MICROCOMPUTERIZED SYSTEM TO ASSESS THE
PERFORMANCE OF INFANT INCUBATORS
Mário Anderson de Oliveira, Maurício Campelo Tavares
Institute of Biomedical Engineering, Federal University of Santa Catarina, campus trindade
88040 – 970, Florianópolis, SC, Brazil
Laboratory of Biomedical Engineering, Catholic University of Pelotas, campus I, number: 412
Félix da Cunha 109C, 96010-000, Pelotas, RS, Brazil
Raimes Moraes
Department of Electrical Engineering, Federal University of Santa Catarina, 88040 – 900, Florianópolis, SC, Brazil
Keywords: NBR IEC 601.2-19 (1999), Bluetooth, neonate incubators, clinical engineering, assessment.
Abstract: This work presents a system based on a microcomputer to assess the performance of infant incubators in a
semi-automatic manner. It carries out the tests described by the section 8 of the NBR IEC 601- 2-19 (1999).
The developed electronic circuit acquires data from the sensors using a microcontroller. A set of sensors are
used: five for temperature, one for humidity and one for air flow. The sampled data is sent to the PC via
Bluetooth. The software running on the PC manages the data sampling, as well as guides the user through
the test procedure by means of messages and sound alerts at the end of each stage. The sampled data is
shown on the screen and also stored in a database that can be remotely accessed. The results are presented
on a graph where the measurements (temperature, humidity and air flow) performed during the whole test
can be seen. The procedure to calibrate the sensors and an infant incubator assessment carried out with the
developed system is presented.
1 INTRODUCTION
There is a high percentage of mortality associated to
low birth weight newborns (Ministério da Saúde,
2002). These neonates have an immature thermal
control mechanism, preventing them to keep
constant their body temperature (Gonzáles, 2001).
The infant incubator (InI) aims to provide a thermo-
neutral environment where the infant does not
exchange heat, consuming a minimum amount of
oxygen. This environment is obtained by controling
the temperature, humidity and air flow. Inside the
InI, the infant has a reduced metabolism that helps
its healthier and faster growth (Ministério da Saúde,
2002). Therefore, InIs shall be periodically checked
to assure that they offer a suitable environment to
the neonate.
This work describes a system developed to
assess the InI performance according to the Section
8 of the NBR IEC 601-2-19 (1999).
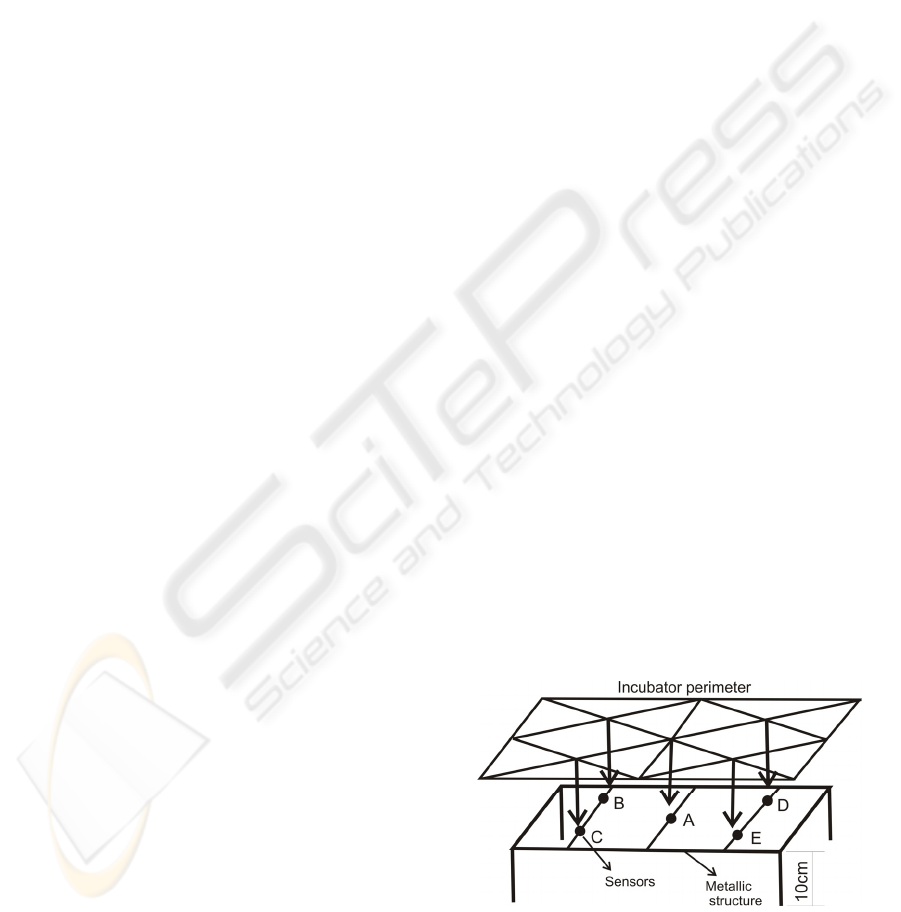
To evaluate the InI performance, the standard
demands measurements of the following parameters:
air temperature at five different points, relative
humidity and air velocity. The temperature sensors
are placed at five points 10 cm above the mattress
surface (A, B, C, D and E) as shown in Figure 1.
Figure 1: The NBR IEC 601-2-19 demands temperature
measurements at 5 different points (10 cm above the
mattress surface): A, B, C, D and E. A metallic structure is
used to hold the sensors at the required positions.
119
Anderson de Oliveira M., Campelo Tavares M. and Moraes R. (2008).
MICROCOMPUTERIZED SYSTEM TO ASSESS THE PERFORMANCE OF INFANT INCUBATORS.
In Proceedings of the First International Conference on Biomedical Electronics and Devices, pages 119-122
DOI: 10.5220/0001053501190122
Copyright
c
SciTePress
2 MATERIALS AND METHODS
The performance testing required by the standard is
relatively difficult to be carried out manually, being
desirable to automate it. Thus, a microcontrolled
system was developed to automatically sample InI
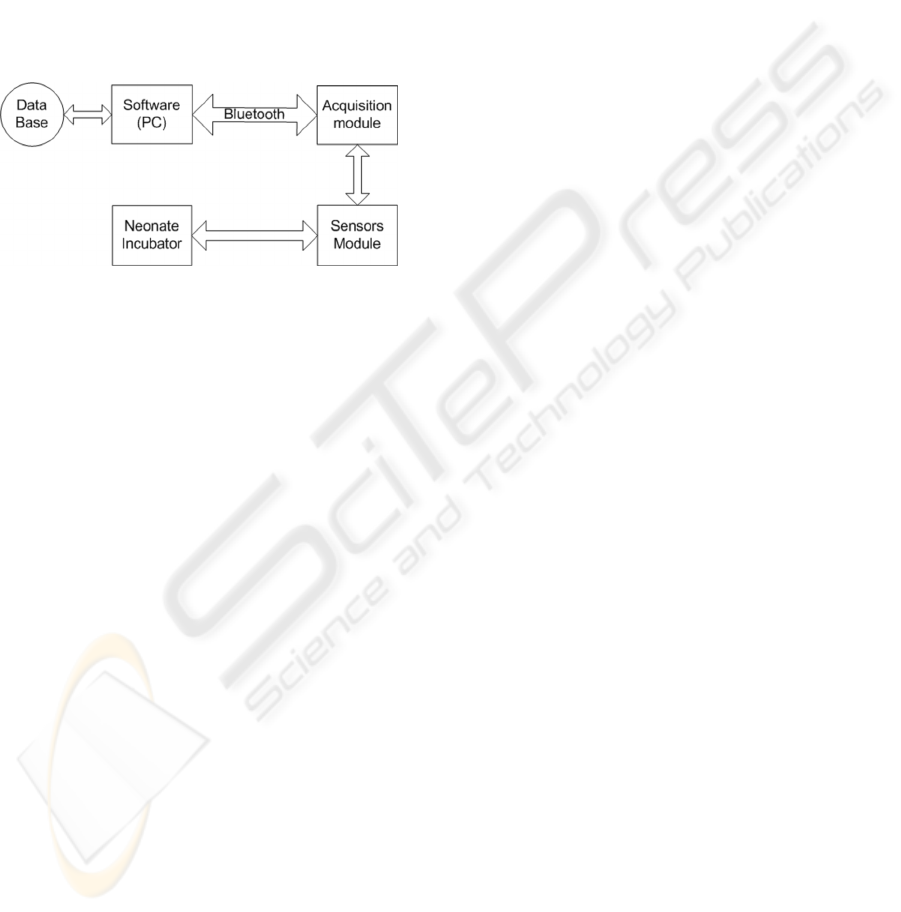
data that are sent to a microcomputer (Figure 2).
The developed system consists of 5 modules:
sensors, acquisition, communication, control
software and database.
The remote control software running on a PC
communicates with the acquisition module to
require samples from the sensors placed into the InI.
Figure 2: The developed system has five modules: sensor,
acquisition, communication (Bluetooth), control software
and database.
2.1 Sensors Module
The sensors are placed into the InI as shown by the
Figure 1.
The sensor SHT75 (Sensirion Inc) is used to
measure the Incubator Temperature (temperature at
the point A of Figure 1) and the relative humidity
(RH). It measures temperature in a range from -40 to
123.8°C and RH from 0 to 100%RH. For
temperature, the SHT75 has a typical accuracy of
±0.5°C and resolution of 0.01°C. For humidity,
typical accuracy of ±1.8%RH and resolution of
0.03%RH.
The measurements executed by the SHT75 are
available in digital format via the 2-wire protocol.
This is a bi-directional protocol, allowing the sensor
to receive commands as well.
To measure the temperature at the other points
(B, C, D and E), 4 TMP05 (Analog Devices) sensors
are used. The TMP05 measures temperature in a
range from -40 to 123.8°C with a typical accuracy of
±0.5°C and resolution of 0.01 °C.
The TMP05 sensors can be connected in
cascade, establishing a net. Thus, only two
microcontroller pins are needed to acquire the
temperature measurements from the sensors: one
generates a start pulse and the other receives the
PWM signal.
The sensor F900 (Degree Controls Inc.) is used
to measure the air velocity in the InI. It has a linear
output voltage for a range from 0.15 to 2m/s with a
typical accuracy of ±0.05m/s and resolution of
0.05m/s.
2.2 Acquisition Module
The acquisition module contains the microcontroller
ADuC841 (Analog Devices) that has the following
characteristics: 8052 core, 20 MIPS, 8 ADC
channels (12 bits), 2 DAC channels (12 bits), 3
timers/counters (16 bits) and serial communication
interfaces (UART, I2C and SPI).
The ADuC establishes serial communication
with the SHT75 and TMP05 to get the humidity and
temperature measurements. Its ADC samples the
F900 voltage output to figure the air velocity out.
These measurements are sent to the PC when
demanded by the control software.
2.3 Communication Module
The Bluetooth module implements wireless link
between the acquisition module and the PC. To
provide a communication range up to 100 meters, a
KC-11 unit (KCWirefree) is connected to the
acquisition module and a KC-210 is inserted into the
PC USB port.
A virtual serial port driver is used by the
control software to communicate with the KC-210.
2.4 Control Software Module
About 6 hours are necessary to carry out the
measurements required by the NBR IEC 601-2-19
(1999). To simplify the task, a control software was
developed for WindowsOS® in Borland C++
Builder.
At 20 second intervals, the software demands
a new set of measurements. For that, an ASCII
command is sent to the acquisition module. After
receiving the command, the microcontroller
communicates with the sensors to get the
measurements that are sent back to the PC. The
received data are presented on the PC screen and
stored into a database.
Besides managing the measurements, the
developed software guides the user through the
testing procedure by means of messages and sound
alerts at the end of each stage. The software displays
messages on the PC screen asking the user to change
the InI settings or the mattress position. After doing
so, the user shall click the OK button of the message
BIODEVICES 2008 - International Conference on Biomedical Electronics and Devices
120
box. Then, the software keeps executing the
measurements.
2.5 Database
The database (DB) was developed with PostgreSQL
8.0, a free software object-relational database
management system (Matthew and Stones, 2005).
The implemented DB has two entry tables, one
registers the equipment identification key; the other
stores the measurements carried out during the InI
testing.
3 SENSORS CALIBRATION
The F900 and SHT75 sensors are individually
calibrated. They both have calibration certificate.
The F900 is supplied with its calibration curve. The
SHT75 has calibration coefficients programmed into
its internal memory.
The TMP05 sensors were calibrated using the
SHT75 as reference since there was no other
traceable reference sensor with better accuracy and
resolution available in this laboratory.
To calibrate the four TMP05 sensors, they were
placed together with the reference sensor into a
container with low heat transmission walls. The air
inside the container was heated up to 60ºC and then,
the heat source was turned off. During the air
cooling, 17 sets of temperature measurements were
obtained for each sensor within the range from 25 to
41ºC. For each sensor, a third order polynomials was
fitted to the experimental data to correct the
systematic error observed with respect to the
reference sensor. These polynomials are used by the
software running on the PC to reduce the
measurement errors.
To evaluate the described calibration, 3
measurements were executed for 13 different
temperatures within the calibrated range to find out
the resultant errors. This procedure was repeated for
each sensor.
For each sensor, the systematic error (bias) and
the random error (repeatability) were surveyed.
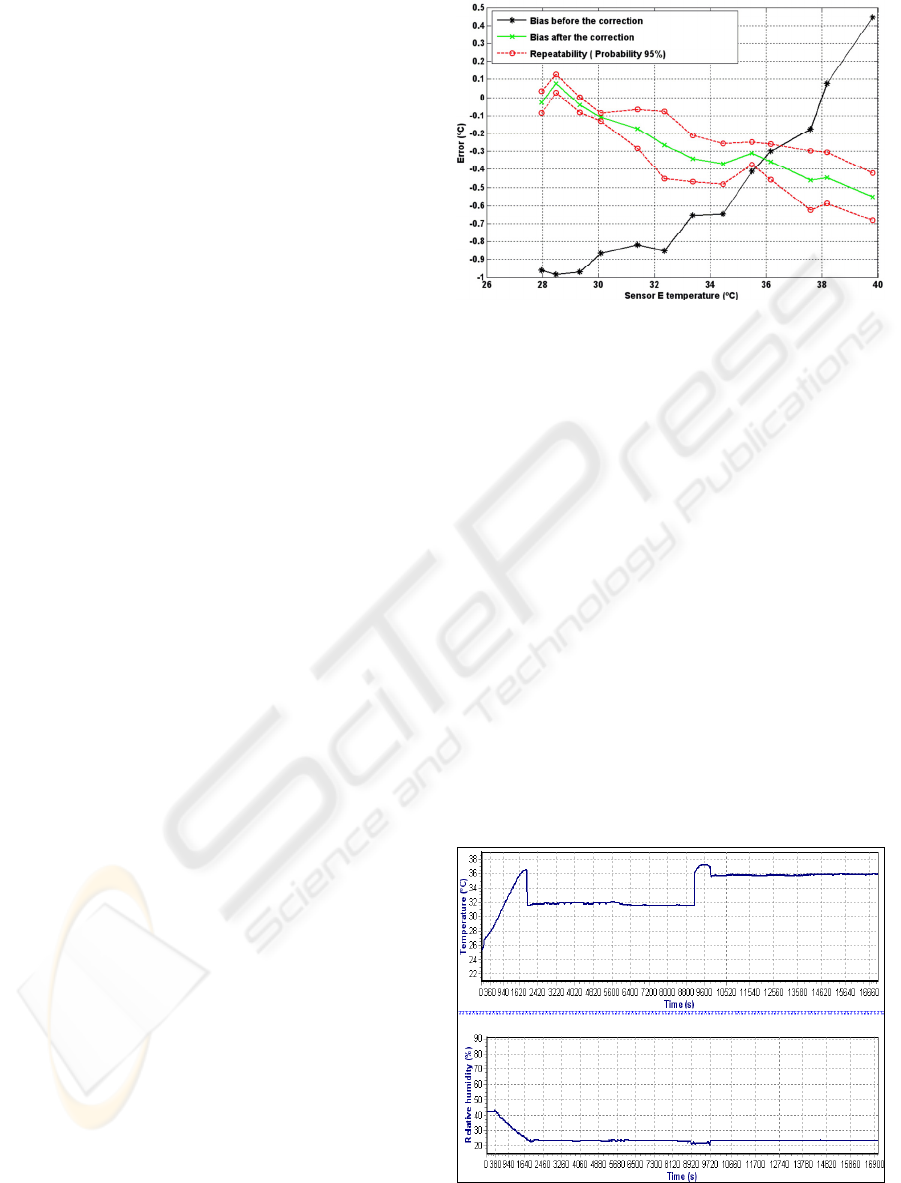
As example of the result achieved with this
sensor calibration procedure, the Figure 3 shows the
curves obtained for the sensor to be placed at the
point E (Figure 1). This one presented the larger
errors (about ± 0.5ºC) within the InI operating range
(32 to 36ºC).
Figure 3: Measured error curve versus temperature is
plotted for the sensor E. It is also shown the systematic
error (before and after the application of the polynomial
adjustment) and the random error.
4 RESULTS
To evaluate the performance of the developed
system, assessment of an InI was carried out.
At the end of test, the results are stored in the
DB. The results can be consulted on text format or
graphically visualized. In text format, statements on
the standard conformance are presented as shown in
Figure 4.
For ethical reason, since the developed system
was not certificated by an accredited laboratory,
reference to the model and manufacturer of the InI
as well as the health institution to which it belongs
are omitted.
In the graph, the behaviour of the measured
parameters (Incubator Temperature, RH and the air
velocity) during the whole test can be observed.
Figure 4: Incubator temperature measured during the
assessment. Relative humidity and air flow curves are also
presented in the graphical report.
MICROCOMPUTERIZED SYSTEM TO ASSESS THE PERFORMANCE OF INFANT INCUBATORS
121

5 CONCLUSION
The developed system does not comprise a sound
level meter. The sound measurements required by
the standard are relatively simple to be made.
Equipments available in the market can be employed
to this end, having a layout that allows their use in
other applications.
The maximum uncertainties in the temperature
measurements (taking into account the propagation
of the reference sensor uncertainty: ±0.3ºC) obtained
with the sensors B, C, D and E are ±0.4 ºC, ±0.5 ºC,
±0.5 ºC and ± 0.61 ºC, respectively.
Assessment of InIs according to the NBR IEC
601-2-19 standard can be performed by the
developed system in a semi-automatic manner, since
the user has to change the InI operating settings
during the test. Throughout the procedure, the
control software beeps at the end of each stage and
shows messages on the PC screen. These messages
ask the operator to adjust the InI settings before
performing the next set of measurements.
The control software and the used
communication protocol have proven to be suitable
and robust during the InI assessment.
The report generated at the end of the assessment
points out the InI conformance with respect to the
NBR IEC 601-2-19 requirements.
It shall be noted that all the tests carried out with
an InI are stored in a same DB, allowing a better
follow-up of its performance along its lifetime.
For instance, the number of corrective
maintenance underwent by a given InI model can be
very useful to the managers when considering the
purchasing of new equipments.
ACKNOWLEDGEMENTS
The authors thank the CNPq for financial support
(507363/2004-3).
REFERENCES
Ministério da Saúde, 2002. Manual do curso de
humanização do recém – nascido de baixo peso.
Método Canguru - Série A: Normas e Manuais
Técnicos Nº 145. Brasília. 1
nd
edition.
Gonzaléz, L. H., 2001. Termoregulación em Recién
Nacido. In: Servicio Neonatalogia. Servicio
Neonatalogia. Hospital Clinico Universidad de Chile.
Chile. 1
nd
edition. p. 34-40.
NBR IEC 601-2-19, 1999. Equipamento Eletromédico
Parte2: prescrições particulares para segurança de
incubadoras para recém-nascidos RN, Rio de Janeiro.
ABNT.
Matthew N, Stones R., 2005. Beninning databases with
PostgreSQL. Aprress. 2
nd
edition.
Figure 5: Part of the report generated by the developed system.
BIODEVICES 2008 - International Conference on Biomedical Electronics and Devices
122
