
INVESTIGATION OF OPERATING PARAMETERS FOR A
SEMEN QUALITY ANALYSIS SYSTEM
S. Atherton, C. R. Evans, P. Roach, D. C. Hughes, G. McHale and M. I. Newton
School of Science and Technology
Nottingham Trent University, Clifton Lane, Nottingham
NG11 8NS, U.K.
Keywords: Sperm, Semen, Motility, Acoustic wave, QCM, Time of flight.
Abstract: To increase the success rate of Artificial Insemination (AI) in animals, it is important that the semen sample
is of a high quality. The quality is related to both the number and motility of sperm present. Numerous
methods of analysing semen samples exist, but these are generally expensive and/or laboratory based. A
useful alternative would be an inexpensive simple system that could be used in the field immediately prior
to insemination. We present a time of flight (ToF) technique using a quartz crystal microbalance (QCM). In
this system the sperm are introduced at one end of a liquid filled swim channel and self propel to a QCM
sensor at the other end. A chemical coating is applied to the QCM to bind the sperm and from the frequency
change the number of attached sperm and their ToF can be measured. We report the effect of temperature
and the introduction of small quantities of progesterone into the swim channel on the sperm ToF. Results
show the QCM can be used to detect the arrival of the sperm and that increasing temperature and the
presence of progesterone are both shown to decrease the ToF.
1 INTRODUCTION
Within the Artificial Insemination (AI) industry it is
important to be able to measure the concentration of
viable sperm in a sample. AI is a common procedure
in farm animals, more than 100 million inseminations
are performed globally every year. It is not only the
number of sperm in a semen sample that is crucial to
the success of the insemination process, but also the
motility of the sperm. Whilst having a large number
of motile sperm in a sample is not a guarantee of
fertility, it is an excellent indicator of semen quality.
Two optical methods of performing sperm
counts are the haemocytometer and counting
chambers. The drawback of these methods is that
multiple measurements are needed to achieve an
acceptable level of precision, resulting in a more time
consuming procedure. The reason for the relative
inaccuracy of these methods is due to the rapid
movement of the sperm under high magnification
and the tedious nature of the work for the human
operator. It is possible to perform a more objective
assessment of sperm motility using a computer
assisted semen analyzer (Mortimer, 2000), which is a
laboratory based instrument that can measure
different aspects of the sperm movement. To further
increase precision, a combination of fluorescent
staining and flow cytometry can be used to analyze
thousands of sperm in a sample (Christensen et al,
2005). The common drawback with all of the above
techniques is the price of the equipment itself and the
need for a skilled operator.
In some species the sample may be successfully
frozen and thawed before use however this is not
always the case. The sperm of other species cannot
be successfully frozen and so a fresh sample, with a
shelf life of only a few days, must be used.
Increasingly, AI is being used for equine applications
and recent changes in UK legislation (Artificial
Insemination of Mares Order 2004) mean that lay
(non-Veterinary) and farm workers are now being
trained to perform such inseminations. For sport
equine applications the cost of semen for a single
mare to be covered may exceed £1000. Semen
analysis is currently a specialist, subjective and
skilled process that is normally carried out under
laboratory conditions. Given these trends, a low cost,
simple to use and objective technique to assess the
quality of the semen, particularly under field
conditions just before insemination, would greatly
13
Atherton S., R. Evans C., Roach P., C. Hughes D., McHale G. and I. Newton M. (2009).
INVESTIGATION OF OPERATING PARAMETERS FOR A SEMEN QUALITY ANALYSIS SYSTEM.
In Proceedings of the International Conference on Biomedical Electronics and Devices, pages 13-16
DOI: 10.5220/0001429600130016
Copyright
c
SciTePress

improve the quality and practice of artificial
insemination in animals. It would also provide an
easier and more cost effective method for monitoring
male animal fertility and breeding male welfare.
Acoustic wave sensors detect very small changes
in mass attached to their surface and often contain a
sensitizing layer that can recognize and bind the
species to be detected onto the mass sensitive
surface. The quartz crystal microbalance (QCM) is
the most widely used acoustic wave device for sensor
applications.
Δf = -2.26 x 10
-6
f
2
Δm/A (1)
The Sauerbrey equation (Sauerbrey, 1959) relates
the change of the crystals resonant frequency to the
change in rigid mass on the crystal surface; this is
shown for AT cut quartz in equation 1 where Δf (in
Hz) is the change in frequency that occurs for an
increase in mass Δm (in grams) on the surface of area
A (in cm
2
) with a crystal resonant frequency of f (in
Hz) and the constant comes from the crystal
materials properties. A well-designed oscillator
circuit can still resonate a crystal even under the high
damping caused by immersion in a liquid. The
change in mass rigidly attached to the surface still
causes a proportional change in frequency although
changes in other parameters such as the liquids
viscosity and density will also cause changes in
frequency. The acoustic wave will only sense mass
changes within a short distance into the liquid called
the penetration depth. (Kanazawa & Gordon, 1985)
Previous studies have shown up to 70% of the sperm
mass to be made up of water (Da Silva et al, 1992) so
it is not obvious how the attachment of a sperm will
change the QCM response. In a preliminary report
(Newton et al, 2007) we have fitted the resonance
curves of 5MHz QCM to the Butterworth van Dyke
model and this has shown that the sperm may be
treated a rigid mass and so a model based on the
Sauerbrey equation is appropriate when using an
effective mass of around 5pg. For other operating
frequencies or other species sperm this effective mass
would be different.
In this report we extend this preliminary work to
investigate the effect of environmental parameters on
the time of flight (ToF). For any practical
measurement technique it is essential the time the
measurement takes is sufficiently short to be usable.
For a portable field instrument then power
consumption may also be an issue therefore the first
parameter we investigate is operating temperature
and we consider a range from room temperature to
body temperature.
Progesterone is a steroid hormone involved in
female menstrual cycle, pregnancy and
embryogenesis of humans and other species. It is one
of a number of substances said to cause
hyperactivation of mammalian spermatozoa and its
presence may therefore affect the time of flight; the
effect of adding progesterone to the swim medium is
reported.
Figure 1: Magnified view of a boar sperm.
2 EXPERIMENTAL
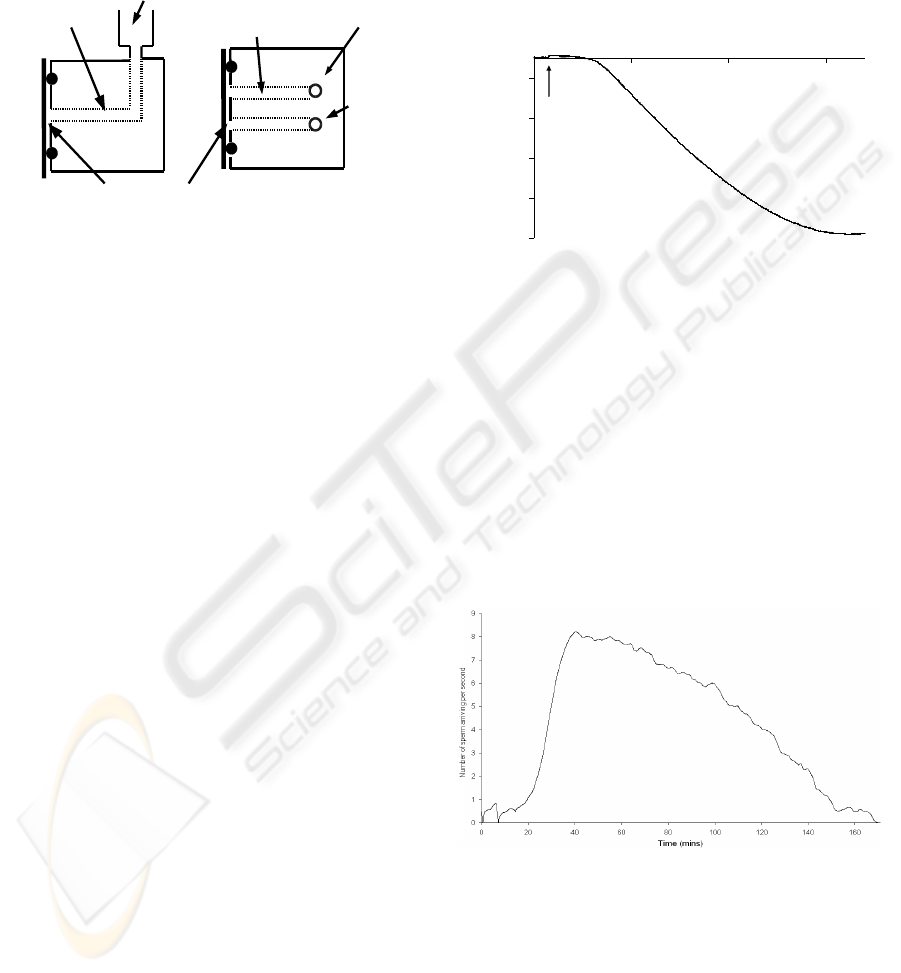
Figure 2 shows a schematic diagram of the
experiment. This consists of an inlet port to a channel
filled with phosphate buffered saline (PBS) buffer.
At the other end of the channel is a quartz crystal
followed by a vent to air preventing pressure changes
being recorded in the QCM response when a semen
sample is added. Sperm are introduced at the inlet
port and are self propelled through the channel to the
QCM where they are detected. A volume of 20μl of
the semen was used and added using a Gilson pipette.
The channel length was set to approximately 14.5 cm
and contained 4ml of PBS; note that for any practical
field instrument the swim channel length could be
considerably reduced to give an analysis time under 5
minutes. The sensing element in the experiments was
a 5MHz AT-cut quartz crystal (Testbourne 149211-
1). A Maxtek PLO-10 phase lock oscillator was used
to drive the crystal and the resonant frequency was
measured with an Agilent universal frequency
counter interfaced to a computer.
To sense the sperm it was necessary to get them
to adhere to the surface of the QCM. To achieve
sperm adhesion to the crystals they were coated in
either Poly-L-Lysine (Sigma-Aldrich) or cysteamine
(Sigma-Aldrich). Crystals were initially cleaning
with ethanol then ozone treated for 30 minutes. They
were then placed in either poly-L-lysine (as
supplied), or a cysteamine solution of 1mmol in
toluene and left overnight. The devices were then
BIODEVICES 2009 - International Conference on Biomedical Electronics and Devices
14
washed in PBS buffer to remove any excess. The
cysteamine coating were found to be the most
reliable method of binding the sperm to the surface as
poly-L-lysine shows significant variability from
batch to batch.
Side view
Top view
sample inlet port
buffer filled
channel
Quartz
crystal
inlet port
buffer filled
channel
vent
Sperm detection region
Figure 2: Schematic diagram of swim channel.
To provide temperature control to the
experiments the swim channel was housed inside an
Octagon 10 incubator. This allowed the temperature
to be controlled so as to investigate the effect this
would have on the sperm ToF. For these the
temperature was varied from 23-45
o
C. The incubator
also allowed the temperature to be kept constant
across all the progesterone experiments.
When looking at the effects of progesterone,
(Sigma-Aldrich) different concentration were added
to the PBS in the swim channel. Firstly 15.7mg of
progesterone was dissolved in 50ml of ethanol. This
mixture was diluted in PBS at concentration of 20-
90μmol.
The porcine semen was supplied by a
commercial artificial insemination centre (JSR
Genetics, Driffield, UK). The semen was received
already mixed with a dilutant (Androhep), cooled to
a temperature of 17
o
C and packaged in plastic
bottles. The androhep allowed the semen to be
stored for up to 5 days at ambient temperature.
However, this does result in the concentration of
semen in the mixture being quite low. To get a more
concentrated sample a centrifuge was used to
separate the sperm from the androhep. To achieve
this, sealable microtubes were filled with 50μl of the
androhep and semen mixture and these were
centrifuged for 40 seconds. This resulted in the
sperm being concentrated at the bottom of the tube.
The androhep was removed with the pipette and
50μl of PBS was added to one of the tubes. The PBS
and sperm were mixed together, this mixture was
removed with the pipette and added to the next tube
and the process was repeated for all 15 tubes. What
was left was a more concentrated sperm sample
mixed with 50μl of PBS.
3 RESULTS AND DISCUSSION
Figure 3 shows the QCM frequency response to
sperm binding on the surface. The arrow shows the
time at which the semen sample was introduce to the
inlet of the swim channel. The arrival of the sperm is
signified by a decrease in the frequency of the sensor
with the fastest ToF of approximately 20 minutes.
-45
-35
-25
-15
-5
0 50 100 150
time (minutes)
frequency change (Hz
)
Figure 3: Graph showing the frequency decrease
indicating sperm arrival.
The frequency continues to drop as more sperm
make their way to the QCM and bind to the surface.
This continues for another 120 minutes until further
arrival of motile sperm finishes.
Using the previously determined sperm effective
mass and taking the rate of frequency change from
figure 1, the Sauerbrey equation can be used to
derive the rate of sperm arrival and this is shown in
figure 3. For use in a screening application, a simple
threshold number of detected sperm would be
required however this demonstrates that quantitative
analysis is also possible with this instrument.
Figure 4: Number of sperm arriving at the QCM over the
course of the experiment.
Figure 5 is a plot of the ToF of the sperm against the
temperature of the environment. The results show a
decrease in the ToF as temperature increases with
almost a 50% fall between room temperature and
body temperature. The scatter observed can be
attributed mainly to the experiments being
performed a differing lengths of time from the
INVESTIGATION OF OPERATING PARAMETERS FOR A SEMEN QUALITY ANALYSIS SYSTEM
15
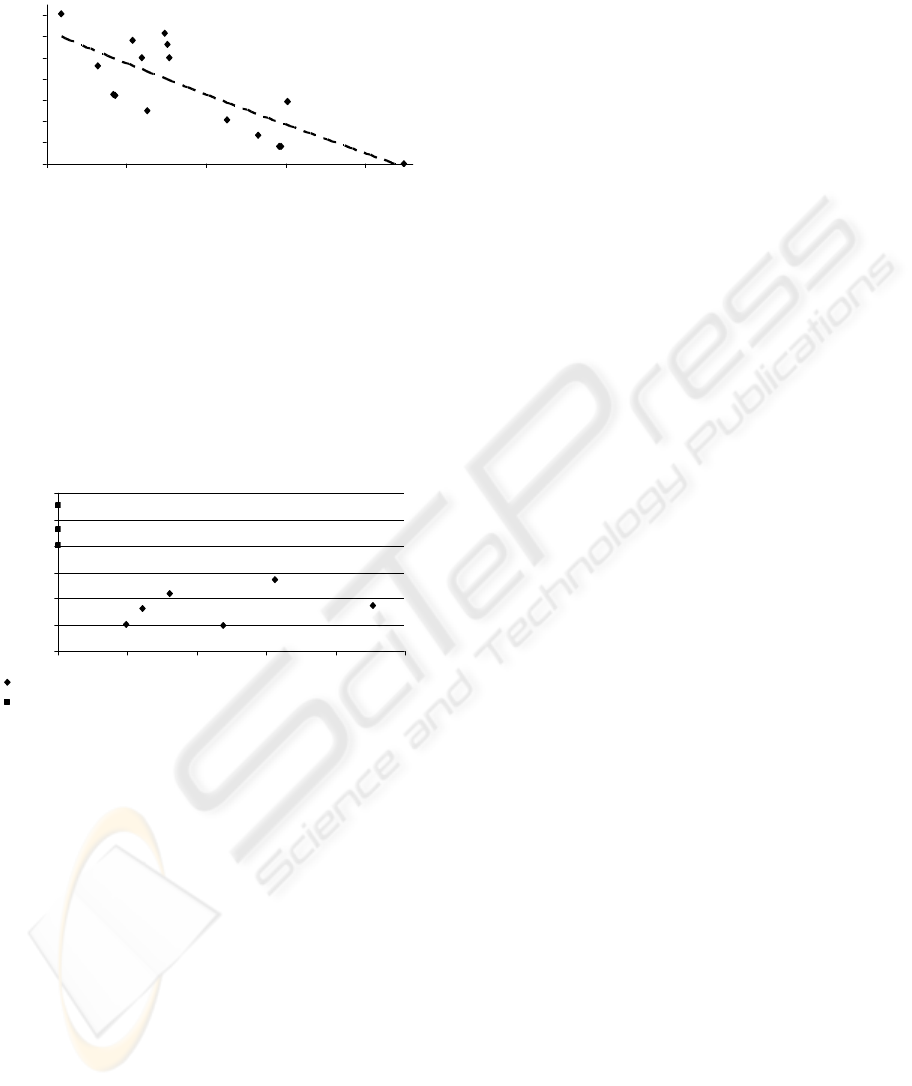
receipt of the samples and the quality of the semen
degrades over time.
10
12
14
16
18
20
22
24
22 27 32 37 42
Temperature (Deg C)
ToF (min)
Figure 5: The time of flight for fastest sperm arrival as a
function of temperature.
To further speed up the measurement,
progesterone was used to cause hyperactivation in an
attempt to decrease the sperm ToF; the results of this
are shown in figure 6. Comparing the non-
progesterone experiments with the progesterone
ones we see a significant decrease in the ToF of the
sperm however for the full range investigated there
was little effect from the progesterone concentration.
300
400
500
600
700
800
900
0.0 20.0 40.0 60.0 80.0 100.0
Progesterone concentration (μmol)
Time of flight (s)
Progesterone
No progesterone
Figure 6: Time of flight as a function of progesterone
concentration.
4 CONCLUSIONS
We have demonstrated that a time of flight
technique with an acoustic wave sensor provides a
viable method for determining the quality of a
semen sample both as a screening technique and as
an analytical tool. The cysteamine coating on the
QCM proved to be the more reliable method of
binding the sperm to the surface. Experiments
varying the temperature showed a general decrease
in ToF as temperature is increased suggesting that
body temperature would be the optimum value. The
presence of progesterone also reduces the ToF
however this was not concentration dependent over
the range investigated. Whilst the laboratory based
instrument reported here used commercial sensor
crystals, the cost of quartz crystals employed more
generally in electronic oscillator circuits are
inexpensive and are still offer a mass sensitive
surface. Using such crystals, pre-treated with
cysteamine, would reduce costs sufficiently to offer
the possibility of a disposable element. With a
modification to the swim channel length to bring
down the measurement time, this technique then
becomes a powerful tool for routine monitoring of
animal reproductive health and investigating factors
that affect the semen motility of animal.
REFERENCES
Mortimer, S. T., 2000, CASA - Practical Aspects, J.ournal
Andrology, 21, p.515-524
Christensen, P., Boelling, D., Pedersen, K. M., Korsgaard,
I. R., & Jensen, J., 2005, Journal of Andrology, 26
Sauerbrey, G., 1959, Zeitschrift für Physik, p.155-206
Da Silva, L. B., Trebes, J. E., Balhorn, R., Mrowaka, S.,
Anderson, E., Attwood, D. T., Barbee, T. W., Brase, J.,
Corzett, M., Gray, J., Koch, J. A., Lee, C., Kern, D.,
London, R. A., MacGowan, B. J., Matthews, D. L., &
Stone, G., 1992, X-ray laser microscopy of rat sperm
nuclei, Science, 258, p.269-271
Kanazawa, K., & Gordon, J. G., 1985, Frequency of a
Quartz Crystal Microbalance in contact with a liquid,
Journal of Analytical Chemistry, 57, p.1770-1771
Newton, M. I., Evans, C. R., Simons, J. J., & Hughes, D.
C., 2007, Semen quality detection using time of flight
and acoustic wave sensors, Applied Physics Letters
90(15)
BIODEVICES 2009 - International Conference on Biomedical Electronics and Devices
16
