
PAIN AND EFFICIENCY IN NEONATAL BLOOD SAMPLE
SCREENINGS
New Devices for Reducing Pain and Improving Blood Sample Quality
Bruno Wacogne, Christian Pieralli
Institut FEMTO-ST, Département d'Optique P.M. Duffieux, UFR Sciences et Techniques
Route de Gray, 25030 Besançon cedex, France
Gonzalo Cabodevila, Nolwenn Baron
Institut FEMTO-ST, Département MN2S, Avenue de l'Observatoire, 25030 Besançon cedex, France
Sandrine Marioli
Service de Pédiatrie 1, CHU Saint Jacques, 2 place Saint Jacques, 25030 Besançon cedex, France
Lionel Pazart
Centre d'Investigation Clinique en Innovation Technologique,CHU Saint Jacques
2 place Saint Jacques, 25030 Besançon cedex, France
Keywords: Neonatal screening, pain evaluation, micro-needles arrays, image processing.
Abstract: Neonatal blood sample screening is recognised as a difficult gesture and painful to the newborns. The
number of detected diseases is still relatively low and depends on the country where it is performed. There
is a real need for new techniques that reduce pain, facilitate the blood sampling, increase the quantity of
sampled blood and improve the collection of blood of the cardboard blotter actually used. In this paper, we
present systems that are currently developed in Besançon (France) in collaboration between the FEMTO-ST
Institute and the University Hospital. They mainly concern micro-needles arrays and pressure free blood
sampling devices. The choice of these systems has been dictated by a study of the pain that newborns feel
during the gesture. The ulterior motive of this work is to improve neonatal blood sample screenings and
therefore, to increase the number of screened diseases and try to generalise this technique to places where it
is not yet done.
1 INTRODUCTION
The blood sample screening of several congenital
diseases (phenylketonuria, hypothyroidism, adrenal
hyperplasia, ...) is performed routinely at birth in
many countries. The number of detected diseases
depends on the screening policy of the country and
the technical limitations of the methods. For
neonates, techniques are mainly limited by the
difficulties of realisation of the sampling gesture, the
small volume of punctionable blood and the pain
caused by the gesture. Currently, the French
Association for the Detection and Prevention of
Handicaps for Children (AFDPHE), responsible for
organising the screening in France, favors (at the
third day of life) a collection of capillary blood after
a bite by a retractable lancet at the heel of the
newborn. This gesture is followed by successive
pressures on the heel in order to collect blood
droplets on a cardboard blotter. This gesture is
recognised as painful (Facchini, 2005), (Owens,
1984). It should be noted that in some places,
screenings are performed with venous blood instead
of capillary blood. In this case, the gesture is more
technical and it presents risks for both the nurse and
the newborn. In this paper, we restrict the discussion
to the case of capillary blood.
290
Wacogne B., Pieralli C., Cabodevila G., Baron N., Marioli S. and Pazart L. (2009).
PAIN AND EFFICIENCY IN NEONATAL BLOOD SAMPLE SCREENINGS - New Devices for Reducing Pain and Improving Blood Sample Quality.
In Proceedings of the International Conference on Biomedical Electronics and Devices, pages 290-295
DOI: 10.5220/0001776902900295
Copyright
c
SciTePress

The orientation of the present works is intended
to develop medical devices less painful and that
offer a higher potential screening of a larger number
of diseases in newborns, replacing technology
currently used.
The context of the study, together with the
evaluation of pain during the blood collection
gesture is presented in section 2. Section 3 deals
with the study of a painless micro-needles array
meant to replace the classical lancet. In section 3, we
present a device developed to obtain blood without
successive pressures on the newborn's heel.
Furthermore, this system can be used to efficiently
deposit the collected blood on the cardboard blotter.
Then, a conclusion and some perspectives will be
proposed at the end of this manuscript.
2 CONTEXT AND PAIN
EVALUATION
Screenings are achieved by nurses on the third day
of life. They require the use of an automatic, sterile
and disposable lancet that pierces the skin of the
heel. The heel is then pressed to obtain droplets of
blood that are collected on circles drawn on a
cardboard blotter (as seen on figure 1). Since the
blood flow is not sufficient, successive manual
pressures to the heel are required. Circles on the card
must be completely filled and the blood must cross
the card (this is not the case for all the circles in
figure 1). If the first attempt does not give enough
blood, a second is made on the same heel or on the
other one. In the same way, if the droplets do not
cross the screening card it is necessary to add blood.
This tracking is recognised as painful.
Newborns feel more or less pain during the
blood sampling gesture. Pain can be estimated using
various behavioural scales (Destuynder, 1991),
(Uyan, 2008). Both D.A.N. (Douleur Aiguë du
Nouveau-Né, Newborn Intense Pain ) and E.D.I.N.
(Echelle de Douleur et d’Inconfort du Nouveau-Né,
Newborn Pain and Discomfort Scale) scales are
adapted to the evaluation of newborn's pain. We
used the D.A.N. scale because it is more specific to
intense pain. It allows quoting three criteria: the
facial answer, the movements of the members and
the vocal answer. Each criterion is quoted either
between 0 and 3 or between 0 and 4. We evaluated
the pain at five different moments: before the
gesture, when the nurse takes the baby's heel, during
the lancet sting, when the nurse presses the heel and
after the gesture.
For a first analysis, we observed 55 children for
which 58 stings were needed. Indeed, no blood was
obtained in 3 cases at the first attempt.
Figure 1: Actual blood sampling technique (top) and
cardboard blotter used to collect blood.
The gesture proving to be painful for a majority
of children, we refined our study at three moments:
the taking of the heel, the sting, the pressing of the
heel. The results obtained showed that the taking of
the heel is not painful (only 1 case). The sting is
painful in 10 cases, what nearly represents 25 % of
the cases. The pressing of the heel is the most
painful moment (26 cases representing 68%).
Therefore, we oriented our works toward two
new systems of blood sampling. The first one is a
micro-needles array meant to reduce the pain during
the sting (in replacement of the lancet). The second
one is used after the heel has been stung with the
lancet. It is designed to collect enough blood without
any pressure on the heel. Moreover, this system
leads to a perfect impregnation of the cardboard
blotter.
3 MICRO-NEEDLES ARRAY
The goal of this work is to estimate the geometry a
micro-needles array used to collect blood at the
newborn's heel. This array should replace the lancet.
The goal is to penetrate the newborn's heel and to let
blood flow in the holes of the micro-needles by
capillarity. In a final version, a tank should be
designed to store the sampled blood. Various studies
concerning micro-needles have been published, but
PAIN AND EFFICIENCY IN NEONATAL BLOOD SAMPLE SCREENINGS - New Devices for Reducing Pain and
Improving Blood Sample Quality
291
very little on blood collection. For example, we can
mention the work presented by (Mukerjee, 2004). In
this case, a liquid is obtained after a relatively long
time but this liquid does not contain only blood.
Other systems, inspired by mosquitos morphology,
are presented but some complementary studies are
still required (Oka, 2001), (Sharaf, 2003). Figure 2
shows an example of a micro-needles array
fabricated in our laboratory. This particular
architecture was designed for drug delivery and not
for blood sampling.
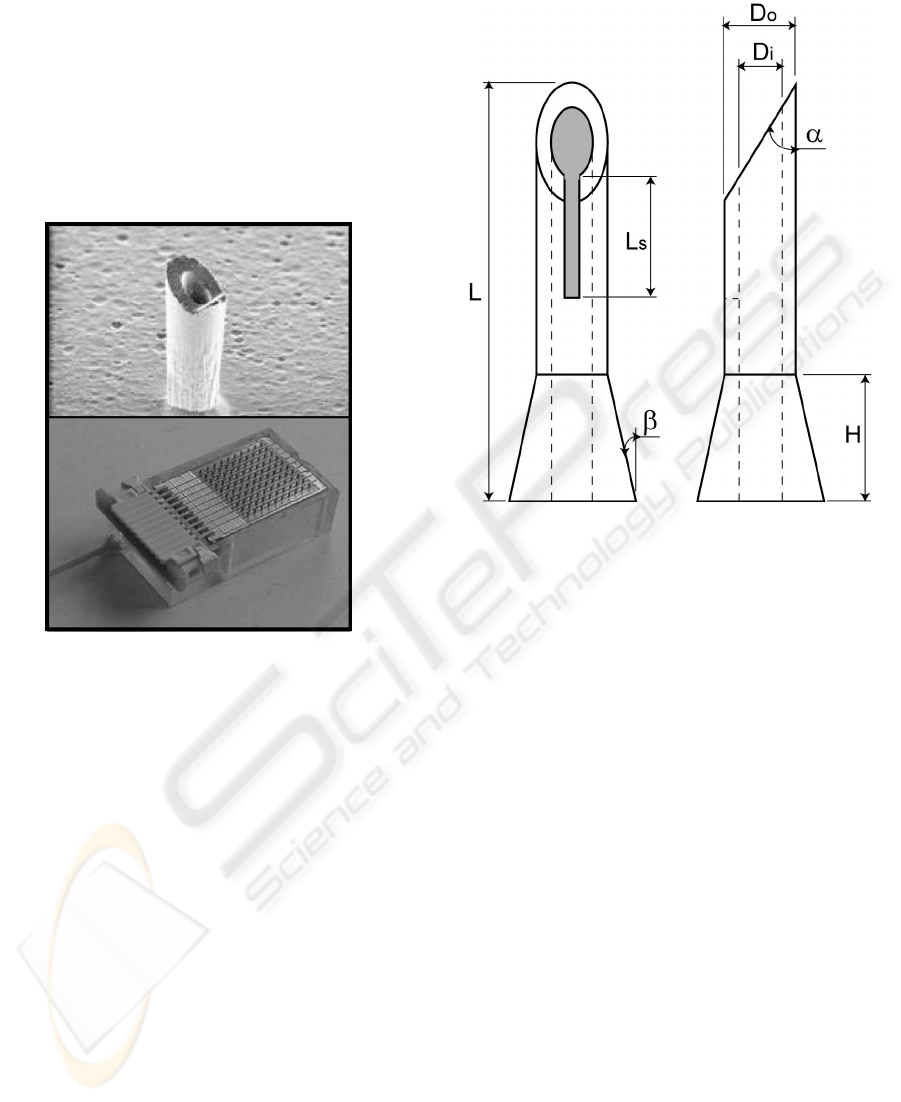
Figure 2: Example of micro-needles array fabricated in our
laboratory.
For our specific application, the characteristics
of the micro-needles have to be rethought according
to the newborn's heel anatomy. The characteristics
we studied are the shape, the dimensions and the
number of micro-needles in a array. This work is
based on a capillaroscopy study of the newborn's
heels that helped to define the depth of the children's
capillary network (350 µm) and its density (70
capillaries by mm
2
). In what follows, we present the
conclusion drawn from our studies. The details of
numerical simulations can be obtained on demand.
Some parameters used in this study come from
the work conducted for several years in our
laboratory. Micro-needles are fabricated with
silicon. The silicon surface is oxidized in order to
make the device bio-compatible. The needles are
cylindrical. In order to improve their strength the
basis of the needles is conical. The tip of the needles
is bevelled in order to facilitate the penetration of the
stratum corneum. Finally, the needles exhibit a
longitudinal slit. It is used to maximize the contact
surface between the capillaries and the hole of the
needle. Figure 3 shows a schematic diagram of one
micro-needle.
Figure 3: Schematic representation of a micro-needle
dedicated for newborns blood sampling.
3.1 Dimensions of the Needles
The height L of the needles is fixed to 1200 µm to
ensure a penetration until the dermis of skin. The
inner and outer diameters (D
i
and D
o
) are 60 and 150
µm respectively. These diameters increase the
probability to meet a capillary.
The bevel exhibits an angle α = 41°. This value
is calculated considering that a third of the needle
penetrates the skin (400 µm) and that the hole of the
needle must be at the level of the capillaries (350
µm).
The slit must not extend outside the area where
the capillaries are located. In this way, we reduce the
risk to collect something else than blood. Its length
is therefore limited to L
s
= 200 µm. The width of the
slit doesn't have an influence on the needle strength.
Numerical simulations showed that the Von Mises
constraints are almost not influenced by this
parameter. For the moment, we consider a slit width
of 60 µm.
The basis of the needle is conical. This cone is
defined by its height H and its angular aperture β.
Numerical simulations showed that the Von Mises
constraints are minimized for a height H = 600 µm
and an angular aperture β < 30°.
BIODEVICES 2009 - International Conference on Biomedical Electronics and Devices
292
3.2 Probability to Collect Blood with
the Needles
To summarize, the micro-needles are cylindrical
with a conical basis. They exhibit an opening used to
collect blood. This opening consists of the hole of
the needle and the longitudinal slit. When the micro-
needle has penetrated the skin, two surfaces can be
defined: the total surface of the needle in skin and
the surface of the opening that is susceptible to be in
contact with the capillaries. The ratio between these
two surfaces allows estimating the probability to
collect blood.
Studies showed that an array of 8 needles gives a
probability to collect blood equal to 67%. It is
necessary to increase the number of needles up to 24
to reach a probability of 96%. We consider that the
distance between two needles must be at least equal
to 1 mm. Below this size, the needles may not
penetrate the skin (fakir effect). Therefore, a 24
needles array is about 4*6 = 24 millimetre square,
which is quite large. Another possibility is to use a 8
needles array (8 millimetre square) three times on
three different parts of the heel.
3.3 Perspectives
To conclude this section, we have to mention that
we are actually working on capillarity studies that
should answer the question: how long will it take to
collect enough blood ? It is indeed possible that a
pumping system will be required in order to reduce
the gesture duration.
Another aspect that must be addressed concerns
the penetration of the needles into the skin.
Techniques developed in the frame of other projects
should be transposable to the specific anatomy of
newborn's heel.
4 PRESSURE FREE
COLLECTION SYSTEM
We recall that most of the children feel pain when
their heel is pressed. The pressure on the heel is
necessary to get the required quantity of blood in
order to correctly fill the circles of the cardboard
blotter. There are two main difficulties:
1. the quantity of the sampled blood with respect
of the felt pain
2. the impregnation of the card circles; blood must
cover the whole surface of the circles and the
rear face of the card must be correctly
impregnated.
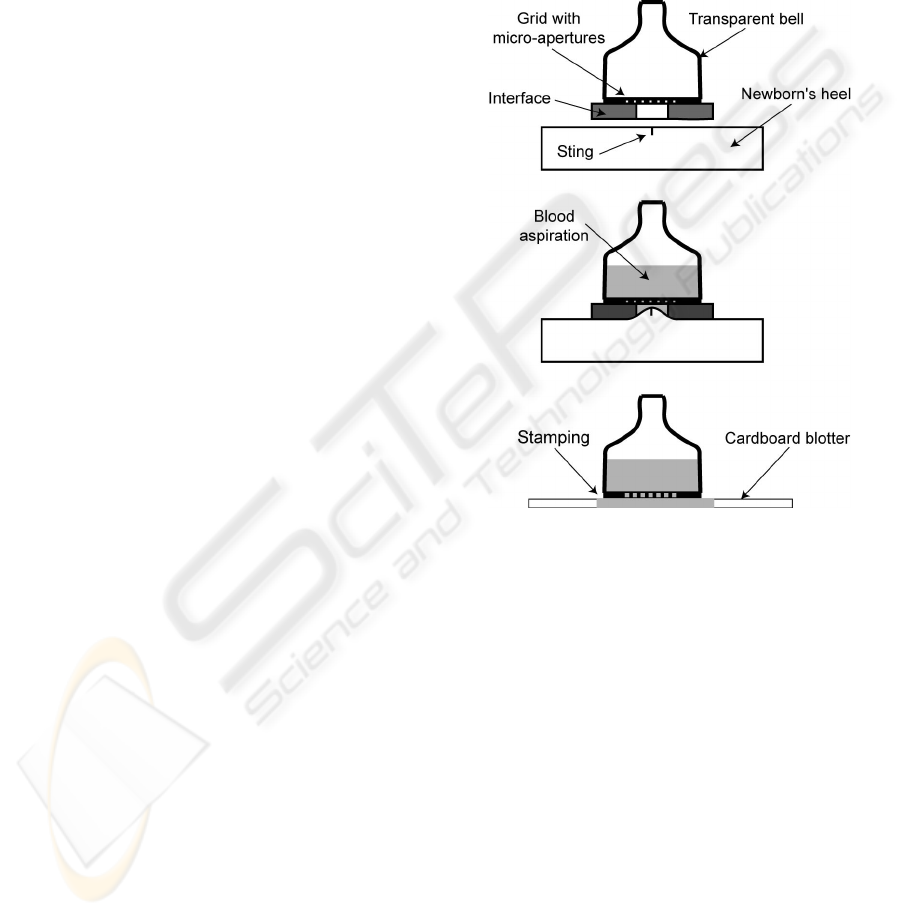
To bring some solutions to these two constraints,
a particular tip is studied (figure 4). This tip is fixed
to the extremity of a reversed syringe that is not
represented on the figure. The tip consists of a
transparent bell onto which a micro-grid is stuck. A
flexible interface is fixed on the micro-grid. The
latter must be removable. It consists of thick disc
with an aperture in its centre. We proceed as
follows.
Figure 4: Principle of the pressure free system.
1. We pierce the newborn's heel with the
conventional lancet.
2. We apply the device onto the cut. The flexible
interface ensures the bloodproofness of the
whole system and prevents the skin to be in
contact with the micro-grid. Without this
interface, the probability that the cut coincides
with a few micro-apertures of the grid would be
very weak and the quantity of sampled blood
likely insufficient.
3. We start the aspiration by means of the reversed
syringe. The pressure on the heel is no longer
required.
4. Blood fills the transparent bell. The volume of
the bell is fixed to 1 ml. We stop the aspiration
when the bell is filled.
5. We withdraw the device from the heel.
6. We remove the interface. The dimension of the
grid apertures is calculated so that blood
remains in the bell during these operations.
PAIN AND EFFICIENCY IN NEONATAL BLOOD SAMPLE SCREENINGS - New Devices for Reducing Pain and
Improving Blood Sample Quality
293

7. We apply the tip on the card. The apertures of
the grid fill the whole surface of the circles
printed on the card. By stamping, the circles are
correctly filled. The capillarity forces are
sufficient for blood to distribute efficiently.
Grids are micro-machined by Deep Reactive Ion
Etching. Three characteristic dimensions of the
apertures have been considered: 300 µm, 200 µm
and 75 µm. Two grid thicknesses have been tested:
300 µm and 525 µm.
The results presented here only concern the tests
of homogeneity of the blood deposited on the cards.
The blood aspiration through the grid will be tested
subsequently. An example of grid attached to the
transparent bell is shown in figure 5.
Figure 5: View of a micro-machined grid fixed on a
transparent bell.
We analyzed the uniformity of the blood
collection with the help of a light source equipped
with a diffuser that permits to illuminate the
cardboard blotter in a homogeneous manner. A CCD
camera is used to acquire pictures of the different
blood impregnated circles. Finally, a suitable image
processing is used to measure the uniformity of
blood. An example of impregnated card is presented
on figure 6 (top). In this case, only 800 µl was
necessary to completely fill the circles. Besides, no
pressure on the syringe was required, the capillarity
forces being sufficient. The homogeneity of the
blood is clearly observed. Also, we can note that
blood completely crosses the card as it can be seen
in figure 6 (bottom). In all, six cards have been
impregnated with grids of different thicknesses and
apertures of various dimensions.
Two steps are required to evaluate the
homogeneity by image processing. The first one
consists in semi-automatically detecting the circle
that contains blood. The second one consists in
evaluating the homogeneity of the blood. To do this,
the pictures are convolved with a test window of 5x5
then of 25x25 pixels. We calculate the standard
deviation on the considered window; the lower the
standard deviation, the better the homogeneity. A
typical result is given in figure 7 where the
homogeneities obtained with apertures of 300 and 75
µm are shown. In abscissa we have the number of
the card and in ordinates the value of the
homogeneity indicator.
Figure 6: Picture of both front and rear side of a cardboard
blotter impregnating with the pressure free device.
Figure 7: Experimental estimation of the homogeneity of
the cardboard blotter impregnation for two aperture sizes.
The conclusion seems obvious. For equal micro-
grid thicknesses, homogeneity is better for large
apertures than for small ones. The grid thickness has
no influence. 300 µm apertures seem interesting for
two reasons. First, the apertures are small enough so
that the blood doesn't escape from the grid before the
latter is in contact with the cardboard blotter.
Secondly, such a dimension is compatible with the
industrial machining. The manufacture of such tips
will therefore be cost-effective.
5 CONCLUSIONS
In this paper, we have presented new medical
devices that reduce pain and should allow screening
a larger number of diseases in newborns. They are
meant to replace the technology currently used in
BIODEVICES 2009 - International Conference on Biomedical Electronics and Devices
294

neonatal blood sample screening. The origin of this
work is a study of the pain felt by the newborns.
This has highlighted that both, the sting and the
pressure required for the blood collection where
painful. We therefore studied a micro-needles array
as well as a system that suppresses pressures on the
children's heel. The ulterior motive of this work is to
improve neonatal blood sample screenings and
therefore to increase the number of screened
diseases and try to generalise this technique to a
places where it is not yet done.
ACKNOWLEDGEMENTS
This work is supported by the French Health
Ministry through a Hospital Protocol for Clinical
Research.
REFERENCES
Destruynder, R., Lassauge, F., Menget, et al., 1991, Pain
in the newborn in an intensive care unit, Pediatrie,
Vol. 46, pp. 535-539.
Facchini, A., Bellieni, CV., 2005, Relating pain intensity
of newborns to onset of nonlinear phenomena in cry
recordings, Physics Letters A, Vol. 338, pp. 332-337.
Mukerjee, E., Collins, S., Isserof, et al., 2004,
Microneedles array for transdermal biological fluid
extraction and in situ analysis, Sensors and Actuators
A, Vol. 114, pp. 267-275.
Oka, K., Aoyagi, S., Isono, et al., 2001, Fabrication of a
micro-needle for a trace blood test, Transducer'01
Digest of Technical Papers, pp. 412-415.
Owens, ME., Todt, EH., Pain in infancy : neonatal
reaction to a heel lance, 1984, Pain, Vol. 20, pp. 77-
86.
Sharaf, R., Aggarwal, P., Kaler, K., et al., 2003, On the
design of an electronic mosquito : design and analysis
of the micro-needle, Proceedings of the International
Conference on MEMS, NANO and Smart Systems,
pp. 32-35.
Uyan, ZS., Bilgen, H., Topuzoglu, et al., Comparison of
three neonatal pain scales during minor painful
procedures, 2008, Journal of Maternal-Fetal and
Neonatal Medicine, Vol. 21, pp. 305-308.
PAIN AND EFFICIENCY IN NEONATAL BLOOD SAMPLE SCREENINGS - New Devices for Reducing Pain and
Improving Blood Sample Quality
295
