
LAB-ON-A-CHIP WITH FLUID ACOUSTIC MICROAGITATION
Piezoelectric Polymer β-PVDF used as Ultrassonic Transducer
V. F. Cardoso, J. G. Rocha, F. O. Soares, G. Minas
Dept. Industrial Electronics,University of Minho, Campus de Azurem, 4800-058 Guimaraes, Portugal
S. Lanceros-Mendez
Dept. Physics,University of Minho, Campus de Gualtar, 4710-057 Braga, Portugal
Keywords: Lab-on-a-chip, PVDF, acoustic microagitation, SU-8.
Abstract: The main objective of this article is to describe the development of a fully-integrated disposable lab-on-a-
chip for point of care testing and monitoring of biochemical parameters in biological fluids. The lab-on-a-
chip is composed mainly by two dies: the fluid and the detection. The fluid die, fabricated in SU-8,
comprises three microfluidic cuvettes, containing the fluids into analysis, and a β-PVDF microagitation
system. The detection die is fabricated in a CMOS standard process and contains the photodetectors and the
electronics for signal actuation and detection. The main innovation of this lab-on-a-chip is the application of
an acoustic microagitation technique by the deposition of the β-PVDF piezoelectric polymer underneath the
microfluidic structures, with automatic electronic control. This piezoelectric polymer produces mechanical
vibrations, which allow the enhancement of the mixing and the reaction.
1 INTRODUCTION
Microfluidic technology has become an important
tool for analytical biochemistry applications. It
enables the fabrication of precise and small
structures in glass, quartz, silicon or polymeric
materials: the lab-on-a-chip concept. The great
interest in that technology stems from the inherent
performance gains: reduced sample size, higher
degree of integration and thus enhanced potential for
automation and control fluids of submicroliter
volumes, shortened response time, potential for
improved analytical performance, reduced storage of
chemicals and hence laboratory safety, and reduced
costs (Kopp et. al, 1997).
There is a large demand in the healthcare system
to develop lab-on-a-chips for rapid and reliable point
of care (POC) testing and monitoring. Such lab-on-
a-chips would significantly enhance the quality of a
diagnostic by offering immediate measurement of
several clinically relevant parameters that can be
used to assess the health of the patient. The
biochemical analysis of the patient’s biological
fluids is a good start. Most diseases leave a
molecular fingerprint in those fluids and by
measuring that fingerprint in the right way, the
precision of the diagnostic can be improved
(Connolly, 1995). However, the physician or the
patient has no routine in performing the advanced
biochemical analysis on-chip. Therefore, to develop
point of care disposable lab-on-a-chips, avoiding
cross-contamination of samples and measurements
errors, it is necessary to have a reliable and highly
automated microfluidic control system. This system
should be fully integrated with the control and the
detection electronics implemented on a low-cost
substrate and performed by a low-cost fabrication
process. The use of MEMS (Micro Electro
Mechanical Systems) based devices, such as
microvalves and micropumps, increases the cost of
the system, needs complex control systems and are
difficult to integrate (Reyes et. al, 2002). Mixing
only by diffusion avoids these drawbacks. However,
long transit times and consequently long
microchannels are necessary, if large molecules with
small diffusivities must react. This illustrates how
dramatic the diffusion limitation is.
To overcome the long transit times due to
diffusion and to enable high-efficient reactions it is
262
F. Cardoso V., G. Rocha J., O. Soares F., Minas G. and Lanceros-Mendez S. (2008).
LAB-ON-A-CHIP WITH FLUID ACOUSTIC MICROAGITATION - Piezoelectric Polymer β-PVDF used as Ultrassonic Transducer.
In Proceedings of the First International Conference on Biomedical Electronics and Devices, pages 262-267
DOI: 10.5220/0001050202620267
Copyright
c
SciTePress

necessary to induce the microfluidic die by a
mechanism that accelerates the mixing and the
reaction, preferably with no moving parts. In this
paper, such mechanism is presented.
2 BACKGROUND
The need for rapid and in-situ measurements with
low sample volumes has led to the development of
miniaturized analyses devices with the fluidic,
detection and readout systems integrated in a single
chip. The vision of those devices was presented in
1990 (Manz et. al., 1990) and has been since then
the inspiration for an intense research effort pursuing
miniaturization of macroscopic biochemical analy-
ses methods. Within the field of clinical diagnostics,
several lab-on-a-chip approaches have been
published (Auroux et. al, 2002, Reyes et. al., 2002).
Our group has developed a lab-on-a-chip for
measuring the concentration of some biomolecules
in urine samples by optical absorption technique
(Minas, et. al., 2005). It comprises three parts in a
multi-chip-module: (1) a microfluidic system die
containing the microchannels fabricated using SU-8
techniques (Ribeiro et. al., 2005); (2) an optical
filtering system based on highly selective Fabry-
Perot optical resonators using a stack of CMOS
process compatible thin-film layers (Minas et. al.,
2006); (3) a detection and readout system fabricated
in a CMOS microelectronic process (Minas et. al.,
2004). The optical filtering system enables the
measurement using white light illumination, thus
avoiding the use of a wavelength dependent light
source. This characteristic makes the lab-on-a-chip
portable and ensures that the analysis can be
performed at any location with instantaneous results.
However, in that lab-on-a-chip, mixing the samples
with the reagents was performed by diffusion, which
leads to long transit times, especially when large
molecules with small diffusion coefficients must be
analysed. Therefore, to be valuable for point of care
testing and monitoring, the microfluidic die of the
lab-on-a-chip requires a microagitation mechanism.
It is desirable that this mechanism does not require
any external apparatus, internal moving parts or
valves.
The flows used in microfluidic systems are very
small, of the order of 100 μm, with velocities of the
order of 1 mm/s, which leads to Reynolds number
less than 1. For this small Reynolds numbers the
flow is dominated by the viscous effects and
turbulent mixing is impossible. Therefore, mixing in
a straight channel rely on diffusion, but since
diffusion coefficients of some biological species
(e.g. enzymes and other proteins) are very small,
mixing may take several minutes or even hours. This
may be undesirable for some applications and
alternative mixing methods must be developed.
Acoustic waves are an interesting solution for
this problem. They have been used both to promote
mixing (Bengtsson et. al., 2004) and to pump fluids
(Rife et. al., 2000). Sound waves that propagate in
the fluid generate pressure differences that induce
the so called acoustic streaming. Secondary flows
can be created in the plane perpendicular to the main
flow in the channel. These secondary flows promote
mixing by convection between otherwise parallel
currents. Therefore, acoustic microagitation could be
a simple actuation source for mixing and promoting
fluids reaction.
In order to produce the ultrasounds, the channels
and the reaction chambers of the microfluidic die
must have an ultrasound transducer, which can be
fabricated from a piezoelectric polymer, such as the
PVDF - Poly(Vinylidene Fluoride) in its beta-phase.
PVDF is a polymer with interesting piezoelectric
properties, which allows important electro-optical,
electromechanical and biomedical applications. This
polymer shows at least four crystalline phases. The
one with the best piezo- and pyroelectric properties,
after poling, is the beta-phase (Sessler, 1987). Until
recently, this phase was exclusively obtained by
mechanical stretching of films originally in the non-
polar alfa-phase, which is the most stable one from a
thermodynamic point of view and directly obtained
from the melt. This process results in films mostly in
the beta-phase, but with a small percentage of alfa-
phase material (Sencadas et. al., 2004). Unoriented
films exclusively in the beta-phase were obtained
from the crystallization of PVDF from solution with
N,N-Dimethyl Formamide or Dimethyl Acetamide
at temperatures below 70ºC. The electromechanical
properties of this film were improved by a patented
process by our group (Lanceros-Mendez et. al.,
2006).
3 DEVICE DESCRIPTION
This paper describes the concept, operation and
implementation of a portable, low-cost, plastic
based, disposable and highly automated lab-on-a-
chip for biochemical analyses of biological fluids. It
uses optical absorption spectrophotometry as
measurement analytical technique and acoustic
microagitation for mixing the fluids. Its application
is for point-of-care systems.
LAB-ON-A-CHIP WITH FLUID ACOUSTIC MICROAGITATION - Piezoelectric Polymer ß-PVDF used as Ultrassonic
Transducer
263

3.1 Measurement Analytical Technique
The spectrophotometric analysis, the study of the
interaction of electromagnetic radiation with
(bio)chemical compounds, is a very convenient and
often used analytical technique in clinical
laboratories for routine tests analyses, especially the
ones based on colorimetric detection (Sigma, 2006).
However, many of the analytes of interest for
clinical analysis do not have chromophores that
absorb light in a useful part of the visible range.
Specific chemical reagents are available to transform
these analytes into colored products that do have
adequate absorbance. Therefore, in that detection
method, the sample (ex: urine) is mixed with a
proper reagent for the biochemical parameter in
analysis (ex: uric acid reagent) and from that
mixture, a visible colour is produced. The intensity
of that colour is proportional to the biochemical
parameter concentration and can be quantified by
measuring the optical absorption of the mixture,
once the mixture has an absorption maximum at a
specific wavelength. As an example, for determining
the uric acid concentration in a urine sample, the
mixture has an absorption maximum at 495 nm
(Figure 1). For an accurate measurement a complete
and homogeneous mixing must be achieved.
495
Uric
acid
Rise of
biochemical
parameter
concentration
Magnesium
Wavelength (nm)
Absobance (arb. units)
Calcium
550
650
600
Albumin
Figure 1: Absorption spectra for some biochemical
parameters with different concentrations in urine.
3.2 Lab-on-a-chip Operation
The lab-on-a-chip is composed by two dies: the
fluidic die and the detection die (Figure 2). For the
measurement of the concentration of the
biochemical parameters, a light beam is directed to
the three fluidic cuvettes. The intensity of the light
beam transmitted through the mixture is measured
using the three photodetectors, placed underneath
the microfluidic cuvettes, forming three optical
channels. The photodetectors signal is converted, by
the readout electronics, into a digital signal that
allows simpler computer interfacing. The accurate
sample and reagent mixing is performed by the
control electronics that actuates an electroactive
polymer (β-PVDF), deposited underneath the
microfluidic cuvettes. This polymer vibrates and
produces the proper acoustic microagitation of the
mixture, e. g., the produced waves are coupled to the
slide and propagate into the microfluidic cuvette.
Chip
Microfluidic cuvettes
β−
PVDF
Photodetectors
Electronics
Figure 2: Schematic drawing of the lab-on-a-chip
structure.
3.3 Microfluidic Die
The microfluidic die includes the microfluidic
cuvettes containing the fluids to be analysed
(Figure 2). Three cuvettes are needed for each
analysis: one for the chemical reagent, in order to
obtain the baseline reference; other for the mixture
of the sample plus the reagent, to perform the
analysis of the coloured mixed solution; and a third
one with a standard sample with a well-known
concentration of the biochemical parameter that is
being analyzed, for the calibration of the
biochemical parameter concentration.
Under the microfluidic cuvettes a piezoelectric
polymer, β-PVDF, is deposited (Figure 2). This
material will be responsible for the acoustic
microagitation of the fluid. β-PVDF is a transparent
piezoelectric polymer that will produce the
necessary vibration, when an electrical alternating
voltage is applied to its contacts. These vibrations
result in the acoustic microagitation necessary to
mix the sample with the reagent inside the cuvettes.
Moreover, it accelerates the required time for the
complete and homogeneous mixing, improving the
efficiency of the reaction.
The microfluidic cuvettes are fabricated using a
photoplastic material: the photoresist SU-8. This
photoresist is an epoxy-based material that offers
relevant properties, such as high mechanical
strength, good adhesion on many different substrate
materials and biocompatibility. The SU-8 based
fabrication is a low-cost process, UV (from 350 nm
to 400 nm) lithography semiconductor compatible
and does not require expensive masks. It can be
processed by using a spincoating and an UV
maskaligner. In addition, the microfluidic system
can be a disposable die, which minimize the costs
associated with cleaning of the microfluidic cuvettes
and avoids the contamination between analyses.
BIODEVICES 2008 - International Conference on Biomedical Electronics and Devices
264
Moreover, SU-8 based processing enables the
fabrication of deep structures with very low sidewall
roughness which is suitable for optical absorption
measurements (IBM, 1989).
The negative mask to be used for patterning the
structure of the microfluidic cuvettes is fabricated
from a regular transparency foil, such as the ones
used in printed circuit boards. The chosen SU-8
photoresist is the SU-8-2150, which has a high
viscosity and is the most appropriate for the required
depth (≥ 500 μm). The following paragraphs
describe the microfluidic cuvettes processing steps,
including the SU-8 and the β-PVDF depositions.
After cleaning and drying the glass substrate, a
thin-film of Itrium-Tin-Oxide (ITO) of approxima-
tely 40 nm is deposited by PVD, in order to form the
bottom contact of the piezoelectric β-PVDF. Then, a
600 nm β-PVDF layer is spun of on top of the ITO.
The coated β-PVDF is activated by applying an
electrical field of tens of megavolts by meter, which
is known as the electrical poling of the polymeric
material. After that, the top electrical contact of the
β-PVDF is deposited using the same procedure used
for the bottom contact. These steps complete the
fabrication of the microagitation mechanism.
Next, the fabrication of the microfluidic cuvettes
starts with the spin of 1.25 ml/cm
2
of SU-8 on top of
the β-PVDF top ITO contact. This process requires
two steps: first, ramping up the spin rotation to
500 rpm during 5 s and keeping that velocity for
10 s; second, ramping up again the spin rotation up
to 1000 rpm during 3 s and keeping it for 30 s. Then,
a soft bake process is needed for evaporation of the
solvent and for the hardening of the SU-8. The soft
bake is performed at 65 ºC during 420 s and at 95 ºC
during 3600 s. The initial temperature allows a
slower evaporation, which results in a better coating
and, mainly and very important, without grooves.
Then, the cuvettes are exposed to UV light, using a
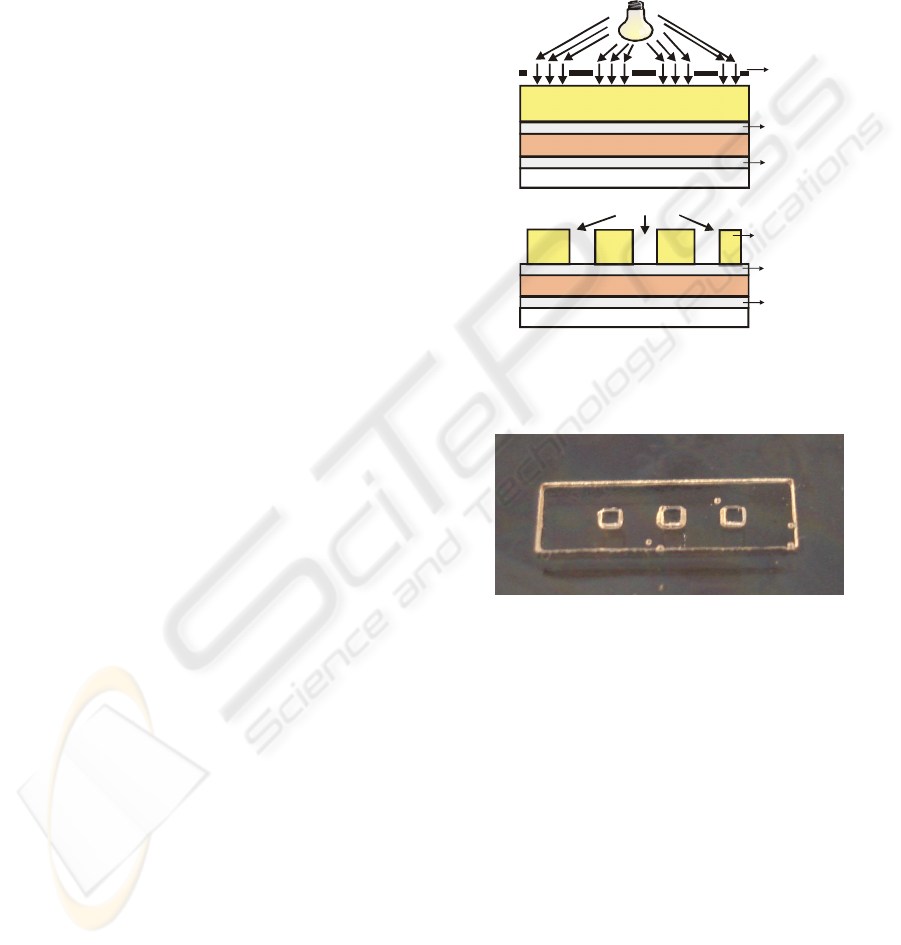
negative mask, for patterning the structure. Figure 3a
shows a cross-section of the device after the
aforementioned steps.
An excessive dose of light with wavelength
lower than 350 nm results in an excessive absorption
at the surface of the film. The effect can be a
pyramidal shape of the microfluidic cuvettes. To
avoid this effect, a commercially available glass
optical filter was used to cut of the wavelengths
lower than 350 nm. The used exposition energy was
700 mJ/cm
2
. The post-exposure bake is performed at
65 ºC during 60 s, followed by 900 s treatment at
95 ºC. The fabrication ends with the development,
during 2400 s, with the SU-8 developer: an agitation
bath of 1 methoxy-2-propanol acetate. In this way,
the unexposed resist is dissolved and the
microfluidic cuvettes are formed. The structure is
then cleaned with isopropanol and deionised water
and dried with nitrogen. Figure 3b shows a cross
section of the patterned structure. The structure of
the microfluidic cuvettes is shown in Figure 4. Each
cuvette is 1 mm wide, long and height, having a 1 μl
sample volume.
(a)
Glass substrate
ITO
ITO
β
-PVDF
SU-8 photoresist
Mask
β
-PVDF top
contact
β
-PVDF bottom
contact
(b)
Glass substrate
ITO
ITO
β
-PVDF
SU-8
Cuvettes
β
-PVDF top
contact
β
-PVDF bottom
contact
SU-8
SU-8
Patterned SU-8
after development
Figure 3: Fabrication sequence of the microfluidic
cuvettes of the prototype: (a) deposition, spin coating, soft
bake, UV exposure; (b) development of the SU-8.
Figure 4: Picture of the microfluidic cuvettes.
3.4 Detection Die
The detection die includes the detectors and the
electronics for signal actuation and detection, all
fabricated in CMOS technology. Specifically, it
comprises the photodetectors, its readout electronics
and the electronics that control the voltages and
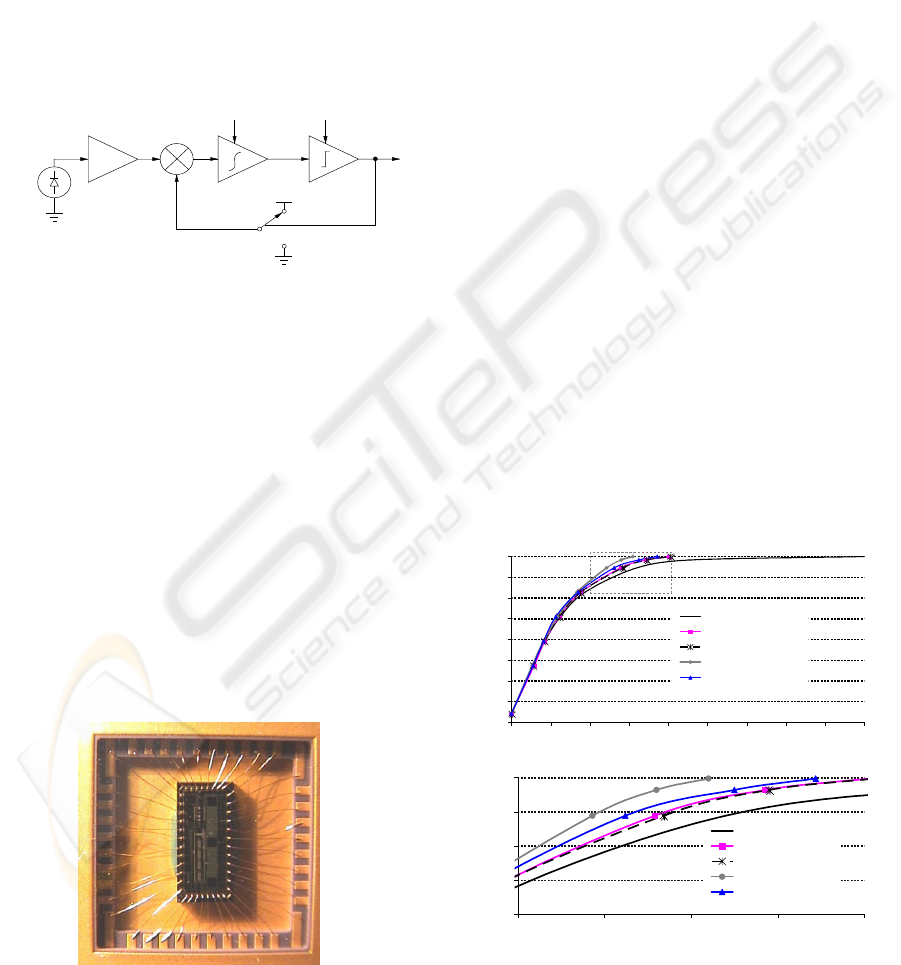
frequencies applied to the β-PVDF. Figure 5 shows a
block diagram of the photodiode and its readout
electronics.
The readout electronics consists basically in a
current amplifier and in a sigma-delta analog to
digital converter. The circuit is repeated for each
microfluidic cuvette. An additional circuit for the
photodiode dark current compensation is also
implemented.
LAB-ON-A-CHIP WITH FLUID ACOUSTIC MICROAGITATION - Piezoelectric Polymer ß-PVDF used as Ultrassonic
Transducer
265
After the light reaches the photodiodes, the four
analog to digital converters start the conversion and
their output signal is placed in four separated lines.
Further computer processing perform additional
calculations of these four output signals to achieve
the concentrations of the biochemical parameters
into analysis. The oversample frequency of the
sigma-delta converters is determined by the desired
number of output bits (signal to noise ratio). For this
particular application, an oversampling ratio of 64
and a first order one-bit sigma-delta analog to digital
converter is adequate since the input signal has no
time variations, allowing a high oversample ratio
without the need for a high clock frequency.
+
Vdd
_
Reset Clk
Out
Amplifier
Current
Photodiode
Figure 5: Block diagram of the readout electronics for
each microfluidic cuvette.
For these sigma-delta converters, the gain of the
integrator is very high, so, it is finite and larger than
the oversampling ratio. At these conditions, the
noise in the signal band increases only 0.15 dB
(Candy et. al., 1992). Finally, the integrator must be
initialized with a known voltage level at the
beginning of each conversion. This initialization
allows an improvement in the sigma-delta modulator
of 3 dB in the signal to noise ratio (Netravali, 1977).
It was used a digital filter with constant weights
(simple average) and the Pearson coefficient
obtained for the response curve is larger than 0.99,
which demonstrates a good linearity of the device.
This result can even be improved by increasing the
oversampling ratio and by using an optimized digital
filter. Figure 6 shows a picture of the detection die.
Figure 6: Picture of the detection die.
4 EXPERIMENTAL RESULTS
The evaluation of the mixing process was carried out
using the Sigma Diagnostic kit (Infinity
TM
Uric Acid
Reagent) and standards of urine with 30 mg/dl of
uric acid concentration (Sigma, 2006). The reagent
reacts with a sample of urine containing uric acid in
a 50:1 ratio, and produces a pink colour with an
absorption maximum at 495 nm. Its manual
procedure states that a gentle inversion of the
cuvette that contains the mixture is enough for the
complete and homogeneous mixing. Performing this
manual agitation, and after approximately 5 minutes
at room temperature, the mixing is complete.
Without any agitation and due to the high diffusion
coefficients of uric acid in the Infinity
TM
reagent, the
complete mixing is achieved but it takes
approximately 15 minutes at room temperature. In
clinical laboratories, the macroscopic equipments
have mechanical agitation of the cuvettes for
improving mixing and reducing the reaction time.
The experimental results for evaluating the
mixing process are presented in Figure 7. The
microagitation was performed by powering the
β-PVDF though the electrical contacts, with a
sinusoidal signal of 5 V amplitude at the frequencies
shown in the Figure 7. The system was calibrated for
an absorbance of 0 a. u. filling the cuvette with
deionised water, and for an absorbance of 1 a. u.
filling the cuvette with the perfectly mixed solution
(perfectly mixing was guaranteed by external
agitation of the mixture using a slow-rotation
vortex).
0.2
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1.0
0 100 200 300 400 500 600 700 800 900
Time (s)
Absorbance (a.u.)
Without oscilation
Oscilation - 100 Hz
Oscilation - 300 Hz
Oscilation - 1 kHz
Oscilation - 10 kHz
(a)
0.80
0.85
0.90
0.95
1.00
200 250 300 350 400
Time (s)
Absorbance (a.u.)
Without oscilation
Oscilation - 100 Hz
Oscilation - 300 Hz
Oscilation - 1 kHz
Oscilation - 10 kHz
(b)
Figure 7: (a) Measured absorbance at 495 nm for 30 mg/dl
of uric acid concentration in urine as a function of time for
different driving frequencies. (b) Zoom of dashed-square.
BIODEVICES 2008 - International Conference on Biomedical Electronics and Devices
266

The analysis cuvette is filled with the reagent. Its
measured absorbance is 0.24 a. u.. Afterwards, the
urine standard with 30 mg/dl of uric acid is
dispensed on the reagent. The absorbance increases
as the sample is being mixed with the reagent. From
Figure 7 it can be seen that with the application of
oscillations, the mixing occurs in a faster way, being
the time necessary to obtain the complete mixing
(absorbance of 1 a. u.) at 1 kHz only one third
(300 s) of the complete mixing time without
oscillation (900 s).
From those results it can be concluded that the
application of acoustic microagitation by the
β-PVDF piezoelectric polymer improves the mixing
time. Moreover, uric acid has high diffusion
coefficients, which is a good characteristic for
mixing by diffusion only: after 15 minutes the
mixture will be completed, even without agitation.
However, when other biochemical parameters
present in biological fluids have to be analysed, such
as enzymes and some macromolecules, the mixing
by diffusion can take hours or can even not occur.
An example is a DNA analysis when PCR
(Polymerase Chain Reaction) must be performed.
5 CONCLUSIONS
A lab-on-a-chip device with fluidic acoustic
microagitation that reduces the mixing time of the
analytes with the reagents was reported. The device
consists of two dies: a microfluidic die, composed
by three cuvettes and a β-PVDF acoustic
microagitator, fabricated on a glass substrate; and an
electronic detection die, composed by the readout
circuits and the microagitation control electronics.
The main innovative concept is the application of a
β-PVDF film that produces acoustic microagitation,
increasing the mixing velocity. Experimental results
show that at 1 kHz microagitation, the mixing time
is reduced to one third of the time needed without
microagitation. As a conclusion, it can be stated that,
for decreasing device sizes, acoustic microagitation
becomes a preferred technology for effective
mixing.
ACKNOWLEDGEMENTS
Work supported by the Portuguese Science
Foundation (grants PTDC/BIO/70017/2006 and
POCI/CTM/59425/2004).
REFERENCES
Kopp, M. U., Crabtree, H. J. Manz, A., 1997,
Developments in technology and applications of
microsystems, Current Opinion in Chemical Biology,
1, p. 410-419.
Connolly, P., 1995, Clinical diagnostics opportunities for
biosensors and bioelectronics, Biosensors &
Bioelectronics, 10, p. 1-6.
Reyes, D. R., Iossifidis, D., Auroux, P., Manz, A., 2002,
Micro Total Analysis Systems. 1.Introduction, Theory,
and Technology, Anal. Chem., 74, p. 2623-2636.
Manz, A., Graber, N., Widmer, H. M., 1990, Miniaturized
total chemical systems: a novel concept for chemical
sensing. Sensors and Actuators B, 1, p. 244-248.
Auroux, P., Iossifidis, D., Reyes, D. R., Manz, A., 2002,
Micro Total Analysis Systems. 2.Analytical Standard
Operations and Applications, Anal. Chem., 74,
p. 2637-2652.
Minas, G., Wolffenbuttel, R. F., Correia, J. H., 2005, A
Lab-on-a-Chip for Spectrophotometric Analysis of
Biological Fluids, Lab-on-a-Chip, 5, p. 1303-1309.
Ribeiro, J. C., Minas, G., Turmezei, P., Wolffenbuttel, R.
F., Correia, J. H., 2005, A SU-8 Fluidic Microsystem
for Biological Fluids Analysis. Sensors and Actuators
A, 123-124 p. 77-81.
Minas, G., Wolffenbuttel, R. F., Correia, J. H., 2006, An
array of highly selective Fabry-Perot optical-channels
for biological fluids analysis by optical absorption
using white light source for illumination. Journal of
Optics A: pure and applied optics, 8, p. 272-278.
Minas, G., Martins, J. S., Ribeiro, J. C., Wolffenbuttel, R.
F., Correia, J. H., 2004, Biological microsystem for
measuring uric acid in biological fluids, Sensors and
Actuators A, 110, p. 33-38.
Bengtsson, M., Laurell, T., 2004, Ultrasonic agitation in
microchannels, Anal Bioanal Chem, 378-7, p. 1716-
1721.
Rife, J.C. et. al., 2000, Miniature valveless ultrasonic
pumps and mixers, Sensors and Actuators B, 86,
p. 135–140.
Sessler, G.M, 1987, Topics in Applied Physics: Electrets,
2nd Ed., Springer, Berlin Heidelberg New York.
Sencadas, V., Lanceros-Mendez, S., Mano, J.F., 2004,
Thermochimica Acta, 424, p. 201.
Lanceros-Mendez, S., Sencadas, V., Gregorio Filho, R.,
2006, Portuguese patent n.º 103318.
Biochemistry and Organic Reagents: for bioscience
investigation. Sigma-Aldrich Diagnostics®, 2006.
IBM, Photoresist composition and printed circuit boards
and packages made therewith. J. D. Gelorme, R. J.
Cox, S. A. R. Gutierrez, US Patent 4882245, 1989.
Candy, J. C., Temes G. C:, 1992, editors, Oversampled
Delta-Sigma Data Converters, IEEE Press, New York.
Netravali, A. N., 1997, Optimum digital filters for
interpolative A/D converters, Bell Syst. Tech. J., 56,
p. 1629.
LAB-ON-A-CHIP WITH FLUID ACOUSTIC MICROAGITATION - Piezoelectric Polymer ß-PVDF used as Ultrassonic
Transducer
267
