
mHealth Use in Healthcare Facilities: Raising Awareness in Data
Protection, Privacy and Safety
Lilian G. Motti Ader
1,2 a
, Bróna MacEntee
1
, Kristina Rutkauskaite
1
, Nutsa Chichilidze
1
,
Dylan Kearney
1
, Sean A. Lynch
1
, Katie Crowley
1,2 b
and Ita Richardson
1,2 c
1
Dept. Computer Science and Information Systems, University of Limerick, Limerick, Ireland
2
Lero Research Centre, University of Limerick, Limerick, Ireland
sean.a.lynch99@gmail.com, krisiiister@gmail.com, katie.crowley@ul.ie, ita.richardson@lero.ie
Keywords: Mobile Devices, Healthcare, mHealth, Medical Device, Cyber Security, Privacy, Data Protection.
Abstract: During the COVID-19 pandemic, many patients and healthcare professionals embraced the possibility of
using available mobile devices and applications, exploring the opportunities to reduce the burden on strained
services. However, despite strict surveillance under the European GDPR or Medical Device (MD) regulations,
users are considered to be primarily responsible for verifying that their application of choice is approved and
certified. We searched academic and grey literature and discuss some of the challenges related to the use of
personal devices and mobile applications for health and medical purposes. Our position is that policies and
technologies should be more considerate of users’ behaviour, which includes use of non-medical software for
medical purposes, and situations where users seem to choose usability over safety.
1 INTRODUCTION
As the COVID-19 pandemic increased the burden in
healthcare, staff, patients and the public turned to
mobile technologies to improve efficiency in
resources, patient care, information and
communication. This raised questions and challenges
relating to the use of personal devices, mainstream
applications and even regulated mobile applications
for health in healthcare facilities.
In Europe, existing regulations for medical
devices and software (MDR, EU Regulation
2017/745), as well as for Data Protection (GDPR, EU
Regulation 2016/679) have been recently reviewed
(2017, 2018, 2020 and 2021) and their extended
applications provide a reassuring framework to
protect personal data and privacy of users. However,
recent news in Ireland, regarding a cyber-attack on
the national health service and a major fine for
WhatsApp (discussed in Section 2), drew public
attention to the issues related to the use of
technologies for health or medical purposes. The risks
a
https://orcid.org/0000-0003-2952-7765
b
https://orcid.org/0000-0003-3596-4363
c
https://orcid.org/0000-0002-5493-2837
of using smartphones in hospital settings may extend
way beyond the control of regulatory agencies and
institutions’ policies.
We searched the literature on the use of mobile
devices and applications in hospital and healthcare
facilities to identify risks associated with data
protection, privacy, and safety, for both patients and
healthcare staff, that might have been overlooked and
need further attention. In this position paper, we
highlight some challenges related to the use of
personal devices and mobile applications for health
and medical purposes. We present our findings
following the People Policy Technology model (PPT)
proposed by Schlarman (Schlarman, 2001), aligning
social and technical dimensions of cybersecurity.
Then, we discuss our proposal on possible solutions
to address risks related to users’ behaviour (e.g.
contamination, misuse, lack of awareness), use of
non-regulated health-related apps (e.g. wellness and
fitness apps, websites), and poor design (e.g. lack of
transparency, difficulties in running user tests or
clinical trials, access to specialized databases).
Ader, L., MacEntee, B., Rutkauskaite, K., Chichilidze, N., Kearney, D., Lynch, S., Crowley, K. and Richardson, I.
mHealth Use in Healthcare Facilities: Raising Awareness in Data Protection, Privacy and Safety.
DOI: 10.5220/0010854100003123
In Proceedings of the 15th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2022) - Volume 5: HEALTHINF, pages 565-572
ISBN: 978-989-758-552-4; ISSN: 2184-4305
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
565

2 MOTIVATION AND GOALS
Europe has strong legislation for data protection and
privacy, with additional reinforcement when it
concerns medical information. For example, in
Ireland, the Heath Products Regulatory Authority –
HPRA, is implementing the new EU MDR/IVDR
regulation – which is now a legal requirement rather
than a directive. Having been postponed due to the
COVID-19 pandemic and its implications, the new
regulations have become fully applicable since the
26
th
May 2021
1
. This results in ‘clearer requirements
for clinical data on medical devices’ and ‘more
specific product requirements’, including technical
standards.
However, users are considered primarily
responsible for verifying that the apps of their choice
are approved and certified. There is some evidence
that more actions are needed to protect users. We
outline below two events in Ireland that recently
featured in worldwide news.
In December 2018 the Data Protection
Commission (DPC) initiated an investigation into the
social media and instant messaging application
WhatsApp. In September 2021, the investigation was
concluded showing lack of compliancy with the
GDPR transparency obligations. WhatsApp was
imposed a fine of €225 million
2
. We highlight this
example as it raises concerns on the use of
applications not compliant with MDR (non-MD apps)
in healthcare settings. Because of the convenience of
using general publicly available applications, the
Ireland Health Service Executive (HSE) had
approved, as an ‘exceptional provision’, the use of
WhatsApp for messaging and video calls in 2020
3
.
In May 2021, the HSE had data stolen in a cyber
security incident
4
. Criminal ransomware groups look
for organisations with highly sensitive data and
insufficient information security, generating big
disruptions on services relying on computers. As a
result of the cyberattack, there were major disruptions
on access to medical records and services which
lasted over five months.
Following this attack, measures and law
enforcements were undertaken to improve the
infrastructure and prevent future cyberattacks.
However, it is unclear whether an improvement in
awareness and training could help users understand
how their actions could prevent future security and
data breaches.
1
https://www.hpra.ie/homepage/medical-devices/regula
tory-information/new-eu-device-regulations
2
https://www.dataprotection.ie/en/news-media/press-relea
ses/data-protection-commission-announces-decision-
whatsapp-inquiry
Mobile Health (mHealth) is medical and public
health practice supported by mobile devices, such as
mobile phones, patient monitoring devices, personal
digital assistants (PDAs), and other wireless devices
(Kay et al., 2011). While there is a trend of increasing
opportunities for design, development and adoption
of mHealth, solutions for enhancing services and
patient care are not restricted to MD regulated
software. When users choose mainstream apps, or
non-MD apps, to support their practices or treatment,
they should pay attention to how personal and health
data is processed, stored and shared. For convenience,
medical staff and patients may be using mHealth
solutions on their own devices, and raises additional
concern (Wani et al., 2020).
The goal of this position paper is to present an
overview of some of the factors related to the use of
personal devices and mobile applications, in hospital
or healthcare facilities, that raise challenges in data
protection, privacy and safety for patients and staff.
3 METHODS
The PPT model (Schlarman, 2001) has been used in
previous studies to evaluate risks of use of mobile
devices in healthcare facilities (Wani et al., 2020). In
the present paper, we define three categories for
exploring the literature research, inspired by the PPT
model (Table 1), or as follows:
User behaviour: in the PPT model, Schlarman
focuses on people responsible for the security
process. We extend this approach to include
general users, from healthcare staff having access
to patient data, to data controllers and processors,
as well as patients and family members. We do
this for two reasons: (1) because there is a trend in
technologies designed to support healthcare to
consider a holistic approach and facilitate
continuous monitoring and communication
between care provider and patients and (2)
because patients, as well as family members and
caregivers, are also interested in safeguarding
their personal and medical data. We argue that
raising awareness for all groups of users could
help them to adopt responsible behaviour.
Policies and regulations: we refer to the existing
regulations in Europe applied to the
commercialization of medical devices and
3
https://healthservice.hse.ie/staff/coronavirus/working-
from-home/virtual-health/guide-to-whatsapp-for-hse-
staff.html
4
https://www2.hse.ie/services/cyber-attack/how-it-may-
affect-you.html
HEALTHINF 2022 - 15th International Conference on Health Informatics
566
software, as well as general data protection
regulations (GDPR). Following the examples
from the literature, we included relevant examples
of grey literature such as codes of conduct and
hospital policies (Garousi et al., 2016; Wani et al.,
2020).
Mobile devices and applications: while the PPT
model refers to all the products, tools and
materials supporting security, we decided to
highlight the challenges of detecting or
controlling the use of personal devices, such as
smartphones, due to their availability, ease of
access and ubiquity. As mobile devices are not
often designed for medical purposes, we decided
to focus on software running on mobile devices,
also referred as mobile applications.
Our literature search is limited to publications
from 2010 up to 2021. We believe this data limit is
sufficient to include literature considered
representative of the recent advances and dynamic
turnaround on mobile devices, network
infrastructure, software availability and users’
practices.
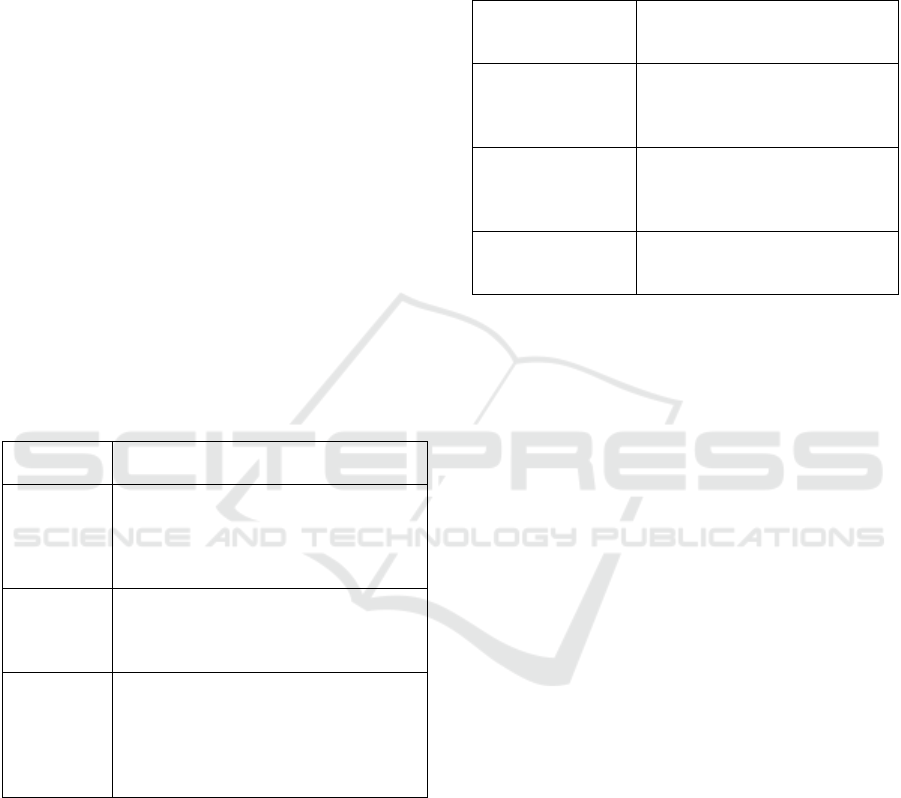
Table 1: Overview of the focus of the present paper after
PPT model.
PPT model Aspect highlighted or extended in the
discussion of the present study
People Users’ behaviour, including all user
groups having access and therefore
responsibilities for the use of personal
data for medical purposes, including
medical staff and patients
Policy EU Medical Device Regulations
(MDR), EU General Data Protection
Regulations (GDPR), Hospital Policies
and Codes of conduct
Technology Mobile devices such as smartphones,
regulated MD software, including
mobile applications, and general public
software and applications, non- MD
apps, having access to personal
information
4 USERS’ BEHAVIOUR
Users are at the centre of our analysis. Data protection
regulations and policies are designed to protect
people’s data: personal information, medical records,
and history of communication between patients and
staff. Users are also the main operators of
technologies, in charge of choices, decisions and
interactions with systems and devices. It is important
to consider factors driving their behaviour, from
acceptance of technology to inappropriate use of
systems. Table 2 presents the main group of users of
mobile devices in healthcare facilities, from our
analysis of existing policies.
Table 2: Groups of users of mobile devices in healthcare
facilities in the context of the present study.
Group of users Actions or behaviours related
to data protection in
healthcare facilities
Patients, visitors,
and informal carers
Usually responsible for their
decisions in following
prescriptions and
recommendations
Medical staff,
interns, students,
and social workers
Usually responsible for making
decisions, monitoring outcomes
and supporting patients during
treatment
Contractors,
administration, and
third-parties
Responsible for providing
general infrastructure and
resources
In the context of this paper, we also consider
factors related to the users’ behaviour actions such as
selecting a device or software, installing, initiating
interaction, learning how to use the app, becoming
familiar with a system, usages related to the system’s
initial intended purpose, appropriation of the system
for different purposes, and the choice to maintain use
or stop.
Human errors are often related to security issues.
Also, we consider whether users’ actions relating to
data protection and safety could be intentional or
unintended.
We would like to highlight the following
behaviours that present risks for data protection,
privacy and safety:
Use of personal devices: There is evidence of
medical staff using their own devices for work
(Wani et al., 2020), for communication (Bautista
& Lin, 2016; Wu et al., 2010), education (Cho &
Lee, 2016) or clinical practice (Koehler, 2013).
Attention is called to issues related to distraction
(Gill et al., 2012), risk of infection (de Jong et al.,
2020) and patient safety (D. McBride et al., 2015).
Inappropriate use of devices or apps: It is
necessary that users keep their devices safe,
password protected, with operating systems and
software up to date. Reasons for inappropriate use
include lack of education, lack of awareness, and
issues related to accessibility and availability of
affordable options.
Use of non-MD apps: The use of general apps
during work could be considered inappropriate
and generate negative attitudes from managers (D.
L. McBride et al., 2015) or patients (Koehler,
mHealth Use in Healthcare Facilities: Raising Awareness in Data Protection, Privacy and Safety
567

2013). Recently, patients and staff acceptance of
mobile applications and devices has improved,
since it has been perceived as useful, facilitating
communication or organisation (Benedictis et al.,
2019; de Jong et al., 2020; Lee Ventola, 2014;
Wyatt et al., 2020). However, general applications
such as calendar, internet browser, and instant
messaging can present risks as there is no
guarantee that users will not disclose or store
personal data (Benedictis et al., 2019; Dexheimer
& Borycki, 2015)
Disclosure of personal or medical data
inadvertently: Users may disclose personal
information when seeking health information
online, or posting reviews for apps and services.
Many are unaware of the fact that their pictures,
full name and contact details may be publicly
displayed. Additionally, some users may not
understand what characterises personal and
medical data, how technologies process and store
them and the risks associated with use of non-MD
apps for health-related purposes.
Lack of awareness and accessibility on policies
and regulations: As with “Terms and
Conditions”, many users only have access to
existing policies and regulations if actively
looking for them. Often the language used can be
a barrier for the users to comply with safety and
security guidelines. Furthermore, users tend not
to read the ‘small print’, and therefore may not be
aware of breaches of specific policies and
regulations which their use of an app may cause.
In summary, many difficulties in misuse of
medical software are not caused by the software itself,
but rather, by the inappropriate use of the software.
Users may not have enough understanding on what
characterises personal and medical data, how
technologies process and store them and the risks
associated with use of non-MD apps for medical
purposes. Additionally, users do not necessarily
understand the importance of MDR, and may not be
on a position to verify compliancy or appropriate use
of them.
5 POLICIES AND REGULATIONS
Given the issues pertaining to Users’ Behaviour as
discussed in the previous section, it is important that
we present and discuss the current regulations and
5
https://ec.europa.eu/health/md_newregulations/overview
_en
6
https://www.hpra.ie/docs/default-source/Safety-Notices/
in201703_mobileappinhealthcare_140917.pdf?sfvrsn=0
policies concerning the use of mobile devices and
apps in healthcare settings.
5.1 GDPR and MDD Devices and
Software
By definition, Medical Devices (MD) include a wide
range of products used in healthcare and are subjected
to strict regulations
5
, which vary depending on the
class of device. In Europe, MDs are reviewed by
notified bodies (e.g. National Standards Authority of
Ireland – NSAI, Health Products Regulatory
Authority HPRA) and, once certified compliant, they
are distinguished by a CE mark (European
Conformity), which indicates, amongst other criteria,
that the product has been adequately tested, that
medical claims are supported by clinical data and that
users are correctly informed about safety of use
6
.
Software, such as mobile applications, once
intended for medical purposes are also considered a
medical device
7
. After a transition period of three
years, the new MDRs are applicable, and include
reinforcement of risk assessment, post-market
surveillance and investigations of clinical evidence.
The collection and processing of personal
information is a sensitive issue, not limited to medical
devices and software. In Europe, the General Data
Protection Regulation (GDPR) was put into effect on
May 25, 2018. Data protection and processing
policies include limitations on data storage, third
party transfers, data anonymisation and
pseudonymisation and disclosure
8
.
We outline some of the issues currently faced by
the users of MDs that seem uncovered by existing
regulations:
Users’ responsibilities It is the responsibility of
the manufacturer, or in the case of software, the
developer, to follow MD regulations.
Compliancy is required before products are placed
in EU markets. However, it is responsibility of the
user to verify that devices and software used for
medical purposes are appropriately CE marked. It
seems to us that users may not be aware of their
responsibilities in ensuring that regulations are
applied, apps are compliant and that their use is
fair and appropriate.
Distinction between MD and non-MD apps: For
mobile apps and software, it is difficult for the
users to distinguish regulated and non-regulated
options in applications stores. Many MD apps
might remain unknown or inaccessible.
7
http://www.hpra.ie/homepage/medical-devices/special-
topics/standalone-software-and-applications
8
https://www.hse.ie/eng/gdpr/hse-data-protection-policy/
hse-data-protection-policy.pdf
HEALTHINF 2022 - 15th International Conference on Health Informatics
568

Additionally, non-MD apps may seem an easy
option to users who are often unaware of the
implications of use on data protection and privacy.
Assessment of compliancy: mHealth apps for use
in the Irish public healthcare system must
complete a Privacy Impact Assessment (PIA) to
ensure compliance with privacy and GDPR. This
process is detailed and can be lengthy (MacEntee,
2021).
PIA not suitable for small systems: An
examination of research has indicated that PIAs
are designed for large scale systems but do not
identify privacy issues when applied to a variety
of smaller scale mHealth apps. This research also
suggests that one size does not fit all with respect
to PIAs and mHealth apps
Transparency of selection and compliancy over
time: As with many apps, mHealth solutions
should be constantly updated and improved, as
they might be dependent on the configurations of
operative systems and devices. Major updates
should be complying with the regulations.
However, the current offer can quickly become
obsolete and present risks of safety and security
for the users.
The gap between designers, practitioners, and
regulatory authorities: From conception to launch
in the market, users’ needs must be identified and
met. The challenges in accessing patients for user
tests and clinical trials are barriers to improving
design, reliability and possibly effectiveness of
digital solutions.
5.2 Policies and Codes of Conduct
Other regulations exist and try to cover aspects
related to the use and application of MDR and GDPR.
With regards to healthcare, hospital and institutions
usually define and circulate their policies, and
members of staff should respect existing codes of
conduct. Examples are the HSE Data Protection
Policy
9
targeting people who may have access to
patients and their data including ‘staff, students,
interns and work experience candidates, contractors,
sub-contractors, agency staff, medical colleges and
authorised third party commercial service providers’.
According to the literature (Bautista & Lin, 2016; de
Jong et al., 2020; Johnston et al., 2015), some issues
around policies and codes of conduct can relate to
Target groups not inclusive: Policies and codes of
conduct are provided to staff, students, interns,
9
https://www.hse.ie/eng/gdpr/hse-data-protection-policy/
hse-data-protection-policy.pdf (June 2019)
and contractors, who are informed and obligated
to comply. Patients are usually not included as
target groups.
Non-generalisable: Policies and procedures are
local, and can apply from institutions, groups, and
localities to national bodies. It is difficult for users
to be aware of these variations, as well as for
developers to adapt the systems to current
guidelines and updates.
Lack of implementation plans and training:
Policies usually do not define how training is
going to be provided.
Finally, there is often a lack of communication,
accessibility and availability. Some users might be
unaware of their responsibilities and general codes of
conduct.
6 MOBILE DEVICES AND
APPLICATIONS
As presented in the introduction, the focus of this
paper is to discuss the use of personal mobile devices
in healthcare settings, and to discuss issues related to
the use of mobile applications having access to
personal data in these settings, potentially with
medical or health information.
Because of the regulations in place, it is important
to distinguish the risks associated with the use of
regulated MD software, which includes MD mobile
apps, to risks of using mainstream non-regulated
mobile apps that can also be used for medical or
health-related purposes.
6.1 MD Mobile Apps
MD software designed for monitoring patients,
facilitating diagnosis or self-management of care is
subject to regulations according to the clinical
category and risk assessment defined by MDR. Some
issues include:
Customisation of MD apps: Some institutions
would support the development of private
solutions, e.g. specially designed communication
platform for staff-staff or staff-patients. This can
be costly, and users might prefer interfaces that
are familiar.
Some mHealth solutions are excluded from
MDR: mHealth apps are currently being
developed to support many medical fields,
including dermatology, paediatrics, cardiology,
mHealth Use in Healthcare Facilities: Raising Awareness in Data Protection, Privacy and Safety
569

oncology, and a variety of chronic conditions
such as diabetes. They may also support
assistive devices. Other areas such as falls, frailty
and clinical trials also are covered under MD
regulations However, some health conditions
such as mental well-being, pregnancy or
menstruation can benefit from mHealth
solutions. If not covered by MDR, users are at
risk of losing control of very sensitive data.
Intentional use of non-MD apps: Users may
intentionally choose apps with which they are
familiar, prioritising usability over safety.
Intentional use may include instant messaging
between staff, teleconsultations, search of
prescriptions or medication details, and booking
appointments.
Networks and communications: As conventional
server−client applications in PCs, mobile
applications communicate with many cloud
services and share information connecting to
many networks and platforms. The use of private
or public networks present risks to secure
transfer of data and many users do not know how
to keep their connections safe.
6.2 mHealth non-MD
Some apps, such as those developed to support
wellness, fitness, period trackers, pregnancy, smoke
cessation, diet, and nutrition, can collect a large
amount of sensitive data from users, and most are not
transparent on how this data is managed. We
highlight the following issues:
Lack of transparency: as for MD apps, it is the
responsibility of designers and developers to
provide users with information on the
transparency of data collection, control of data
capture, storage, and processing such as
anonymisation, pseudonymisation, removal,
time limits, and sharing. Lack of regulation
makes non-MD apps less clear about how data is
managed.
Third parties and shared data: As apps are
selected for available countries, the use of data
by third-party in different countries is not always
clearly indicated. From target advertisements to
data breaches, and risks of safety and privacy to
the users, existing regulations provide a
reassuring framework. However, users do not
always verify if apps developers are ethically
responsible for the management and control of
their sensitive data.
App permission: Upon installation, use or
updates, the users can grant permission, often
unknowingly, as they do not check or read
detailed terms and conditions, to mobile
applications to capture, store, process or even
share the users’ location, camera or audio
recordings, media content or textual information.
Recent mobile devices are equipped with many
sensors enabling data collection: connectivity
such as Wi-Fi and Bluetooth, contactless NFC,
motion sensors as gyroscope, accelerometers are
user to recognise levels of activity, localisation
from GPS as well as Wi-Fi, biometric sensors,
microphones, and others. If GDPR preconises
fair use of data, users are not always attentive
when enabling permissions to the apps installed
in their devices. Apps do not generally give
options to the users to select when and how to
turn data access in or off.
7 CONCLUSION
We reviewed challenges related to the use of mobile
devices and apps for health and medical purposes. As
discussed in the literature, users might prefer to use
their personal devices and familiar interfaces to
improve the effectiveness of their practices,
sometimes without understanding the implications or
risks for data protection, privacy and safety. We argue
that policies and regulations, as well as designers and
developers, should be more considerate of users’
behaviour.
When the purpose of the system and its use of data
is not transparent, there is a risk for data protection
and privacy with unintended data breaches or security
faults. Therefore, there is an onus on the software
engineer or developer to ensure that the MD software
such as MD apps, as well as general apps supporting
mHealth are fit-for-purpose. It is responsibility of
designers and developers to provide users with
information transparency on data collection, control
for data capture, storage, and processing such as
anonymisation, pseudonymisation, removal, time
limits, sharing.
References for this position paper were selected
to enable an initial discussion on the issues raised by
the use of mobile applications and devices in
healthcare facilities, such as hospital and clinics. By
referring to the PPT model, we have provided a
holistic view of the issues related to use of
smartphones in hospital observed in the past 10 years.
Future work should further study the aspects
highlighted in the present paper in the scope of
security and privacy threats for conventional
information systems. It would be important to take
this step, as mobile applications create an additional
HEALTHINF 2022 - 15th International Conference on Health Informatics
570

layer to collect, store and share health and medical
data that are not restricted to MD software. Further
studies could benefit from a historical perspective or
to analyse the trends based on recent advances related
to the adoption of mHealth solutions after the
COVID-19 pandemic (Storni et al., 2021; Webb et al.,
2020).
7.1 Future Work
We argue that the challenges outlined in this paper
could be addressed by policies and regulations
reinforcing the need for public training, education and
awareness, and these could be two-fold:
- for users, about their choices and risks,
- for designers and developers, about
transparency, ethics and their responsibilities.
It is necessary to provide users with information
regarding their responsibilities in ensuring that
regulations are applied, apps are compliant and that
their use is fair and appropriate. Some suggestions
are:
Supporting users to choose safe mHealth: A
possible solution would be a filter in digital stores,
such as Google Play Store or Apple Store, to
differentiate MD from general apps. Apps
available in these stores must already comply with
software development guidelines.
Involving users in the design process: In Ireland,
the digital transformation includes initiatives such
as training and support to healthcare staff in
identifying opportunities to design and develop
technological solutions
10
, offering unique insight
to solving real-world problems.
A database of regulated mHealth: This could help
users, both patients and healthcare staff, to find
the support they need from MD. This solution has
been suggested in the literature as a mean to
address general public and patients’ needs
(Olivero et al., 2019). Recently, initiatives such as
the NHSX
11
in UK have been created to support
the transition to the digital healthcare. As part of
their services, manufacturers can apply to be listed
in a selection of existing MD apps, and centralize
their review and offer to healthcare staff.
Awareness and means to action: Users also might
be informed and provided with easier means to
contact regulatory bodies for verification,
information or indicate possible issues.
Improving user experience: It is important that the
design of mHealth solutions support users in
10
https://www.hse.ie/eng/about/who/communications/
digital/digital-transformation/
making good choices, being aware of policies and
processes, reflecting on transparency, reporting
inappropriate use or system.
Regarding the responsibility of designers and
developers, we highlight the following suggestions:
Education: Guidelines should be presented early
and regularly in educational settings to systems
developers. These would cover best practices so
that they would understand the factors involved in
launching in the MD market and the issues with
the use of non-regulated apps in healthcare. They
should also be familiar with sanctions for security
breaches or threats.
Improved design: MD software should
automatically prompt users with options for
encryption or better management of security
options. For example, public websites or search
engines could detect use of personal data, such as
fields labelled “first name”, “last name”, “date of
birth” when classifying information and alert
users.
ACKNOWLEDGEMENTS
This research has been partially supported by the
Science Foundation Ireland Grant number
13/RC/2094_P2 to Lero - the Science Foundation
Ireland Research Centre for Software (www.lero.ie).
REFERENCES
Bautista, J. R., & Lin, T. T. C. (2016). Sociotechnical
analysis of nurses’ use of personal mobile phones at
work. International Journal of Medical Informatics, 95,
71–80. https://doi.org/10.1016/j.ijmedinf.2016.09.002
Benedictis, A. De, Lettieri, E., Masella, C., Gastaldi, L.,
Macchini, G., Santu, C., & Tartaglini, D. (2019).
WhatsApp in hospital? An empirical investigation of
individual and organizational determinants to use. PLoS
ONE, 14(1), 1–12. https://doi.org/10.1371/journal.po
ne.0209873
Cho, S., & Lee, E. (2016). Distraction by smartphone use
during clinical practice and opinions about smartphone
restriction policies: A cross-sectional descriptive study
of nursing students. Nurse Education Today, 40, 128–
133. https://doi.org/10.1016/j.nedt.2016.02.021
de Jong, A., Donelle, L., & Kerr, M. (2020). Nurses’ use of
personal smartphone technology in the workplace:
Scoping review. JMIR MHealth and UHealth, 8(11), 1–
15. https://doi.org/10.2196/18774
11
https://www.nhsx.nhs.uk/
mHealth Use in Healthcare Facilities: Raising Awareness in Data Protection, Privacy and Safety
571

Dexheimer, J. W., & Borycki, E. M. (2015). Use of mobile
devices in the emergency department: A scoping
review. Health Informatics Journal, 21(4), 306–315.
https://doi.org/10.1177/1460458214530137
Garousi, V., Felderer, M., & Mäntylä, M. V. (2016). The
need for multivocal literature reviews in software
engineering. 1–6. https://doi.org/10.1145/2915970.291
6008
Gill, P. S., Kamath, A., & Gill, T. S. (2012). Distraction:
An assessment of smartphone usage in health care work
settings. Risk Management and Healthcare Policy, 5,
105–114. https://doi.org/10.2147/RMHP.S34813
Johnston, M. J., King, D., Arora, S., Behar, N., Athanasiou,
T., Sevdalis, N., & Darzi, A. (2015). Smartphones let
surgeons know WhatsApp: An analysis of
communication in emergency surgical teams. American
Journal of Surgery, 209(1), 45–51. https://doi.org/
10.1016/j.amjsurg.2014.08.030
Kay, M., Santos, J., & Takane, M. (2011). mHealth - New
horizons for health through mobile technologies (Vol.
3). World Health Organization. https://doi.org/
10.1109/CBMI.2010.5529886
Koehler, N. (2013). Healthcare professionals’ use of mobile
phones and the internet in clinical practice. Journal of
Mobile Technology in Medicine, 2(1), 3–13.
https://doi.org/10.7309/jmtm.76
Lee Ventola, C. (2014). Mobile devices and apps for health
care professionals: Uses and benefits. P and T, 39(5),
356–364.
MacEntee, B. (2021). Are Privacy Impact Assessments for
Mobile Health Applications Fit for Purpose? (Issue
August). University of Limerick.
McBride, D. L., LeVasseur, S. A., & Li, D. (2015). Non-
work-related use of personal mobile phones by hospital
registered nurses. JMIR MHealth and UHealth, 3(1), 1–
5. https://doi.org/10.2196/mhealth.4001
McBride, D., LeVasseur, S. A., & Li, D. (2015). Nursing
performance and mobile phone use: Are nurses aware
of their performance decrements? JMIR Human
Factors, 2(1), 1–6. https://doi.org/10.2196/human
factors.4070
Olivero, E., Bert, F., Thomas, R., Scarmozzino, A., Raciti,
I. M., Gualano, M. R., & Siliquini, R. (2019). E-tools
for hospital management: An overview of smartphone
applications for health professionals. International
Journal of Medical Informatics, 124(January), 58–67.
https://doi.org/10.1016/j.ijmedinf.2019.01.010
Schlarman, S. (2001). The people, policy, technology (PPT)
model: Core elements of the security process.
Information Systems Security
, 10(5), 1–6. https://
doi.org/10.1201/1086/43315.10.5.20011101/31719.6
Storni, C., Tsvyatkova, D., Richardson, I., Buckley, J.,
Abbas, M., Beecham, S., Chochlov, M., Fitzgerald, B.,
Glynn, L., Johnson, K., Laffey, J., Mcnicholas, B.,
Nuseibeh, B., Connell, J. O., Keeffe, D. O., Keeffe, I.
R. O., Callaghan, M. O., Razzaq, A., Rekanar, K.,
Welsh, T. (2021). Toward a Compare and Contrast
Framework for COVID-19 Contact Tracing Mobile
Applications : A Look at Usability. 5(Biostec), 557–
565. https://doi.org/10.5220/0010307005570565
Wani, T. A., Mendoza, A., & Gray, K. (2020). Hospital
Bring-your-own-device security challenges and
solutions: Systematic review of gray literature. JMIR
MHealth and UHealth, 8(6), 1–13. https://doi.org/
10.2196/18175
Webb, H., Parson, M., Hodgson, L. E., & Daswani, K.
(2020). Virtual visiting and other technological
adaptations for critical care. Future Healthcare
Journal, 7(3), e93–e95. https://doi.org/10.7861/
fhj.2020-0088
Wu, R. C., Morra, D., Quan, S., Lai, S., Zanjani, S.,
Abrams, H., & Rossos, P. G. (2010). The use of
smartphones for clinical communication on internal
medicine wards. Journal of Hospital Medicine, 5(9),
553–559. https://doi.org/10.1002/jhm.775
Wyatt, K. D., Finley, A., Uribe, R., Pallagi, P., Willaert, B.,
Ommen, S., Yiannias, J., & Hellmich, T. (2020).
Patients’ experiences and attitudes of using a secure
mobile phone app for medical photography: Qualitative
survey study. Journal of Medical Internet Research,
22(5), 1–10. https://doi.org/10.2196/14412
HEALTHINF 2022 - 15th International Conference on Health Informatics
572
