within a certain
range.
2.4 Treatment of the Hypertension
At present, the treatment of hypertension advocates
comprehensive treatment, that is, non-drug treatment
and drugs are combined to control the patient's blood
pressure.
Non-drug treatment is the change in the patient’s
living habits to control risk factors. This method
advocate reducing sodium and salt intake, reasonable
dietary arrangements, regular exercise, quitting
smoking and drinking, and maintaining a good mood
(J Geriatr Cardiol 2019).
In terms of drug treatment, currently, diuretics,
calcium channel blockers, β receptor blockers,
angiotensin converting enzyme inhibitors (ACEI),
AT
1
receptor blockers, which are widely used or
called first-line antihypertensive drugs, are widely
used medicine. Clinical treatment of hypertension
often adopts combination medications to reduce the
damage to the patient's target organs and reduce
complications (Tsioufis, and Thomopoulos 2017).
3 DESCRIPTION OF CHEMICAL
STRUCTURES OF DRUGS
3.1 Structure and Nomenclature
3.1.1 Structure and Nomenclature of
amlodipine
2D Structure: 3D Structure:
Figure 1: 2D and 3D structures of amlodipine.
The figure 1 shows the 2D and 3D structures of
amlodipine. The amlodipine contains a benzene ring
and a dihydropyridine ring, from the 3D structure can
be seen that the two rings are not co-plane, but in a
state perpendicular to each other. Dihydropyridine
ring 3, 5-bit replacement group is different, so C4 is
the stereoscopic center, and the activity of S
configuration is stronger than R configuration
(Coelho, et al. 2021).
Nomenclature:
The IUPAC Name of amlodipine is 3-O-ethyl 5-
O-methyl 2-(2-aminoethoxymethyl)-4-(2-
chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-
dicarboxylate. This drug has many brand names in the
market, such as Amlobenz, Azor, Caduet, Consensi,
Dafiro, Exforge, Exforge Hct, Katerzia, Lotrel,
Norvasc, Prestalia, Tribenzor, Twynsta, Viacoram.
Clinical drugs amlodipine maleate and amlodipine
besylate are widely used.
3.1.2 Structure and Nomenclature of
enalapril
2D structure: 3D Structure:
Figure 2: 2D and 3D structures of enalapril
The figure 2 shows the 2D and 3D structures of
enalapril. The structure of enalapril contains a
benzene ring, tetrahydropyrrole ring, carboxyl group,
carbonyl group, an ester bond, and other structures.
There are two chiral carbon atoms in the structure.
Nomenclature:
The IUPAC Name of enalapril is (2S)-1-[(2S)-2-
[[(2S)-1-ethoxy-1-oxo-4-phenylbutan-2-
yl]amino]propanoyl]pyrrolidine-2-carboxylic acid.
This drug has three brand names on market, Epaned,
Vaseretic, and Vasotec.
3.2 Chemical and Physical Properties
3.2.1
Chemical and Physical Properties
of amlodipine
The molecular formula is C
20
H
25
ClN
2
O
5
, this drug is
a small molecule drug. Its molecular weight is 408.9,
solid, slightly soluble in water. The 𝑝𝐾
of
ICHIH 2022 - International Conference on Health Big Data and Intelligent Healthcare
208

amlodipine is 9.4(amine)
3.2.2 Chemical and Physical Properties
of enalapril
The molecular formula of enalapril is C
20
H
28
N
2
O
5
,
this drug is a small molecule drug. Its molecular
weight is 376.4, solid, Its solubility in water is
1.64 × 10
mg/L at 25 °C. The pKa of enalapril
p𝐾
1 = 3.0 (carboxylic acid) and p𝐾
2 =
5.5 (secondary amide).
4 DISCUSSION OF DRUG
PHARMACOLOGY
4.1 Origin and History of Drugs
4.1.1 Origin and history of Amlodipine
The study of calcium channel blockers on the
cardiovascular system began around the 1960s.
Amlodipine is the third generation dihydropyridine
calcium channel blocker, which is modified from the
first marketed dihydropyridine CCB nifedipine.
Nifedipine was launched in 1975 and has been
effective in controlling hypertension since it was
launched. Compared with nifedipine, amlodipine has
a long-lasting and stable lowering of blood pressure.
It has become a once-a-day antihypertensive drug,
which greatly improves the compliance of
hypertensive patients (Burges, and Moisey 1994).
Amlodipine can also reverse left ventricular
hypertrophy (Lu, et al. 2016), especially for patients
with hypertension and left ventricular hypertrophy.
4.1.2 Origin and History of Enalapril
In 1971, teprotide, the first active substance with an
inhibitory effect on angiotensin converting enzyme,
was isolated from the venom of a Brazilian snake.
However, the oral administration of teprotide was
ineffective, so it was necessary to find a more stable
structure. Inspired by the inhibitor of
carboxypeptidase A (an exopeptidase containing zinc
ion active center, which was thought to be similar to
ACE at that time) (Cushman, et al. 1982),
succinylproline was synthesized. The product had a
certain inhibitory effect on ACE, but its activity was
low. To further increase the inhibitory activity,
succinylproline was structurally modified by adding
chiral carbon and replacing carboxyl with sulfhydryl.
Surprisingly, this structural transformation increased
the inhibitory effect by 2000 times (Cushman, and
Ondetti 1991). This is the first ACEI drug captopril,
which was listed by Squibb in 1981.
But then captopril was reported to have serious
adverse reactions, such as proteinuria and taste loss.
These adverse reactions were related to the sulfhydryl
group contained in its structure (Jaffe 1986).
Therefore, to eliminate these adverse reactions, the
structure of captopril was modified α- Amphetamine
replaces sulfhydryl group and is made into monoethyl
ester as a prodrug (Patchett 1984). After enalapril
enters the body, it can be metabolized into Enalapril
to exert its efficacy, which was later listed by Merck
in the United States.
4.2 Description of Drug Targets and
How Their Function is Affected by
the Drugs
4.2.1 Pharmacology of Amlodipine
Calcium ion plays an important role in the excitation-
contraction coupling of the myocardium and vascular
smooth muscle. The higher the concentration of
calcium ions entering the cell, the stronger the
contractility of the myocardium and vascular smooth
muscle (Bers 2014). The entry of calcium ions into
cells mainly depends on the calcium channels on the
cell membrane. Calcium channels play an important
physiological role in the body and can be divided into
many subtypes (Zamponi, et al. 2015). The calcium
channels distributed in the cells of the cardiovascular
system are mainly L-type calcium channels.
Amlodipine is the third generation
dihydropyridine (DHP) calcium channel blocker
(CCB). Dihydropyridine drugs mainly inhibit L-type
Ca2 + voltage-gated calcium channels widely
distributed in the cell membrane of the cardiovascular
system (Gao, and Yan 2021), prevent calcium influx,
and inhibit the contractility of myocardium and
vascular smooth muscle. In this way, the myocardial
contractility is weakened, the heart rate is slowed
down, the cardiac ejection is reduced, and the arterial
blood pressure is reduced.
The calcium ion channel consists of α1 subunit
that determines their characteristics and α2, δ, β, γ
subunits help regulate the function of calcium ion
channels (Catterall 2011). Calcium channel blockers
can combine with calcium ion channels when α1 is in
subunit inactivate condition to keep them inactive and
reduce the flow of calcium ions into cells. According
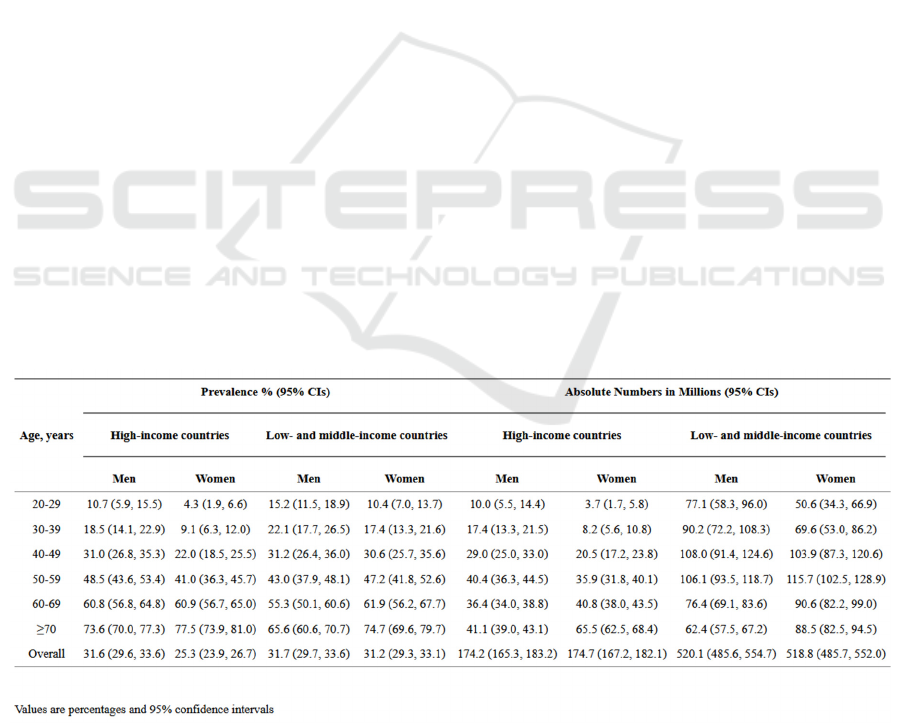
to Tang’s research (Tang, et al. 2016), amlodipine can
be combined with the dihydropyridine combined
pockets of Ca
v
Ab, and the crystal structure of the
Progress of Amlodipine and Enalapril in the Treatment of Hypertension
209
compound can well represent the state of Amlodipine
regulate calcium channels, as shown in the Figure 3.
Unlike non-dihydropyridine calcium channel
blockers (e.g. Verapamil, Diltiazem),
dihydropyridine drugs do not limit the internal flow
of calcium ions by blocking pores but act as a gated
regulator (Catterall, and Swanson, 2015).
Figure 3: Crystal Structure of Compound of Amlodipine
and Ca
v
Ab (Tang, et al. 2016).
The above is the traditional and recognized blood
pressure reduction mechanism of amlodipine. In
addition, recent studies have shown that amlodipine
can also regulate arterial blood pressure in other
ways. For example, amlodipine can inhibit the growth
of vascular smooth muscle, regulate phenotypic
conversion and reduce arterial blood pressure (Fang,
et al. 2019, Stepien, et al. 1998).
4.2.2 Pharmacology of Enalapril
Enalapril is an antihypertensive drug that acts on the
renin angiotensin system (RAS). It belongs to the
angiotensin converting enzyme inhibitor (ACEI). The
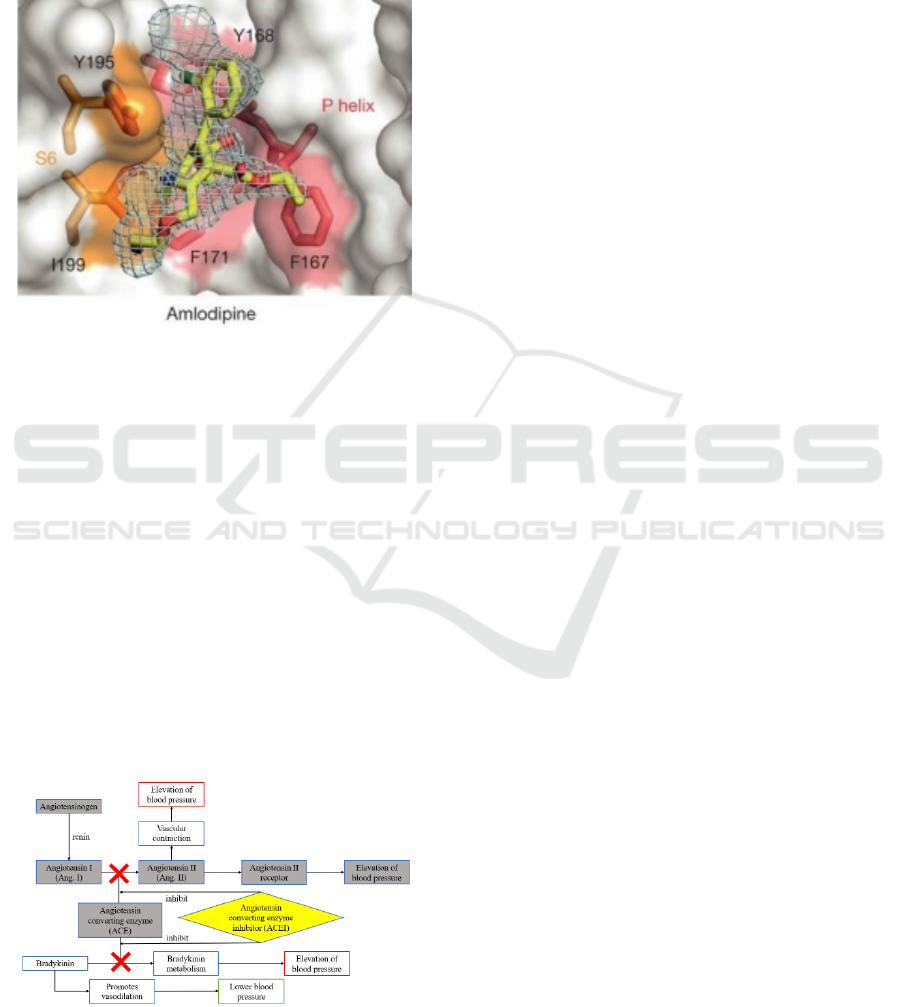
mechanism of ACEI lowers blood pressure is shown
in figure 4.
Figure 4: The mechanism of ACEI lowers blood pressure.
The renin angiotensin system (RAS) is an
important system of body fluid regulation in the
human body. It plays an important role in maintaining
the stability of cardiovascular function and the
balance of body fluid and electrolytes. However,
excessive activation of RAS will lead to pathological
phenomena such as hypertension and congestive
heart failure (Forrester, et al. 2018). Therefore, a
major idea for the treatment of hypertension is to
down-regulate the over-excited RAS.
Renin is a protease synthesized and secreted by renal
near globular cells. It can hydrolyze angiotensinogen
(a polypeptide) into angiotensin I (a decapeptide
molecule) (Fyhrquist, and Saijonmaa 2008).
Angiotensin I has no obvious biological activity and
has no obvious contractile effect on the heart and
vascular smooth muscle (Li, et al. 1979).
Angiotensin converting enzyme (ACE) can
catalyze the conversion of angiotensin I (Ang I) to
angiotensin II (Ang II). Angiotensin II is the strongest
vasoconstrictor in the angiotensin family, and it is a
polypeptide substance. It can act on the AT1 receptors
of the myocardium and vascular smooth muscle cells
to directly constrict blood vessels and cause an
increase in blood pressure (Peach, and Dostal 1990).
In addition, Ang II can also promote the secretion of
norepinephrine and aldosterone, thereby promoting
the effect of sympathetic nerves on blood vessels,
increasing the reabsorption of sodium ions and water,
increasing blood volume, and leading to increased
blood pressure (Schlaich, et al. 2005, Xanthakis, and
Vasan 2013, Yatabe, et al. 2011).
When ACE is combined with ACEI, it can make
ACE lose its catalytic activity, reduce the production
of AngⅡ, and relax blood vessels, thereby lowering
blood pressure. In addition, ACEI can also enhance
the effect of bradykinin (a substance that promotes
vasodilation), make it accumulate, and lower blood
pressure (Tom, et al. 2002).
4.3 Mode of Delivery
4.3.1 Delivery of Amlodipine
Amlodipine can be administered in many ways.
Different modes of administration have their unique
characteristics and adapt to the condition or disease
severity.
Amlodipine can be salted with benzenesulfonic
acid and maleic acid and administered orally. Tablet
is the most common oral dosage form in the market.
Although there are great differences in the activity of
S- and R- configuration, there is no significant
difference in the clinical effect between racemate and
ICHIH 2022 - International Conference on Health Big Data and Intelligent Healthcare
210
amlodipine with single S-configuration, and there are
no serious adverse reactions. Therefore, it is often
administered in the form of a racemate (Park, et al.
2006). Oral instant amlodipine tablet is suitable for
patients with dysphagia. After entering the mouth,
this tablet disintegrates rapidly and is absorbed by the
human body, but it has high requirements for the
palatability of the tablet (Fukui-Soubou, et al. 2011).
Layered tablets can release drugs at two different
release rates. The effect of oral nanoemulsion on the
target site is greatly improved compared with
ordinary oral tablets (Chhabra, et al. 2011).
In addition to oral administration, there is also a
form of intranasal administration. The biodegradable
polymer hydroxypropyl guar is used to make
amlodipine microspheres and penetrate the nasal
mucosa (Swamy, and Abbas 2011).
4.3.2 Delivery of Enalapril
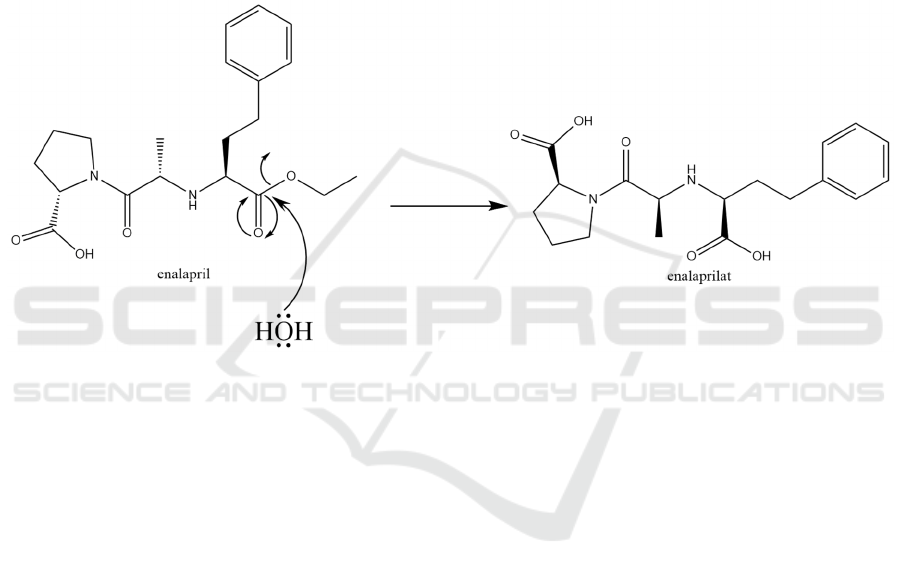
Enalapril is characterized as an oral prodrug, which is
hydrolyzed and metabolized in the body to produce
enalaprilat to produce pharmacological activity
(Patchett 1984). The process of hydrolyze and
metabolize is shown in Figure 5.
Figure 5: The prodrug enalapril is hydrolyzed and metabolized into enalapril in the body.
Enalapril is mainly administered orally. Enalapril
maleate has a low degree of oral absorption, and the
active form produced by first-pass elimination is
greatly reduced. Existing studies have improved the
bioavailability of oral enalapril in a variety of ways.
Preparation of enalapril maleate suspended
microspheres to control drug release (Abbas, and
Alhamdany 2020). The cross-linked hard gelatin
capsules are prepared, and the pulse drug delivery
mode is designed to control the drug release rate
(Tosha, et al. 2015). The oral mucosa is designed to
adhere to the double-layer tablet to allow the drug to
be absorbed through the oral mucosa to prevent the
first-pass metabolism of the drug (Shah,
Gadhethariya, and Shah 2014).
4.4 Comparison of Amlodipine and
Enalapril
Amlodipine and enalapril are two drugs with different
action mechanisms. Amlodipine belongs to
dihydropyridine calcium channel antagonist, and
enalapril belongs to angiotensin converting enzyme
inhibitor ACEI. Because of their different
mechanisms of action, they will have different effects
on patients with hypertension.
From the perspective of pharmacological action,
amlodipine can increase cardiac output, but enalapril
has no significant change in cardiac output (Murdoch,
and Heel 1991, Todd, and Heel 1986). Amlodipine
increased the glomerular filtration rate, while
enalapril did not affect the glomerular filtration rate
(Murdoch, and Heel 1991, Todd, and Heel 1986). In
known experiments, amlodipine has the effect of
antiplatelet aggregation, but enalapril has no such
effect (Hernández-Hernández, et al. 1999).
Amlodipine can decrease the demand for oxygen of
the myocardium and increase the protective effect on
the myocardium (Murdoch, and Heel 1991). Enalapril
can reverse the symptoms of cardiac hypertrophy in
patients with left ventricular hypertrophy (Todd, and
Heel 1986).
From the perspective of metabolism, the oral
absorption of amlodipine is slow, reaching the peak
of drug plasma concentration in 6 ~ 12 hours, and the
oral absorption of enalapril is rapid, reaching the peak
Progress of Amlodipine and Enalapril in the Treatment of Hypertension
211

in only one hour (Murdoch, and Heel 1991, Todd, and
Heel 1986).
From the perspective of adverse reactions, the
common adverse reactions of amlodipine include
edema, muscle spasm, and so on (Galappatthy, et al.
2016). The common adverse reaction of enalapril is
cough, which is a common adverse reaction in ACEI
(Simon, et al. 1992).
From an application point of view, the
combination of calcium channel antagonists and
ACEI in the treatment of hypertension can better
protect the cardiovascular system (Taddei 2015).
5 DISCUSSION OF DRUG
ECONOMICS
5.1 Cost of Amlodipine and Enalapril
According to the data of clincalc.com, in 2018, the
average total drug cost of amlodipine in the United
States was $24.71 per prescription, the average out-
of-pocket cost of patients was $4.79 per prescription,
and the daily treatment cost (i.e., the average cost of
each prescription divided by the number of days
required for treatment, the same below) was
$0.11/day. In the same year, the average total drug
cost of enalapril maleate (the maleate of enalapril) in
the United States was $42.88 per prescription, the
average out-of-pocket cost of patients was $11.29 per
prescription, and the daily treatment cost was
$0.24/day (Enalapril Maleate Drug Usage Statistics,
United States 2021). In contrast, the price of enalapril
maleate is higher than that of amlodipine.
Compare the prices of amlodipine in China and
the United States, unifying the purchase price of
patients as USD per tablet. Only in terms of the price
of tablets, the cost paid by Chinese patients with
hypertension is 2.3 times that of American patients
(Bai, et al. 2018).
5.2 The Number of Prescriptions and
Sales
In 2018, the number of prescriptions for amlodipine
exceeded 75 million, ranking fifth among prescription
drugs in the United States, and it has maintained its top
ten rankings in the past ten years, indicating that
amlodipine has been used in huge amounts. The
number of patients using amlodipine reached
15,851,641. Since 2008, the number of patients using
amlodipine and the number of prescriptions has been
slowly increasing (Amlodipine Drug Usage Statistics
2021).
Enalapril ranks 135th in the ranking of
prescription drugs in the United States, with more
than 5 million prescriptions and an estimated number
of patients of 1.07 million. Unlike amlodipine, since
2008, the number of enalapril prescriptions and the
number of patients has shown a downward trend, and
the ranking of commonly used prescription drugs in
the United States has also been declining in recent
years (Enalapril Maleate Drug Usage Statistics,
United States 2021).
Calcium channel blockers and β-blockers are the
most prescribed drugs for hypertension. At the same
time, in the treatment of elderly hypertensive patients,
amlodipine is the most common drug, reaching 37%
(Altaf, et al. 2014). In China, the defined daily dose
of CCB in the five years from 2007 to 2015 accounted
for about 42.8% of the five types of antihypertensive
drugs, with an average annual growth rate of about
13.2%. In China, in the five years from 2007 to 2015,
the defined daily dose of ACEI accounted for
approximately 13.3% of the five types of
antihypertensive drugs, with an average annual
growth rate of approximately 1.4% (Xu, et al. 2015).
6 CONCLUSIONS
Hypertension is widely concerned all over the world
because of its prevalence, difficulty to cure, and great
harm. The pathogenesis of hypertension is not clear,
but one of the possible trends in the research on the
pathogenesis of hypertension is integration, that is,
considering the cumulative effect of multiple factors,
such as eating habits, gene inheritance, and
microbiota. At present, the exploration of the possible
pathogenic factors of hypertension is not exhaustive.
However, based on the correlation between the
known important pathogenic factors and the
incidence rate of hypertension, a rough model can be
established, which is important for preventing and
controlling hypertension.
In the treatment of hypertension, amlodipine, a
calcium channel blocker, and enalapril, an
angiotensin converting enzyme inhibitor, have
antihypertensive effects through different
pharmacological effects. The research on the
potential pharmacological mechanism of known
drugs plays an important role in deeply understanding
the pathogenesis and developing new targeted drugs.
In recent studies on the discovery and research of new
antihypertensive mechanisms of two drugs,
amlodipine can regulate the configuration conversion
of vascular smooth muscle to reduce blood pressure,
ICHIH 2022 - International Conference on Health Big Data and Intelligent Healthcare
212

and the targets and real compounds that promote the
configuration conversion are good research objects.
For the antihypertensive drug market, the number of
patients using amlodipine to reduce blood pressure is
increasing year by year, which will promote the in-
depth study of amlodipine. For other types of
antihypertensive drugs, to occupy a larger market
share, enterprises are required to optimize the
production process and improve antihypertensive
drugs and develop drugs or dosage forms with better
efficacy, safety, and stability, and appropriate price.
REFERENCES
Abbas, A.K. and A.T. Alhamdany, Floating Microspheres
of Enalapril Maleate as a Developed Controlled
Release Dosage Form: Investigation of the Effect of an
Ionotropic Gelation Technique. Turkish journal of
pharmaceutical sciences, 2020. 17(2): p. 159-171.
Altaf, M., et al., Drug utilisation evaluation of
antihypertensives in geriatric patients in a tertiary care
hospital. 2014. 6(9): p. 261-264.
Amlodipine Drug Usage Statistics, United States, 2008 -
2018. 2021; Available from:
https://clincalc.com/DrugStats/Drugs/Amlodipine.
Bai, G., et al., Access to Antihypertensive Drugs in China.
Circulation, 2018. 138(17): p. 1777-1779.
Bers, D.M., Cardiac Sarcoplasmic Reticulum Calcium
Leak: Basis and Roles in Cardiac Dysfunction. 2014.
76(1): p. 107-127.
Burges, R. and D. Moisey, Unique pharmacologic
properties of amlodipine. American Journal of
Cardiology, 1994. 73(3): p. A2-A9.
Catterall, W.A. and T.M. Swanson, Structural Basis for
Pharmacology of Voltage-Gated Sodium and Calcium
Channels. Mol Pharmacol, 2015. 88(1): p. 141-50.
Catterall, W.A., Voltage-gated calcium channels. Cold
Spring Harb Perspect Biol, 2011. 3(8): p. a003947.
Chakraborty, S., et al., Metabolites and Hypertension:
Insights into Hypertension as a Metabolic Disorder:
2019 Harriet Dustan Award. Hypertension, 2020.
75(6): p. 1386-1396.
Chhabra, G., et al., Design and development of
nanoemulsion drug delivery system of amlodipine
besilate for improvement of oral bioavailability. Drug
Development and Industrial Pharmacy, 2011. 37(8): p.
907-916.
Coelho, M.M., et al., Enantioselectivity in Drug
Pharmacokinetics and Toxicity: Pharmacological
Relevance and Analytical Methods. Molecules, 2021.
26(11).
Cushman, D.W. and M.A. Ondetti, History of the design of
captopril and related inhibitors of angiotensin
converting enzyme. 1991. 17(4): p. 589-592.
Cushman, D.W., et al., Development and design of specific
inhibitors of angiotensin-converting enzyme. American
Journal of Cardiology, 1982. 49(6): p. 1390-1394.
Enalapril Maleate Drug Usage Statistics, United States,
2008 - 2018. 2021.
Fang, Q., et al., Amlodipine induces vasodilation via
Akt2/Sp1-activated miR-21 in smooth muscle cells. Br J
Pharmacol, 2019. 176(13): p. 2306-2320.
Flack, J.M., et al., Prevention of Hypertension and Its
Complications: Theoretical Basis and Guidelines for
Treatment. Journal of the American Society of
Nephrology, 2003. 14(suppl 2): p. S92.
Forrester, S.J., et al., Angiotensin II Signal Transduction:
An Update on Mechanisms of Physiology and
Pathophysiology. Physiological reviews, 2018. 98(3):
p. 1627-1738.
Fukui-Soubou, M., et al.,
Efficacy, Safety, and Palatability
of RACTAB® Formulation Amlodipine Orally
Disintegrating Tablets. Drugs in R & D, 2011. 11(4): p.
327-336.
Fyhrquist, F. and O. Saijonmaa, Renin-angiotensin system
revisited. Journal of internal medicine, 2008. 264(3): p.
224-236.
Galappatthy, P., et al., Leg edema with (S)-amlodipine vs
conventional amlodipine given in triple therapy for
hypertension: a randomized double blind controlled
clinical trial. BMC cardiovascular disorders, 2016.
16(1): p. 168-168.
Gao, S. and N. Yan, Structural Basis of the Modulation of
the Voltage-Gated Calcium Ion Channel Cav 1.1 by
Dihydropyridine Compounds*. Angew Chem Int Ed
Engl, 2021. 60(6): p. 3131-3137.
Hernández-Hernández, R., et al., Effects of amlodipine and
enalapril on platelet function in patients with mild to
moderate hypertension. International journal of clinical
pharmacology and therapeutics, 1999. 37(7): p. 323-
331.
Jaffe, I.A., Adverse effects profile of sulfhydryl compounds
in man. The American Journal of Medicine, 1986.
80(3): p. 471-476.
Joint Committee for Guideline, R., 2018 Chinese
Guidelines for Prevention and Treatment of
Hypertension-A report of the Revision Committee of
Chinese Guidelines for Prevention and Treatment of
Hypertension. J Geriatr Cardiol, 2019. 16(3): p. 182-
241.
Li, X.C., et al., Intratubular and intracellular renin-
angiotensin system in the kidney: a unifying perspective
in blood pressure control. Clinical science (London,
England : 1979), 2018. 132(13): p. 1383-1401.
Lu, J., et al., Amlodipine and atorvastatin improved
hypertensive cardiac hypertrophy through regulation of
receptor activator of nuclear factor kappa B
ligand/receptor activator of nuclear factor kappa
B/osteoprotegerin system in spontaneous hypertension
rats. Exp Biol Med (Maywood), 2016. 241(11): p.
1237-49.
Manosroi, W. and G.H. Williams, Genetics of Human
Primary Hypertension: Focus on Hormonal
Mechanisms. Endocrine reviews, 2019. 40(3): p. 825-
856.
Mills, K.T., et al., Global Disparities of Hypertension
Prevalence and Control: A Systematic Analysis of
Progress of Amlodipine and Enalapril in the Treatment of Hypertension
213

Population-Based Studies From 90 Countries.
Circulation, 2016. 134(6): p. 441-50.
Murdoch, D. and R.C. Heel, Amlodipine. Drugs, 1991.
41(3): p. 478-505.
Oparil, S., et al., Hypertension. Nat Rev Dis Primers, 2018.
4: p. 18014.
Park, J.-Y., et al., Pharmacokinetic and pharmacodynamic
characteristics of a new <em>S</em>-amlodipine
formulation in healthy Korean male subjects: A
randomized, open-label, two-period, comparative,
crossover study. Clinical Therapeutics, 2006. 28(11): p.
1837-1847.
Patchett, A., The chemistry of enalapril. 1984. 18(S2): p.
201S-207S.
Peach, M.J. and D.E. Dostal, The Angiotensin II Receptor
and the Actions of Angiotensin II. 1990. 16: p. S25-S30.
Rimoldi, S.F., U. Scherrer, and F.H. Messerli, Secondary
arterial hypertension: when, who, and how to screen?
Eur Heart J, 2014. 35(19): p. 1245-54.
Schlaich, M.P., et al., Angiotensin II and norepinephrine
release: interaction and effects on the heart. 2005.
23(5): p. 1077-1082.
Shah, P., Y. Gadhethariya, and S.J.T.i.D.D. Shah,
Formulation and Evaluation of Buccal Mucoadhesive
Bilayered Tablets of Enalapril Maleate. 2014. 1(1): p.
1-15.
Simon, S.R., et al., Cough and ACE Inhibitors. Archives of
Internal Medicine, 1992. 152(8): p. 1698-1700.
Stepien, O., et al., Amlodipine Inhibition of Serum-,
Thrombin-, or Fibroblast Growth Factor-Induced
Vascular Smooth-Muscle Cell Proliferation. 1998.
31(5): p. 786-793.
Swamy, N.G.N. and Z. Abbas, Preparation and in vitro
characterization of mucoadhesive hydroxypropyl guar
microspheres containing amlodipine besylate for nasal
administration. Indian journal of pharmaceutical
sciences, 2011. 73(6): p. 608-614.
Taddei, S., Combination Therapy in Hypertension: What
Are the Best Options According to Clinical
Pharmacology Principles and Controlled Clinical Trial
Evidence? American Journal of Cardiovascular Drugs,
2015. 15(3): p. 185-194.
Tang, L., et al., Structural basis for inhibition of a voltage-
gated Ca(2+) channel by Ca(2+) antagonist drugs.
Nature, 2016. 537(7618): p. 117-121.
Todd, P.A. and R.C. Heel,
Enalapril. Drugs, 1986. 31(3):
p. 198-248.
Tom, B., et al., Bradykinin potentiation by ACE inhibitors:
a matter of metabolism. Br J Pharmacol, 2002. 137(2):
p. 276-84.
Tosha, S.M., et al., Development and in vitro evaluation of
pulsatile drug delivery system of enalapril maleate.
Bangladesh Pharmaceutical Journal, 2015. 18(1): p. 66-
71.
Tsioufis, C. and C. Thomopoulos, Combination drug
treatment in hypertension. Pharmacol Res, 2017.
125(Pt B): p. 266-271.
Wei, J., et al., Factors associated with awareness, treatment
and control of hypertension among 3579 hypertensive
adults in China: data from the China Health and
Nutrition Survey. BMC Public Health, 2021. 21(1): p.
423.
Xanthakis, V. and R.S. Vasan, Aldosterone and the risk of
hypertension. Curr Hypertens Rep, 2013. 15(2): p. 102-
7.
Xu, H., et al., Trends and patterns of five antihypertensive
drug classes between 2007 and 2012 in China using
hospital prescription data. 2015. 53(6): p. 430-437.
Yatabe, J., et al., Angiotensin III Stimulates Aldosterone
Secretion from Adrenal Gland Partially via
Angiotensin II Type 2 Receptor But Not Angiotensin II
Type 1 Receptor. Endocrinology, 2011. 152(4): p.
1582-1588.
Zamponi, G.W., et al., The Physiology, Pathology, and
Pharmacology of Voltage-Gated Calcium Channels
and Their Future Therapeutic Potential. Pharmacol
Rev, 2015. 67(4): p. 821-70.
ICHIH 2022 - International Conference on Health Big Data and Intelligent Healthcare
214
