PUNCTURE DEPTH AND THE MECHANICAL STABILITY OF
MICRONEEDLES
D. C. C. Lam, Y. H. Lee, K. T. Shek
Department of Mechanical Engineering, The Hong Kong University of Science and Technology
Clear Water Bay, Kowloon, Hong Kong SAR China
G. Pang
Department of Electrical and Electronic Engineering, The University of Hong Kong
Keywords: Drug delivery, microneedles, mechanical behaviour, punctures, silicone rubber.
Abstract: Microneedles penetrating less than 1mm beneath the skin can deliver the drugs directly without piercing
blood vessels or damaging nerves. The mechanical stability and the puncture behaviour were investigated
experimentally by inserting steel needles into silicone rubber and pig skin. Puncture tests revealed that the
length of needle buried in the flesh is less than 50% of the nominal insertion depth when the insertion depth
is less 1mm. The mechanical stability of the buried needle-flesh assembly, characterized by the force needed
to retract the needle, decreased with buried depth and needle diameter. Analysis of the load data suggested
that a 100-micron diameter microneedle buried 100 microns deep in pig skin would have a retraction force
of 0.1mN, which is only 1% of the retraction force of a conventional needle inserted 5mm into the skin.
This suggests that the usage of microneedles in arrays is necessary to increase stability and to enable stable
drug delivery.
1 DRUG INJECTION
Drug delivery in hospitals involves modalities as
diverse as hypodermal injection, intravenous
delivery and oral intake. Drugs taken orally must
pass through the digestive system and deal with the
first-pass effect of the liver. Numerous drugs,
particularly those developed in genetics laboratories,
are chemically incompatible with the oral intake
route (Orive et al., 2004) They must be directly
injected into the body. Hypodermal injection
involves training, some pain and potential blood
loss, making self-injection at home unwelcome for
patients.
Other than direct injection, drugs can be
delivered via absorption through the skin. The skin
is a natural barrier to foreign chemicals and
biological agents. A few drugs such as nicotine and
its substitutes have been customized for absorption
by the skin. Chemical customization of individual
drugs for such absorption is technically challenging,
and in many cases, economically unfeasible. Instead
of traversing the skin via absorption, solid or hollow
microneedles can be used to physically puncture the
skin. Drugs can pass through a capillary-scale
channel in the hollow needle, or can be delivered as
a soluble coating on the needle surface. For such.
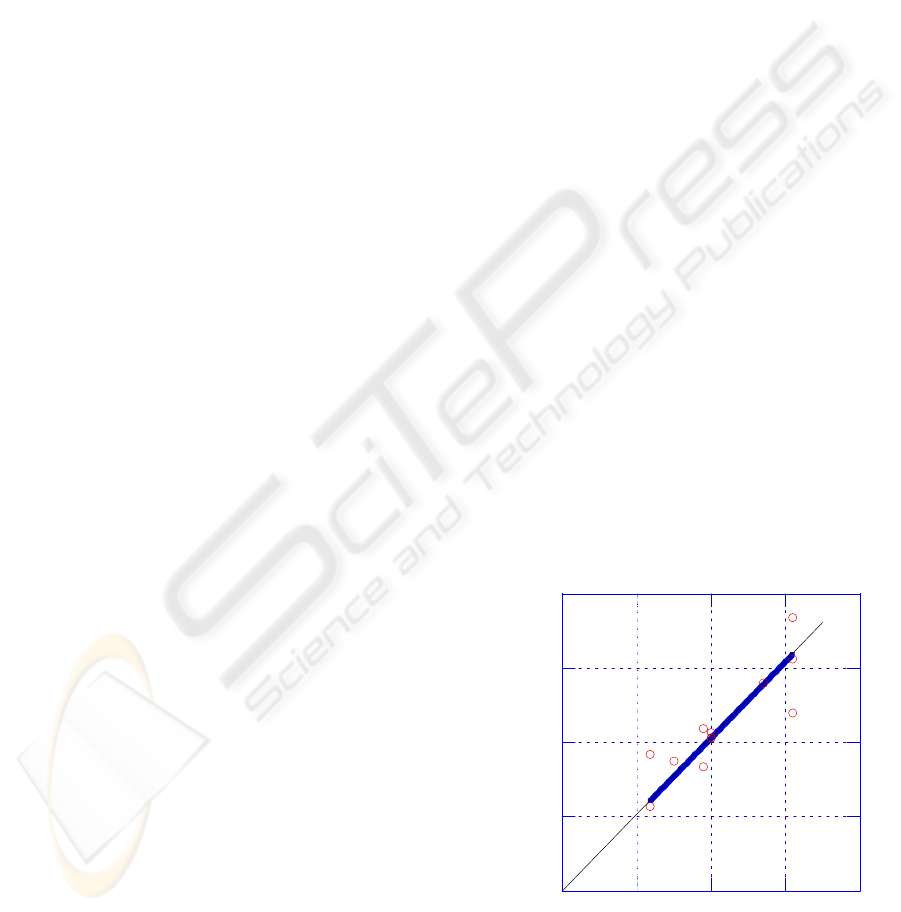
0
0.05
0.1
0.15
0.2
00.511.52
Liver insertion
Retraction force (N/mm)
Needle diameter (mm)
m=0.1N/mm/mm
Figure 1: Retraction load for needles with conical and
beveled tips. Data from (Okamura).
291
C. C. Lam D., H. Lee Y., T. Shek K. and Pang G. (2008).
PUNCTURE DEPTH AND THE MECHANICAL STABILITY OF MICRONEEDLES.
In Proceedings of the First International Conference on Biomedical Electronics and Devices, pages 291-296
DOI: 10.5220/0001054802910296
Copyright
c
SciTePress
applications, the microneedles must be long enough
to bridge the surface skin layer, but short enough to
avoid piercing the blood vessels and nerves that are
typically 0.5 – 1mm under the skin surface
There is now a large body of knowledge,
processes and tools that have been developed for the
design and fabrication of micro- and
nanoelectromechanical systems (MEMS and
NEMS). A number of researchers have developed
silicon microneedles using MEMS processes.
(Stoeber & Liepmann, 2005; Staples et al., 2006; Ji,
et al., 2006). Silicon microneedle arrays have been
fabricated successfully and tested in trials with rats
(Nordquist et al., 2007). These tests were concept
demonstrations showing that drugs can be delivered
using microneedles. However, a variety of issues,
including safety concerns about the use of brittle
silicon microneedles, remains to be solved. Titanium
and polymers are potentially better material choices,
and the fabrications of titanium and polymeric
microneedles (Parker et al., 2007; Park, Allen &
Prausnitz, 2006) has been investigated using
MEMS-based processes.
As another alternative to MEMS-based
technologies, lasers have been used to create arrays
of holes in substrates (Davis et al., 2005).
Microneedle pads have been created by plating a
metal shell onto the holed surface. The microneedles
in arrays fabricated in this fashion are hundreds of
microns long, with walls limited to several microns
thick because of plating limits. Silicon or metal
microneedles fabricated in this way are inherently
small because the processes are inherently micron
and submicron processes. Recognizing the inherent
process limits, microinjection molding suited for
fabricating millimeter-scale parts has been used to
make polymer microneedles (Sammoura et al.,
2007).
Conventional hypodermic needles are typically
more than a few millimetres long with diameters
typically ranging from 220 microns on up. Such
needles are designed to puncture skin and deliver
drugs deep underneath the skin. They are not
designed for drug delivery at insertion depths of
1mm or less. Limited studies had been conducted to
examine systematically the puncture behaviour of
such large needles at deep and shallow depths. Long
needle puncture studies were conducted by Nguyen
& Vu-Khanh on rubber sheets (Nguyen & Vu-
Khanh,, 2004) and by Sherwood’s group (Sherwood
& Fleck, 2004) on solid rubber. They observed
significant indentation deformation occurred before
puncture in these tests.
For microneedles, where penetration is less than
1mm into the skin, indentation is expected to be
significant and to affect drug delivery. The effect of
needle diameter had been examined in a study of
needle insertion into the liver (Okamura, 2004). The
study revealed that the force required to retract the
needle was strongly dependent on the needle
diameter and the insertion depth (Figure 1). Small
microneedles inserted into the surface of the skin
should have retraction forces in the mN range and
each individual puncture may have low mechanical
stability. The effect of depth and needle diameter on
the insertion and retraction behaviour were
examined this study.
2 INVESTIGATIVE APPROACH
The puncture behaviour of single steel needles
pressed to depths of 0.2 to 8mm was examined in
this study. Following the methods pioneered by
(Sherwood & Fleck, 2004), microstructurally
uniform and homogeneous silicone rubber was used
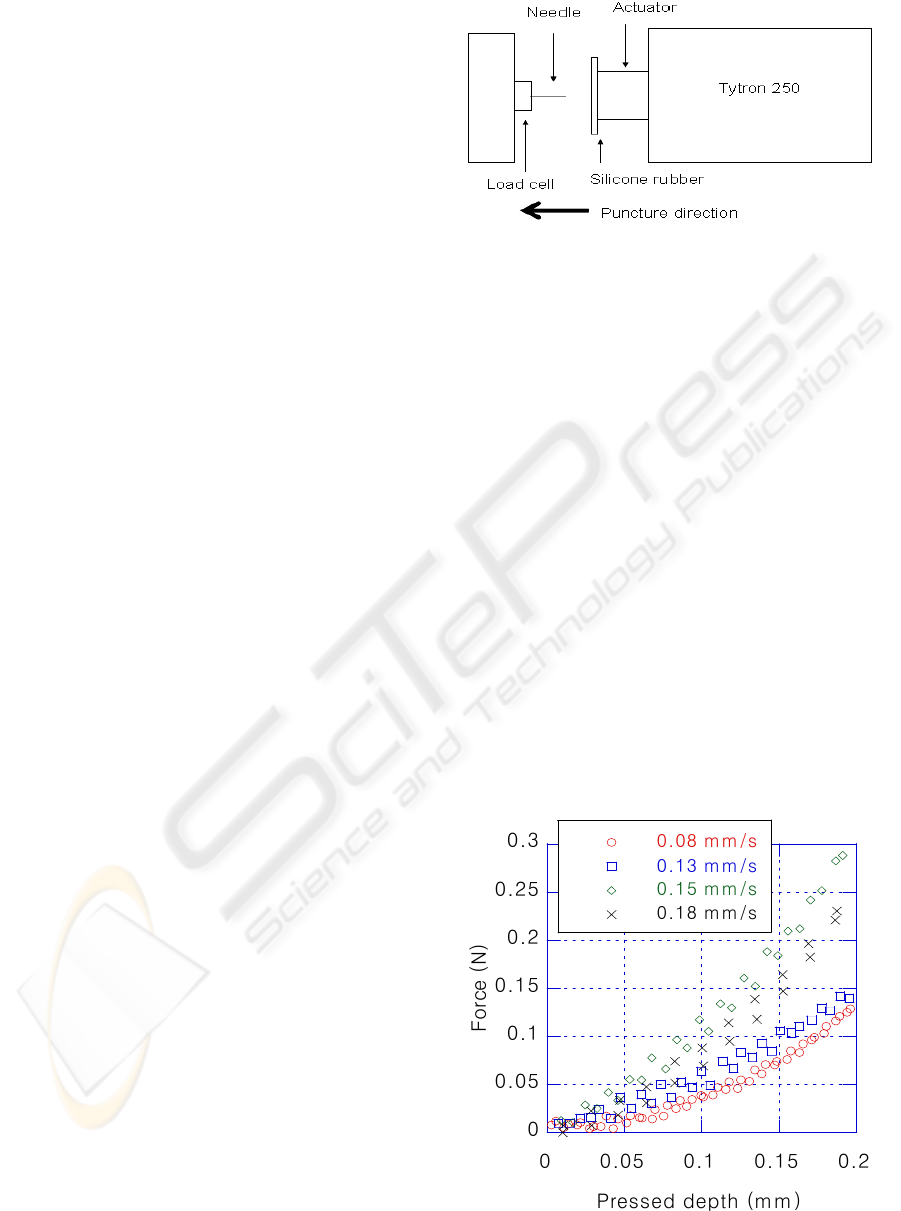
Figure 2: Mechanical testing setup.
Figure 3: 130-micron needle insertion behaviour i
n
silicone rubber at 0.2mm pressed depth.
BIODEVICES 2008 - International Conference on Biomedical Electronics and Devices
292
as a reference puncture material to examine the
puncture behaviour. The puncture behaviour of pig
skin was also examined to determine the load and
depth range of puncture behaviour in biological
materials.
The puncture experiments were conducted on a
Tytron 250 press from MTS Systems Corporation
equipped with a linear drive actuator. Silicone
rubber blocks were cast from a Shin-Etsu silicone
premix and cured according to the manufacturer’s
specifications. Medical grade stainless steel needles
130, 220 or 300 microns in diameter were mounted
on a custom designed jig for puncture testing (Figure
2). Once mounted, the loads and positions were
calibrated. The needle was held fixed while the
silicone rubber casting was moved toward the needle
at displacement rates of 0.1 mm/s, 0.13 mm/s, 0.15
mm/s or 0.18 mm/s to a target maximum pressed
depth (MPD), and then reversed at the same rate.
The experiment ended when the displacement
reached zero. The experiment was repeated for a
range of MPDs from 0.2 mm to 8 mm.
3 RESULTS AND ANALYSES
3.1 Shallow Insertion Depths
The load-displacement results at 0.2mm MPD are
shown in Figure 3. The curves indicate that the peak
load was displacement rate dependent. At each
displacement rate, the unload curve collapsed onto
the load curve. This indicates that the deformation
recovered elastically without puncture. The absence
of a hysteresis between the load and unload curves
indicates that no energy was dissipated at 0.2mm
MPD. In addition, the higher peak loads at higher
displacement rates did not result in puncture. This
suggests that needles less than 0.2mm long have
difficulty in puncturing soft silicone rubber.
3.2 Puncture Depth
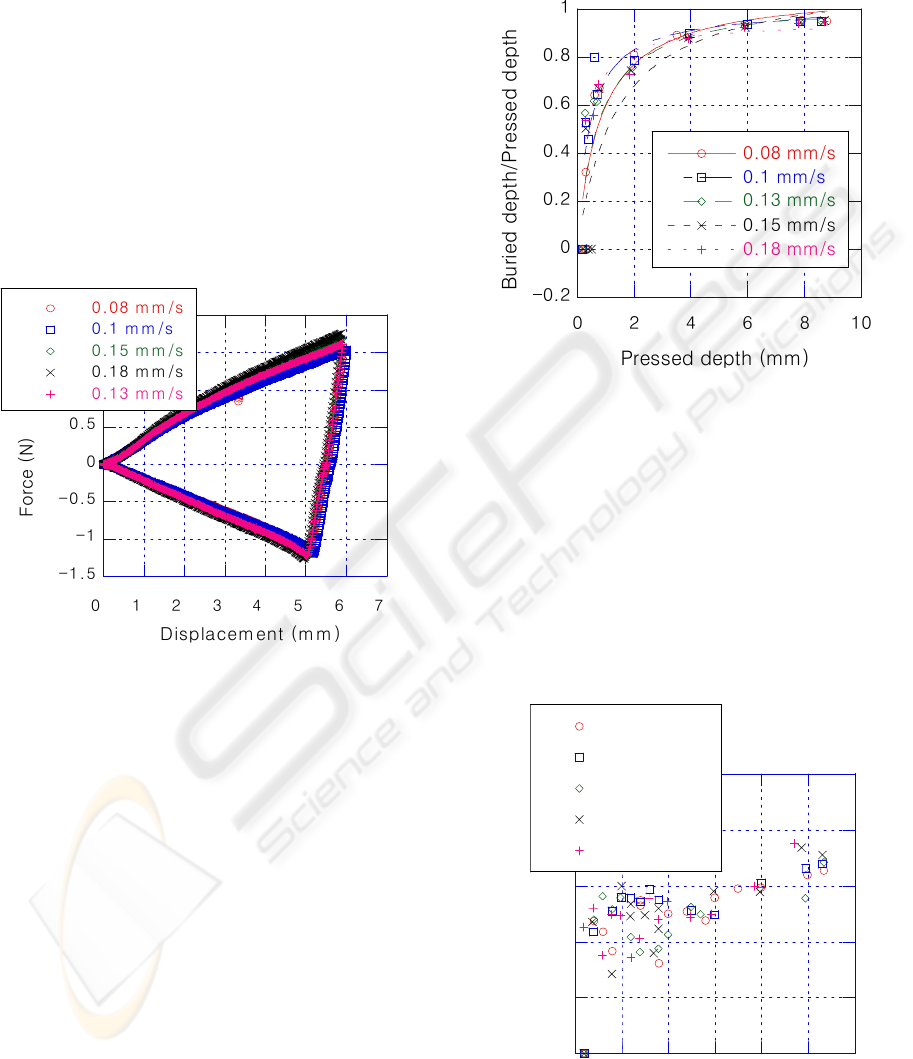
The results at 6mm MPD are plotted in Figure 4. As
with the tests in Figure 3, the displacement was
reversed from loading to unloading in each trial
upon reaching the 6mm maximum set displacement.
The unloading curves did not retrace the loading
Figure 4: 130-micron needle insertion behaviour in silicone
rubber at 6mm maximum pressed depth.
Figure 5: Ratio of buried to pressed depth for needle
insertion into silicone rubber using 130-micron needles.
0
0.2
0.4
0.6
0.8
1
0123456
0.08 mm/s
0.1 mm/s
0.13 mm/s
0.15 mm/s
0.18 mm/s
Buried depth/Pressed depth
Maximum Pressed depth (mm)
Figure 6: Ratio of buried to pressed depth for needle
insertion in pig skin using 130-micron needles.
PUNCTURE DEPTH AND THE MECHANICAL STABILITY OF MICRONEEDLES
293
curves, but rather intersected the zero load line to the
right of the triangle, crossing into the tension region.
Unlike the penetration behaviour at 0.2mm MPD,
where the needle was fully withdrawn at zero load,
the needle retracting from 6mm MPD remained
buried in the rubber. This zero load depth on
retraction was taken to represent the buried depth on
insertion. A plot of the buried needle depth
normalized by the MPD is plotted in Figure 5. At
large MPDs, the ratio approached unity, but at
MPDs less than 1mm, the ratio was significantly less
than one. The low ratios at settings below 1mm
indicate that an increasingly significant proportion
of the needle was left outside the solid during the
loading phase.
The MPD for needle insertion into pig skin is plotted
in Figure 6. Biological materials are less uniform
and the ratio is more scattered. Despite this, the
trend is similar to that observed with the silicone
rubber in that the ratio approaches unity at large
pressed depth, but is 0.5 or less when the pressed
depth was 2mm or less.
3.3 Retraction Force
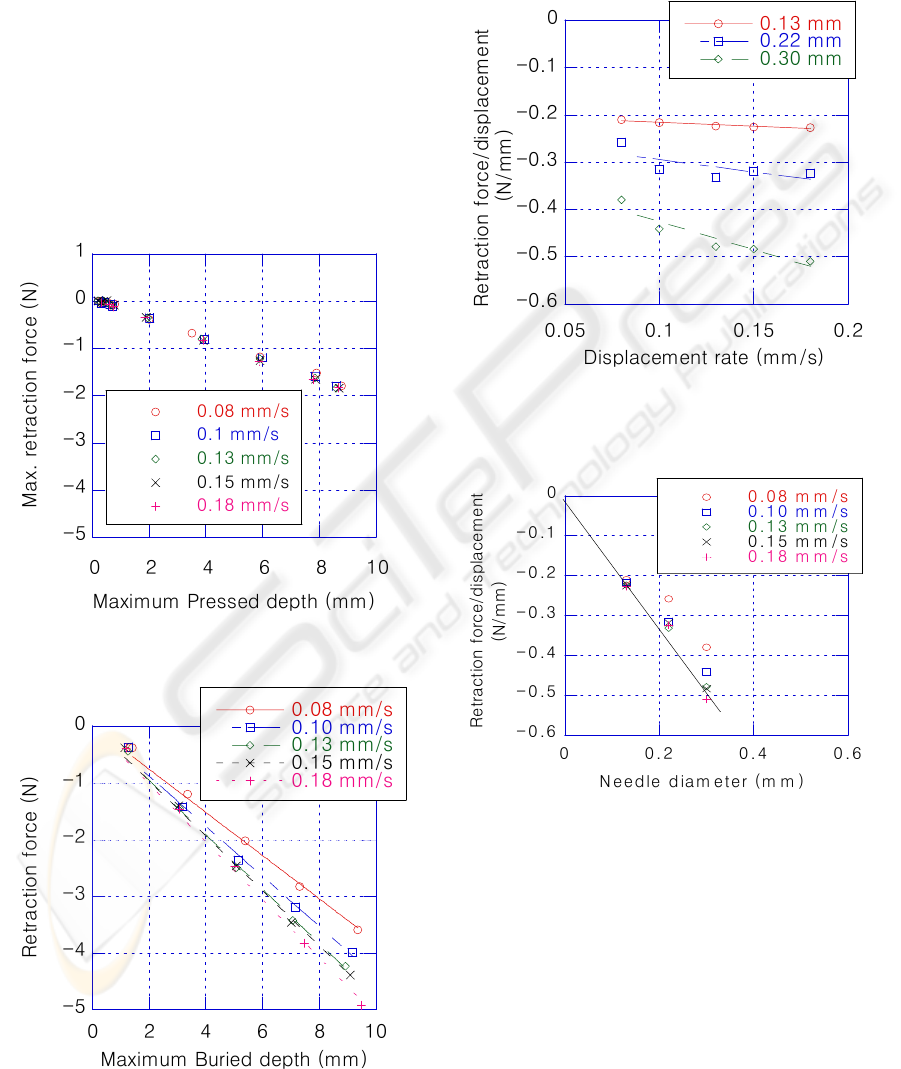
The maximum retraction load (MRL; bottom right
apex in Figure 3) required to pull a 130-micron
needle out after pressing is shown in Figure 7. The
data show that the retraction forces at different
displacement rates collapsed onto a single line for
the 130 micron-needle. The MRL for the 300-
micron needle is plotted in Figure 8. The MRL for
this larger needle was clearly displacement
Figure 7: Maximum retraction force for 130-micron
needle insertion into silicone rubber.
Figure 8: Maximum retraction force for a 300-micron
needle inserted into silicone rubber.
Figure 9: Rate dependence of retraction force per unit o
f
b
uried needle length for needle insertion into silicone
rubber.
Figure 10: Diameter dependence of retraction force
p
e
r
unit of buried depth in silicone rubber.
BIODEVICES 2008 - International Conference on Biomedical Electronics and Devices
294
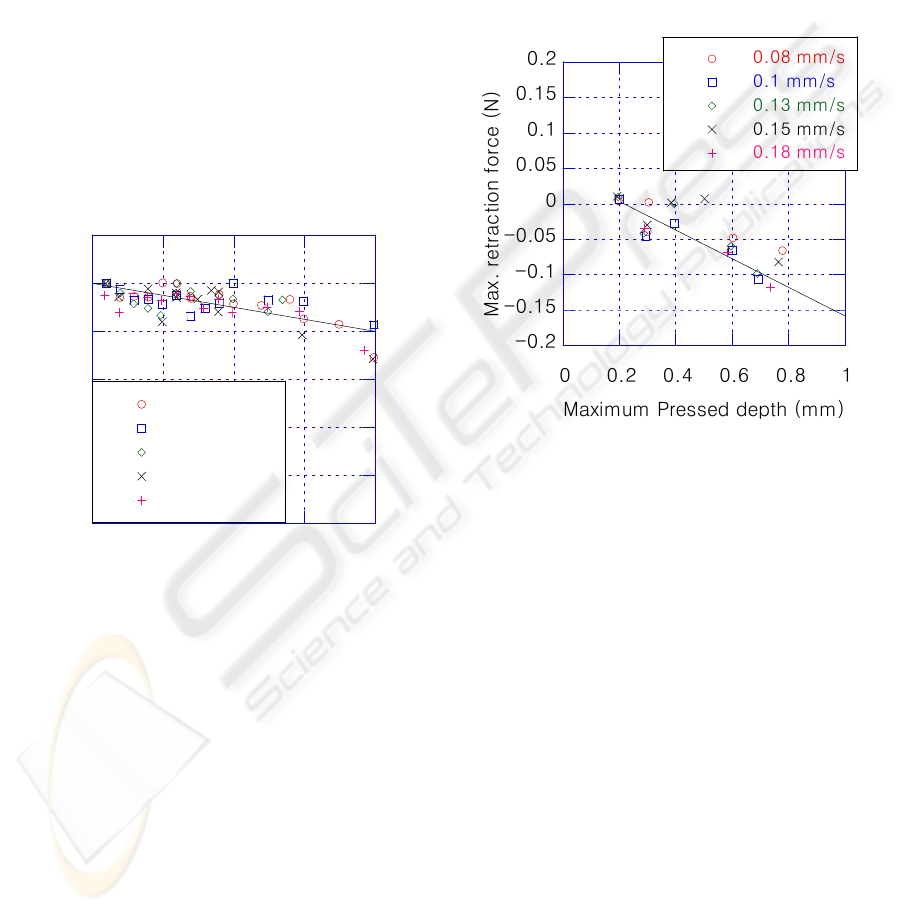
-0.25
-0.2
-0.15
-0.1
-0.05
0
0.05
01234
0.08 mm/s
0.1 mm/s
0.13 mm/s
0.15 mm/s
0.18 mm/s
Retraction force (N)
Maximum Pressed depth (mm)
Figure 11: Comparison of maximum retraction force wit
h
model (line) at shallow pressed depth.
dependent. The MRL was markedly higher at higher
retraction rates. Thus, while the 130-micron needle’s
rate dependence appeared negligible, this was not
the case with larger diameter needles. The rate of
change of the MRL with buried depth as a function
of the displacement rate is plotted in Figure 9. The
plot shows that the rate dependence was linear for all
the needles tested, and that the MRL decreased with
decreasing needle diameter. The influence of needle
diameter is plotted in Figure 10. The MRL was
greatest at high displacement rates. Fitting a straight
line through the data with the highest displacement
gives a slope of -1.6N/mm/mm, which is the MRL
per unit of buried depth and needle diameter. Using
this as a basis for estimate, in Figure 11 a line is
plotted and compared with the retraction loads
observed with a 130-micron needle at depths less
than 1000 microns. The comparison showed
reasonable agreement between the data and the
prediction.
The MRL for insertion into pig skin is plotted in
Figure 12. As expected, the magnitude of the MRL
increased with the actual buried depth, but the
displacement rate dependence cannot be delineated
given the greater scatter. The rate of change of the
MRL per unit of buried depth and needle diameter
was approximately 0.1N/mm/mm.
4 DISCUSSION
The length of the needle buried in the flesh during
hypodermal insertion is generally assumed to equal
to the pressed depth. This assumption is reasonable
for deep insertions, but breaks down when the
needle is less than 2000 microns long. At pressed
depths under 1000 microns, less than half of the
needle maybe buried; and no penetration was
observed at pressed depths less than 200 microns.
Since the lengths of microneedles fabricated using
MEMS processes are limited by the thickness of the
wafer, which is typically less than 300 microns
thick. Silicon microneedles less than 300 microns
long may have difficulty in puncturing the skin. If
punctured, the maximum buried depth will likely be
less than 100 microns.
Another issue is the mechanical stability of the
microneedles. When punctured, the surrounding
material in the puncture will exert traction onto the
buried needle and prevents it from slipping out of
the puncture hole. The retraction force was found to
decrease at a rate of -1.6N/mm of buried depth/mm
of needle diameter for silicone rubber, and -
0.1N/mm/mm for pig skin (Figure 12). In
comparison, the corresponding value for the case of
needle puncture into liver is -0.1N/mm/mm. Using
this as an estimate for biological materials, a 0.1mm
microneedle buried 0.1mm into the skin would have
a retraction force of 1mN. A conventional 0.22mm
needle inserted into flesh 5mm would have a
retraction force of 110mN, which is 100 times
greater. If an array of 110 microneedles were used,
the retraction force can be increased to 100mN such
that the microneedles can remain stably buried to
allow stable drug delivery. The puncture behavior
and the design guidelines for stably drug delivery
are reported in another paper.
Figure 12: Retraction force for 130-micron insertion in pig
skin.
PUNCTURE DEPTH AND THE MECHANICAL STABILITY OF MICRONEEDLES
295

5 CONCLUSIONS
Examination of the puncture behaviour needles with
a silicone rubber model indicated that indentation
effects are significant at shallow insertion depths for
both silicone rubber and pig skin. Less than 50% of
the needle may be buried because of indentation
when short microneedles are used. The investigation
also revealed that the retraction force providing
mechanical stability to the needle in the skin was
linearly proportional to the buried depth and the
needle diameter. Short microneedles with small
diameters will have low retraction forces and poor
mechanical resistance against being dislodged.
ACKNOWLEDGEMENTS
This work was supported by grants from Hong
Kong’s Research Grants Council. The authors also
acknowledge the use of equipment provided by the
Advanced Engineering Materials facility at the
HKUST.
REFERENCES
Orive, G., Gascon, A.R., Hernandez, R.M., Dominguez-
Gil, A., Pedraz, J.L., 2004. Techniques: New
approaches to the delivery of biopharmaceuticals,
Pharma. Sci., Vol 25 No. 7 pp. 382-7.
Stoeber, B., Liepmann, D., 2005. Arrays of hollow out-of
plane microneedles for drug delivery, J. MEMS Vol
14 No. 3 pp. 474-9.
Staples, M., Daniel, K., Cima, M.J., Langer, R., 2006.
Application of micro- and nano-electromechanical
devices to drug delivery, Pharma Res. Vol 23 No. 5
pp. 847-63.
Ji, J., Tay, F.E.H., Miao, J., Iliescu, C., 2006.
Microfabricated microneedle with porous tip for drug
delivery, J. Micromech. Microeng. Vol 16 pp. 958-
964.
Nordquist, L., Roxhed, N., Griss, P., Stemme, G., 2007.
Novel microneedle patches for active insulin delivery
are efficient in maintaining glycaemic control: An
initial comparison with subcutaneous administration,
Phama. Res. (in print).
Parker, E.R., Rao, M.P., Turner, K.L., Meinhart, C. D.,
MacDonald, N.C., 2007. Bulk micromachined titanium
microneedles, J. MEMS Vol 16 No. 2 pp. 289-95.
Park, J., Allen, M.G., Prausnitz, M.R., 2006. Polymer
microneedles for controlled-released drug delivery,
Pharma. Res. Vol 23 No. 5 pp. 1008-19.
Davis, S. P., Wijaya, M., Allen, M. G., Prausnitz, M. R.,
2005. Hollow metal microneedles for insulin delivery
to diabetic rats, IEEE Trans. Biomed. Eng. Vol 52
No.5 pp. 909-15.
Sammoura, F., Kang, J.J., Heo, Y., Jung, T.S., Lin, L.,
2007. Polymeric microneedle fabrication using a
microinjection molding technique, Microsys. Tech..
vol 13 Issue 5 pp. 517-22.
Nguyen, C.T., Vu-Khanh, T., 2004. Mechanics and
mechanisms of puncture of elastomer membranes, J.
Mater. Sci. Vol 39 No.4 pp. 7361-4.
Sherwood O.A., Fleck, N.A., 2004. Mechanisms of deep
penetration of soft solids with application to the
injection and wounding of skin, Proc. R. Soc. Lon. A
vol. 460 pp. 3037-68.
Okamura, A. M., Simone, C., O’Leary, M. D., 2004.
Force Modeling for Needle Insertion Into Soft
Tissue,IEEE Trans. Biomed. Eng. Vol 51 No.10 pp.
1707-1716.
BIODEVICES 2008 - International Conference on Biomedical Electronics and Devices
296
