
Targeting GABA to Cure Anxiety Disorder in Various Methods
Yihui Lu
1,
†
a
,
Jiayi Yuan
2,
†
b
and Xichen Zhang
3,
†
c
1
Environmental and Life Sciences Faculty University of Southampton, Southampton, U.K.
2
Qingdao Chengyang No.1 Senior High School, Qingdao, China
3
Shanghai Shangde Experimental School, Shanghai, China
†
These authors contributed equally
Keywords: GABA, GABAA Receptor, Benzodiazepine, GABAB Receptor, Anxiety Disorder.
Abstract: The number of people suffering from anxiety disorders has risen sharply in recent years. Gamma-
aminobutyric acid (GABA) is a neurotransmitter in the central nervous system that inhibits (prevents) nerve
activity by restricting nerve transmission. A large number of studies have shown that low GABA levels or
GABA system dysfunction can cause anxiety. This article mainly summarizes the treatment and mechanism
of anxiety disorders for GABA metabolic uptake, GABAA receptor, and GABAB receptor. The metabolic
process of GABA was outlined, by using drugs that increase the expression of GAD, inhibit GABA-T, and
block GAT to increased the content of GABA and treat anxiety. The structure of GABAA receptors and how
benzodiazepine targets GABAA receptors to treat anxiety disorders were detailed. Finally, anxiety disorders
can be treated by GABAB receptor agonist baclofen and positive allosteric modulators (PAMs).
1 INTRODUCTION
Anxiety is a common unpleasant emotional state
marked by emotions of fear and dread, as well as
distinct physical, cognitive, and behavioral
symptoms. Anxiety is a normal element of one's
behavioral repertoire, and it can be useful as a
protective mechanism for increasing awareness and
response to unexpected situations. However, when it
is overly intense or frequent, or when it occurs in
inappropriate situations, it can interfere with regular
functioning and thus be called abnormal (Roy-Byrne,
2005). The people who are in this state are suffering
from an anxiety disorder. Anxiety disorder is a major
mental health problem that affects people all over the
world and the proportion of people who are suffering
from this disease are increasing. In a 2020 survey, 62
percent of respondents said they were anxious in
some way (Team 2021). And the proportion of those
who are suffering from an anxiety disorder is
increasing year after year. The global prevalence of
all mental disorders increased by 50% between 1990
and 2013, from 416 million to 615 million persons
a
https://orcid.org/0000-0003-3963-7140
b
https://orcid.org/0000-0001-8315-7609
c
https://orcid.org/0000-0003-2712-1091
(World Health Organizations 2021). Besides,
Anxiety disorders impacted around 273 million
people worldwide in 2010. In 2017, an estimated 264
million people worldwide suffered from anxiety
(Figure 1). Thus, there are such a large number of
people are struggling with this disease, our research
aims to provide a great number of meaningful
methods to relieve symptoms to some extent.
Figure 1: The number of mental disorder patients (World
Health Organizations, 2021).
Gamma-aminobutyric acid (GABA), which is
found in more than a third of central nervous system
366
Lu, Y., Yuan, J. and Zhang, X.
Targeting GABA to Cure Anxiety Disorder in Various Methods.
DOI: 10.5220/0011208900003443
In Proceedings of the 4th International Conference on Biomedical Engineering and Bioinformatics (ICBEB 2022), pages 366-373
ISBN: 978-989-758-595-1
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

(CNS) synapses, is one of the most widely distributed
neurotransmitters in the brain (Peter P Roy-Byrne
2021). The etiology of anxiety disorders is related to
changes in the GABA system. Allosteric sites on the
GABA receptor allow for precise modulation of the
level of inhibition of neurons in the amygdala, and
these sites are the molecular targets of the most
common types of anxiolytic medicines (Gauthier
2015).
To explain the content of GABA and the
treatment of anxiety, we first detailed the metabolic
process of GABA by employing medications that
boost the expression of GAD, inhibit GABA-T, and
block GAT. The structure of GABAA receptors was
then discussed, as well as how benzodiazepines target
GABAA receptors to treat anxiety disorders. Finally,
anxiety disorders can be treated using the GABAB
receptor agonist baclofen and positive reinforcement.
2 GABA METABOLISM
GABA transport, sequestration, synthesis, and
degradation are all mediated by various specific
molecular pathways (Roth 2021). GABA metabolism
and uptake both play a role in the factors that
influence anxiety pathogenesis (Kalueff, 2007).
The enzyme glutamic acid decarboxylase (GAD)
and the cofactor pyridoxal phosphate are used to
convert glutamate, the principal excitatory
neurotransmitter, into GABA in the GABAergic
neurons of the central nervous system. Only cells that
employ GABA as a neurotransmitter produce GAD.
GABA is packaged into vesicles by vesicle GABA
transporters (VGAT), released into the synaptic cleft,
and diffused across the cleft to the target receptors
located on the postsynaptic surface when the
presynaptic neuron is depolarized. Furthermore,
Presynaptic terminals and surrounding glial cells can
resorb GABA released into the synapse cleft for a
variety of objectives. Membrane GABA transporters
(GAT) allow GABA to be re-used in presynaptic
terminals, however GABA in glial cells is converted
to succinic semialdehyde by GABA-T and cannot be
re-synthesised in this compartment because glia lack
GAD. Through a circuitous route that involves the
Krebs cycle, GABA can eventually be recovered
from this source. GABA is transformed to glutamine
in glia by the GABA shunt, which subsequently
transports glutamine back to presynaptic neurons,
where glutamine is converted to glutamate by
glutaminase. Anxiety is dependent on GABA
metabolism, which may be disrupted in the
pathophysiology (Siegel 1999).
2.1 Glutamic Acid Decarboxylase
The enzyme glutamic acid decarboxylase (GAD) is
responsible for converting glutamate to GABA. GAD
comes in two molecular forms in the mammalian
brain: Glutamic acid decarboxylase 67 (GAD67) and
Glutamic acid decarboxylase (GAD65), which are
produced by two independently regulated genes,
GAD1 and GAD2, respectively (Tillakaratne, 1995)
(Hettema 2006). In terms of intraneuronal expression,
GAD65 appears to be more restricted to axon
terminals, whereas GAD67 appears to be more
equally distributed throughout the neuron.
Furthermore, the active cofactor-bound holoenzyme
form of GAD67 makes up nearly all of the enzyme,
whereas majority of GAD65 is found as a reserve
pool of inactive apoenzyme. One study suggests that
acute stress may enhance GAD67 production,
whereas chronic stress may increase GAD65
availability (Hettema 2006).
Mice missing a short 65-kDa GAD isoform
(GAD65), which is responsible for fine-tuning
GABAergic neurotransmission, GABA levels were
lower and anxiety levels were higher, whereas AD
reboxetine increased GAD65 expression in the
septum of stressed rats. Recent research has
discovered a relationship has been discovered
between a GAD65 gene polymorphism and anxiety-
related behavioral inhibition in childre, as well as
lower levels of GAD65 and GAD67 in the prefrontal
cortex and cerebellum in depressed people (Kalueff
2007). In human studies, neurotic patients and those
with mood disorders showed lower GAD plasma
activity. In a small family-based study of children,
researchers discovered a small link between GAD2
and behavioral inhibition, and anxiety-related traits
(Hettema 2006). Therefore, a decrease in GAD level
will result in less synthesis of GABA, which will lead
to increased anxiety.
2.2 GABA Transaminase
Another important enzyme in GABA turnover is
GABA transaminase (GABA-T), which catabolizes
GABA (Kalueff 2007). GABA-T inhibition increases
brain levels of the main inhibitory neurotransmitter
GABA, which has been linked to a range of
functional consequences, including behavioral
alterations (Sherif 1995). Its inhibitors, such as
Vigabatrin (gamma-vinyl GABA, GVG), can raise
brain GABA levels by irreversibly blocking GABA-
T. In the plus-maze test, treatment with vigabatrin at
a dose of 250 mg/kg, i.p. lowered anxiety levels in
both groups of differentially housed rats
Targeting GABA to Cure Anxiety Disorder in Various Methods
367

(Tillakaratne, 1995). Inhibiting GABA-T, elevating
brain GABA levels, producing anxiolytic-like effects
in animals, and modulating glutamate and
catecholamine neurotransmission are all effects of the
AD phenelzine (In humans, it is beneficial in the
treatment of social anxiety and panic disorders)
(Kalueff 2007). In various experimental models,
phenylethylidenehydrazine (PEH), an derivative of
the monoamine oxidase inhibitor beta-phenylmethyl
hydrazine (phenelzine), which inhibits the gamma-
aminobutyric acid (GABA) catabolic enzyme
GABA-transaminase and elevates GABA levels in
the brain and has strong anxiolytic properties (Duffy
2004).
2.3 GABA Transporter
GABA transporters (GATs) are important molecules
in the transfer of GABA and are found on the
membrane of cells. At the synapse, GATs can
regulate the duration and intensity of GABAergic
activity by reabsorbing GABA. GAT1, GAT2,
GAT3, and GAT4 are among the GABA transporter
subtypes discovered. GAT1 is the most common
subtype in the brain, and it can be found in both
synaptic and extrasynaptic sites (Liu 2007). It's also
in charge of maintaining 75 percent of synaptic
GABA concentration and delivering it to GABAA
receptors to initiate receptor-mediated postsynaptic
neuron inhibition. GAT1 is largely involved in
GABA binding and transport from the cytoplasm to
the extracellular space (reverse mode) and back
(forward mode). As a result, if GAT1 is
malfunctioning, communication with postsynaptic
GABA receptors may be delayed (Zafar 2018).
GAT1 deficiency results in increased extracellular
GABA levels and GABAA receptor overactivation.
Tremor, ataxia, and nervousness are all symptoms of
GAT1 deficiency, according to behavioral tests.
GAT1 is involved in the development of anxiety
disorders, according to several behavioral tasks such
as the tail-suspension test, forced swim test, and
open-field test. Tiagabine hydrochloride is a selective
GABA reuptake inhibitor that blocks GAT1 and
hence raises GABA tone. It is used to treat anxiety
disorders. For decades, GAT1 has been recognized as
a potential therapeutic target due to its critical role in
the GABAergic transport mechanism (Liu 2007).
2.4 Neuropeptide
Neuropeptides are small proteins that operate as
neuronal signaling molecules and have a role in a
variety of brain functions, including analgesia,
reward systems, social behaviors, learning, and
memory (Garakani 2020). It's also important to
consider neuropeptides' role in modulating
GABAergic function and anxiety/depression
interplay. Melatonin, for example, has been shown in
animals to have both anxiolytic and anti-properties
(Kalueff 2007). Melatonin improved GABAergic
inhibitory transmission by increasing the amplitude
and frequency of GABAergic mIPSCs. As a result,
we've discovered that melatonin boosts the
GABAergic system's performance (Cheng 2021).
Cholecystokinin antagonists are neuropeptides that
regulate the GABAergic system and is implicated in
both anxiety pathogenesis and treatment, have been
found to have similar properties (Kalueff 2007).
Oxytocin has been associated to high anxiety in
healthy individuals and has been proven to have a
good influence on emotion modulation in healthy
adults. Animal research imply that oxytocin has
anxiolytic qualities, however human studies suggest
that oxytocin can produce acute anxiety. In animal
models, arginine vasopressin (AVP) has been linked
to anxiety, anxiolytic properties may be found in
vasopressin V1a and V1b receptor antagonists.
Despite the fact that neuropeptides appear to be a
promising new therapy option for anxiety disorders,
no specific treatment candidates have been identified
(Garakani 2020).
3 GABAA RECEPTOR
The GABAA receptor is one of the most common
types of receptor for the inhibitory neurotransmitter
y-aminobutyric acid (GABA) (OLSEN, Tobin 1990).
GABAA receptors are made up of five protein
subunits that bridge a lipid bilayer to produce a
cylindrical structure. There is a ligand-gated ion
channel in the center. (Roy-Byrne 2005) (OLSEN,
Tobin 1990) (Bruce 2021). It allows chloride ions to
move in and move out, using this mechanism, it can
regulate excitability. And there are many agonists and
antagonists to accommodate the process of GABA
binding with GABAA receptor, which plays role in
curing anxiety disorders, like benzodiazepines.
3.1 The Structure of GABAA Receptor
Electric organs of electric rays and eels, as well as
vertebrate skeletal muscle, contain GABAA
receptors. All are pentameric oligomers with a mass
of around 250 kilodaltons (kDa) and four different
types of subunits, each with a mass of about 50 kDa.
GABAA receptors in the central nervous system of
ICBEB 2022 - The International Conference on Biomedical Engineering and Bioinformatics
368

vertebrates appear to be made up of solely a, β and γ
polypeptides, with a total of four or five subunits. The
oligomeric subgroups differ in terms of
developmental stage, tissue type, and brain region, as
well as pharmacological qualities (OLSEN, Tobin
1990). As a result, the GABAA receptor is a
pentamer. It's also a heteropentamer, according to a
number of studies. There are several molecular
families of subunits that have been found, including
those α with 6 isoforms, β with 3 isoforms, γ with 3
isoforms, θ with 1 isoform, and ρ with 3 isoforms
(Tallman 2002). Furthermore, when viewed from the
extracellular space, the receptor complex is known to
consist of 2 α subunits alternating with 2 β subunits
ordered b-a-b-g-a in a clockwise orientation and a
single γ subunit (Sherif 1995) (Duffy 2004) (Liu
2007) (Zafar 2018). The tri-heteromeric receptor,
which consists of two α, two β, and one γ subunit, in
the vertebrate brain, is the most common subunit
combination, despite the fact that there are multiple
possible layouts (Chang 1996) (Farrar 1999) (Tretter
1997).
3.2 Ligand-gated Channel
There are two GABA binding sites in each receptor
complex, but only one benzodiazepine binding site.
The GABA binding sites are found at the junction of
the two β subunit pairs that alternate, while the lone
benzodiazepine binding site is found at the crossroads
of the single α and γ subunit pairings (Roy-Byrne
2005). There are many different subtypes to choose
from due to the numerous diverse subunits and their
arrangements, each with its own affinity for GABA,
chloride channel kinetics, and affinity for different
benzodiazepine ligands (Roy-Byrne 2005). The vast
majority of these receptor complexes, according to
evidence, are made up of α1 subunit in combination
with β2 and γ2 subunits (Mohler HF 2001). The α2 or
α3 receptor subtype is likely to be responsible for
benzodiazepine sedation (Rudolph U 1999) (Lydiard
2003).
3.3 GABAA Receptor Channel
There are GABAA receptors play a critical role in
balancing excitatory transmission. When activated by
GABA, the protein subunits undergo conformational
changes, resulting in the brief development of a
channel along the cylinder's axis via which chloride
ions can flow from the outside to the interior (Kalueff
2007). The chloride ions flowing into the cytoplasm
will cause hyperpolarization, so GABA is considered
an inhibitory neurotransmitter. As a result, GABA is
known to counteract the excitatory neurotransmitter
glutamate's effect (Tillakaratne 1995).
3.4 Pharmacology
The GABA-benzodiazepine receptor, also known as
the GABAA receptor or the benzodiazepine receptor,
may play a role in the pathophysiology of anxiety as
well as its treatment (Figure 2). GABAA receptors
are important since they are where benzodiazepines
act. It refers to a complex glycoprotein that has
binding sites for a variety of benzodiazepine drugs
with potent anxiolytic properties (Roy-Byrne 2005).
The reason why benzodiazepine can be used to treat
anxiety disorder on GABAA receptor is that
benzodiazepines allosterically modulated GABAA
receptors, which are used for their sedative,
anxiolytic, anticonvulsant, and muscle relaxant
actions (Rudolph 2018). To be specific, the
neurotransmitter GABA opens the chloride channel
when it binds to GABAA receptor while
benzodiazepines control this opening (Sigel 2018).
Furthermore, benzodiazepines influence this channel
opening triggered by either agonist binding site (Baur
2005).
Figure 2: The Structure of GABAA receptor.
Benzodiazepines are effective as a drug to treat
anxiety disorder when they bind with GABAA
receptors containing specific subunits. The most
sophisticated drugs, which target GABAA receptors
with α2 and α3 (positive allosteric modulation) and
α5 subunits (negative allosteric modulation), are now
being evaluated in clinical trials for anxiolytic effects,
and using one subunit (negative allosteric
modulation) to avoid functional effects at GABAA
receptors, while using only α1 subunit to avoid
functional effects at GABAA receptors (Rudolph
2018). In addition to their anxiolytic effect, which is
Targeting GABA to Cure Anxiety Disorder in Various Methods
369
mediated by α2 and potentially also by α3-containing
GABAA receptors, benzodiazepines exhibit sedative
properties mediated by α1-containing GABAA
receptors (Rudolph, 2018). Benzodiazepines are only
sensitive to receptor assemblies that have an a1
subunit next to g2 (Minier 2004). As a result, when,
benzodiazepines combine with different sub-type of
GABAA receptors, they have sedative, hypnotic,
muscle relaxant, and anticonvulsant properties, with
extremely minimal risk of overdosing (Sigel 2018).
4 GABAB RECEPTOR
The GABAB receptor, which is a G-protein-coupled
receptor, suppresses adenylate cyclase activity and
mediates synaptic inhibition's gradual and sustained
component (Bowery N 2004). GABAB receptors,
highly expressing in the limbic systemare, were
found in almost all neuronal cells, especially GABAB
(1) receptor. GABAB (1) and GABAB (2) are two
subunits of the GABAB receptor that heterodimerize
to create the functional GABAB receptor. The
orthosteric ligand binding site is found in the
GABAB (1) subunit, whereas the GABAB (2)
subunit, responsible for G-protein activation,
contains positive allosteric modulator binding sites
(Gassmann M 2012).
4.1 Baclofen
Baclofen was a powerful and selective agonist of
GABAB receptor in 1980, and it was shown that
baclofen inhibited neurotransmitter release in the
central nervous system when it acted on it. GABAB
receptor agonists, including baclofen, have a lot of
preclinical evidence that they could be useful in the
treatment of anxiety disorders (Felice D 2016)
(Vinkers CH 2010). Baclofen, the first known GABA
derivative, was created in 1962 by combining a
halogenated phenylring with carbon to create a
molecule that could cross the blood-brain barrier
(BBB). The functions of baclofen are mainly used as
a muscle relaxant and antispastic. Research of
baclofen revealed a number of drawbacks: it couldn't
passively penetrate the BBB and had a short duration
of action and rapid tolerance development in the
patient (Felice 2020).
Efforts have been made to obtain baclofen
analogs, but no superior drug options have emerged.
One of these compounds was phenibut, which was
made by merely removing the chlorine atom from
baclofen. The molecule has anxiolytic and nootropic
properties, however it was quickly determined that it
was not GABAB receptor-selective. Other
experiments attempted but failed, to rigidify the
baclofen structure by inserting groups like ethylene
and propylene. Some efforts to obtain baclofen
analogs resulted in clinically authorized medications,
such as pregabalin, vigabatrin, and gabapentin,
although these were discovered to act through distinct
processes and bind receptors other than the GABAB
receptor. Although some inconsistent outcomes have
been reported, baclofen treatment has been
demonstrated to attenuate anxiety-like behavior in
numerous rat models and mouse models. For
example, one study found that baclofen was useful in
the Vogel conflict test, whereas another found that it
had no impact (Lu Y 2016). The side effects of
Baclofen included muscular relaxation,sedation,
vertigo , somnolence, and hypothermia (Agabio
2013). PAMs, one of GABAB receptors, have a lower
risk of receptor desensitization/tolerance as compared
to baclofen, which was typical GABAB receptor
agonists.
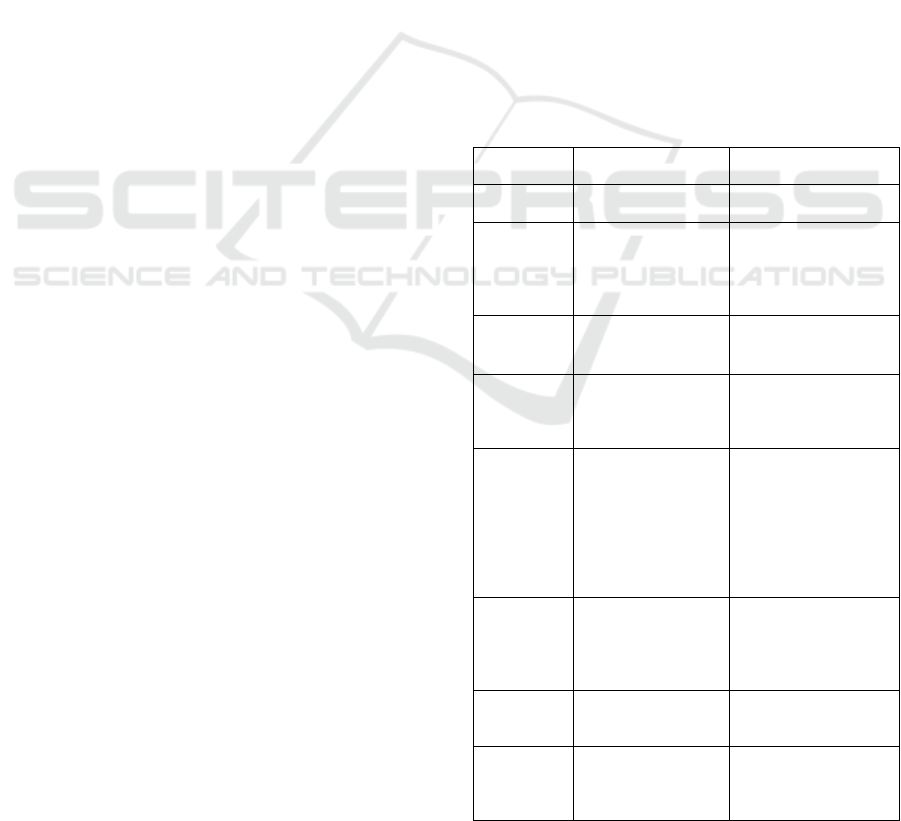
Table 1: The typical medicine, target receptor, mechanism
and side-effects.
Name of
Medicine
Target
receptor/Mechanism
Side-effect(s)
Reboxetine Increased GAD65
expression
Dry mouth,
Constipation.
Vigabatrin GABA-T inhibitor,
raise brain
GABA levels by
irreversibly blocking
GABA-T
Problems walking or
feeling uncoordinated,
feeling dizzy, shaking,
joint pain.
Phenylethyli
denehydrazin
e (PEH)
Inhibit GABA-T No clinical trials, thus
no known side-effect.
Tiagabine
hydrochlorid
e
Selective GABA-
reuptake inhibitor,
increases GABA tone
via GAT1 blockade
Inability to concentrate,
dizziness, drowsiness,
nervousness, irritability.
Antagonists
of
cholecystoki
nin
Blocks the receptor
sites for the peptide
hormone
cholecystokinin
(CCK), a
neuropeptide that
modulates the
GABAergic system
Nausea, vomiting,
constipation,
drowsiness.
Benzodiazepi
ne
GABAA Dependence, rebound
anxiety, memory
impairment, and
discontinuation
syndrome.
Baclofen GABAB Short duration of action
and rapid tolerance
development.
Positive
Allosteric
modulators
(PAMs)
GABAB Addiction liability and
respiratory depression.
ICBEB 2022 - The International Conference on Biomedical Engineering and Bioinformatics
370

4.2 Positive Allosteric Modulators
(PAMs)
Compared with receptor agonists such as baclofen,
PAMs have several advantages: (1) PAM binding
causes receptor potentiation rather than direct
activation; (2) When the allosteric binding site is
saturated, the target receptor will not be down-
regulated or over-stimulated (Gjoni T 2008) (Gjoni
2009). GABAB receptor agonists, such as PAMs,
will produce anxiolytic-like effects, whereas
defunction of GABAB receptor (in GABAB (1) and
GABAB (2) animals models) will produces
anxiogenic-like effects. Losing the function of either
the GABAB (1a, 2) or GABAB (1b, 2) receptor
subunit isoform alone, however, exhibited no effect
on anxiety-like behavior in these mice, most likely
because they still retained functioning GABAB
receptors (GABAB(1b,2) or GABAB(1a,2),
respectively). GABAB receptor antagonists have an
unknown effect on anxiety, however they appear to
be anxiolytic in some situations, similar to
agonists/PAMs. The exact mechanism of the
anxiolytic effects of GABAB receptor antagonists
and agonists/PAMS, which should have opposing
pharmacological actions, are still unknown. This
could be because GABAB receptors are located both
pre- and post-synaptically, and medicines' efficacy
and timings may differ (Freyd T 2017). Despite this,
data suggested that the GABAB receptor could be a
promising therapeutic target for anxiety disorders.
Moreover, the typical treatments for anxiety disorders
are as long as their respective characteristics, as
shown in Table 1.
5 CONCLUSIONS
There are various methods of anxiety treatment by
using GABA. Includes modulate GABA metabolism,
regulating GABAA and GABAB receptor. There are
several ways of GABA regulations such as GAD,
which could increase the expression of GABA-
related gene and increase the amount of GABA, then
the number of receptor increases which cause anxiety
to be inhibited. The second way is to use a GABA
transporter, which could inhibit the metabolism of
GABA, then the number of GABA increases, and
result in the rise of GABA level. GAT also plays an
important role, which regulates the duration and
intensity of GABAergic activity presynaptically and
postsynaptically. These are the method that
introduced in modulating GABA metabolism.
GABAA is crucial, and the main therapeutic target in
anxiety disorder. Benzodiazepine is an essential part
of anxiety. It reacts and combines with specific
subunits in the GABAA receptor which ultimately
reduces anxiety. GABAB is also a significant but less
developed receptor to cure anxiety. Baclofen which is
a GABAB agonist and PAM increases the effect of
enzymes which could ultimately decrease the level of
anxiety. However, recent studies and research shows
there is a sea of side effects and obstacles during
therapy. As benzodiazepine can cause sleep and
baclofen had a short duration of action and rapid
tolerance development. Fixing these problems is the
real question that is facing and hope to fix in the
future.
REFERENCES
Agabio R, Preti A, Gessa GL (2013) Efficacy and
tolerability of baclofen in substance use disorders: a
systematic review. Eur Addict Res 19:325–345.
Baur, R. and Sigel, E. (2005) Benzodiazepines affect
channel opening of GABAA receptors induced by
either agonist binding site. Mol. Pharmacol. 67, 1005–
1008.
Bowery N, Enna SJ, Olsen RW (2004) Six decades of
GABA. Biochem Pharmacol 68:1477–1478 Car H,
Wisniewska RJ (2006) Antidepressant-like effects of
baclofen and LY367385 in the forced swim test in rats.
Pharmacol Rep 58:758–764.
Chang Y, Wang R, Barot S, Weiss DS. 1996. Stoichiometry
of a recombinant GABAA receptor. The Journal of
Neuroscience 16:5415–5424.
Cheng, X. P., Sun, H., Ye, Z. Y., & Zhou, J. N. (2012).
Melatonin modulates the GABAergic response in
cultured rat hippocampal neurons. Journal of
pharmacological sciences, 119(2), 177–185.
Duffy, S., Nguyen, P. V., & Baker, G. B. (2004).
Phenylethylidenehydrazine, a novel GABA-
transaminase inhibitor, reduces epileptiform activity in
rat hippocampal slices. Neuroscience, 126(2), 423–432.
Duffy, S., Nguyen, P. V., & Baker, G. B. (2004).
Phenylethylidenehydrazine, a novel GABA-
transaminase inhibitor, reduces epileptiform activity in
rat hippocampal slices. Neuroscience, 126(2), 423–432.
https://doi.org/10.1016/j.neuroscience.2004.03.007.
Farrar SJ, Whiting PJ, Bonnert TP, McKernan RM. 1999.
Stoichiometry of a ligand-gated ion channel determined
by fluorescence energy transfer. Journal of Biological
Chemistry 274:10100–10104.
Felice D, O’Leary OF, Cryan JF (2016) Targeting the
GABAB receptor for the treatment of depression and
anxiety disorders. In: Colombo G (ed) GABAB
receptor the receptors, vol 29. Humana Press, Cham.
Felice, D., Cryan, J. F., & O'Leary, O. F. (2020). GABAB
Receptors: Anxiety and Mood Disorders. Current
topics in behavioral neurosciences,
10.1007/7854_2020_171. Advance online publication.
Targeting GABA to Cure Anxiety Disorder in Various Methods
371

Freyd T, Warszycki D, Mordalski S, Bojarski AJ, Sylte I,
Gabrielsen M (2017) Ligand-guided homology
modelling of the GABAB2 subunit of the GABAB
receptor. PLoS One 12:e0173889. FNM Surname
(2018). Article Title. Journal Title, 10(3), 1–10.
Conference Name: ACM Woodstock conference
Garakani, A., Murrough, J. W., Freire, R. C., Thom, R. P.,
Larkin, K., Buono, F. D., & Iosifescu, D. V. (2020).
Pharmacotherapy of Anxiety Disorders: Current and
Emerging Treatment Options. Frontiers in psychiatry,
11, 595584.
https://doi.org/10.3389/fpsyt.2020.595584.
Gassmann M, Bettler B (2012) Regulation of neuronal
GABAB receptor functions by subunit composition.
Nat Rev Neurosci 13:380–394.
Gauthier, I., & Nuss, P. (2015). Anxiety disorders and
GABA neurotransmission: a disturbance of
modulation. Neuropsychiatric Disease and Treatment,
165. doi: 10.2147/ndt.s58841.
Gjoni T, Urwyler S (2008) Receptor activation involving
positive allosteric modulation, unlike full agonism,
does not result in GABAB receptor desensitization.
Neuropharmacology 55:1293–1299.
Gjoni T, Urwyler S (2009) Changes in the properties of
allosteric and orthosteric GABAB receptor ligands
after a continuous, desensitizing agonist pretreatment.
Eur J Pharmacol 603:37–41.
Hettema, J. M., An, S. S., Neale, M. C., Bukszar, J., van
den Oord, E. J., Kendler, K. S., & Chen, X. (2006).
Association between glutamic acid decarboxylase
genes and anxiety disorders, major depression, and
neuroticism. Molecular psychiatry, 11(8), 752–762.
https://doi.org/10.1038/sj.mp.4001845.
Kalueff, A. V., & Nutt, D. J. (2007). Role of GABA in
anxiety and depression. Depression and anxiety, 24(7),
495–517. https://doi.org/10.1002/da.20262.
Liu, G. X., Cai, G. Q., Cai, Y. Q., Sheng, Z. J., Jiang, J.,
Mei, Z., Wang, Z. G., Guo, L., & Fei, J. (2007).
Reduced anxiety and depression-like behaviors in mice
lacking GABA transporter subtype 1.
Neuropsychopharmacology: official publication of the
American College of Neuropsychopharmacology,
32(7), 1531–1539.
Liu, G. X., Cai, G. Q., Cai, Y. Q., Sheng, Z. J., Jiang, J.,
Mei, Z., Wang, Z. G., Guo, L., & Fei, J. (2007).
Reduced anxiety and depression-like behaviors in mice
lacking GABA transporter subtype 1.
Neuropsychopharmacology: official publication of the
American College of Neuropsychopharmacology,
32(7), 1531–1539.
https://doi.org/10.1038/sj.npp.1301281.
Lu Y, Li CJ, Chen C, Luo P, Zhou M, Li C, Xu XL, Lu Q,
He Z, Guo LJ (2016) Activation of GABAB2 subunits
alleviates chronic cerebral hypoperfusion-induced
anxiety-like behaviours: a role for BDNF signalling and
Kir3 channels. Neuropharmacology 110:308–321.
Lydiard RB. The role of GABA in anxiety disorders. J Clin
Psychiatry 2003;64(suppl 3):21–27.
Minier, F. and Sigel, E. (2004) Positioning of a subunit
isoforms confers a functional signature to
GABAAreceptors. Proc. Natl. Acad. Sci. U. S. A. 101,
7769–7774.
Mohler HF, Crestani D, Rudolph U. GABAA receptor
subtypes: a new pharmacology. Curr Opin Pharmacol
2001; 1: 22–25.
Olsen, R. W., & Tobin, A. J. (1990). Molecular biology of
GABAA receptors. FASEB journal: official
publication of the Federation of American Societies for
Experimental Biology, 4(5), 1469–1480.
https://doi.org/10.1096/fasebj.4.5.2155149
R. Bruce, L. (2021). Retrieved 10 September 2021, from
https://www.psychiatrist.com.
Roth, F. C., & Draguhn, A. (2012). GABA metabolism and
transport: effects on synaptic efficacy. Neural
plasticity, 2012, 805830.
https://doi.org/10.1155/2012/805830.
Roy-Byrne P. P. (2005). The GABA-benzodiazepine
receptor complex: structure, function, and role in
anxiety. The Journal of clinical psychiatry, 66 Suppl 2,
14–20.
Rudolph U, Crestani F, Benke D, et al. Benzodiazepine
actions mediated by specific gamma-aminobutyric
acid(A) receptor subtypes. Nature 1999; 401:796–800.
Rudolph, U., & Knoflach, F. (2011). Beyond classical
benzodiazepines: novel therapeutic potential of
GABAA receptor subtypes. Nature reviews. Drug
discovery, 10(9), 685–697.
Sherif, F., & Oreland, L. (1995). Effect of the GABA-
transaminase inhibitor vigabatrin on exploratory
behaviour in socially isolated rats. Behavioural brain
research, 72(1-2), 135–140.
Sherif, F., & Oreland, L. (1995). Effect of the GABA-
transaminase inhibitor vigabatrin on exploratory
behaviour in socially isolated rats. Behavioural brain
research,72(1-2),135–140.
https://doi.org/10.1016/0166-4328(96)00047-2.
Siegel GJ, Agranoff BW, Albers RW, Basic
Neurochemistry: Molecular, Cellular and Medical
Aspects. 6th edition, 1999.
Sigel, E., & Ernst, M. (2018). The Benzodiazepine Binding
Sites of GABAA Receptors. Trends in pharmacological
sciences, 39(7), 659–671.
Tallman JF, Casella J, Kehne J. Mechanism of action of
anxiolytics. In: Davis KL, Charney D, Coyle JT, et al,
eds. Neuropsychopharmacology: The Fifth Generation
of Progress. Philadelphia, Pa: Lippincott Williams &
Wilkins; 2002:993–1008.
Team, S. (2021). Anxiety stats in the U.S. Retrieved 26
September 2021, from: https://www.singlecare.com.
Tillakaratne, N. J., Medina-Kauwe, L., & Gibson, K. M.
(1995). Comparative biochemistry and physiology.
Part A, Physiology, 112(2), 247–263.
https://doi.org/10.1016/0300-9629(95)00099-2.
Tretter V, Ehya N, Fuchs K, Sieghart W. 1997.
Stoichiometry and assembly of a recombinant GABAA
receptor subtype. The Journal of Neuroscience
17:2728–2737.
Vinkers CH, Cryan JF, Berend Olivier B, Groenink L
(2010) Elucidating GABAA and GABAB receptor
functions in anxiety using the stress-induced
ICBEB 2022 - The International Conference on Biomedical Engineering and Bioinformatics
372

hyperthermia paradigm: a review. Open Pharmacol
J:1–14.
World Health Organizations Says Being Trans Is Not A
Mental Disorder. (2021). Retrieved 27 September
2021, from https://outrightinternational.org.
Zafar, S., & Jabeen, I. (2018). Structure, Function, and
Modulation of γ-Aminobutyric Acid Transporter 1
(GAT1) in Neurological Disorders: A
Pharmacoinformatic Prospective. Frontiers in
5chemistry, 6, 397.
Zafar, S., & Jabeen, I. (2018). Structure, Function, and
Modulation of γ-Aminobutyric Acid Transporter 1
(GAT1) in Neurological Disorders: A
Pharmacoinformatic Prospective. Frontiers in
5chemistry, 6, 397.
https://doi.org/10.3389/fchem.2018.00397.
Targeting GABA to Cure Anxiety Disorder in Various Methods
373
