
ARTIFICIAL NEURAL NETWORKS FOR DIAGNOSES OF
DYSFUNCTIONS IN UROLOGY
1
David Gil Mendez,
2
Magnus Johnsson,
1
Antonio Soriano Paya and
1
Daniel Ruiz Fernandez
1
Computing Technology and Data Processing, University of Alicante, Spain
2
Lund University Cognitive Science & Dept. of Computer Science, Lund University, Lund, Sweden
Keywords:
Artificial neural networks, urology, artificial intelligence, medical diagnosis, decision support systems.
Abstract:
In this article we evaluate the work out of artificial neural networks as tools for helping and support in the
medical diagnosis. In particular we compare the usability of one supervised and two unsupervised neural
network architectures for medical diagnoses of lower urinary tract dysfunctions. The purpose is to develop a
system that aid urologists in obtaining diagnoses, which will yield improved diagnostic accuracy and lower
medical treatment costs. The clinical study has been carried out using the medical registers of patients with
dysfunctions in the lower urinary tract. The current system is able to distinguish and classify dysfunctions as
areflexia, hyperreflexia, obstruction of the lower urinary tract and patients free from dysfunction.
1 INTRODUCTION
Nowadays, the urologists have different tools avail-
able to obtain urodynamical data. However, it still re-
mains very complicated to make a correct diagnosis:
the knowledge concerning the origin of the detected
dysfunctions depends mainly on acquired experience
and on the research, which is constantly carried out
within the field of urology. The specialists in urology
are quite often dealing with situations that are poorly
described or that are not described in the medical lit-
erature. In addition there are numerous dysfunctions
whose precise diagnoses are complicated. This is a
consequence of the interaction with the neurological
system and the limited knowledge available on how
this operates.
Various techniques are used to diagnose dysfunc-
tions of the lower urinary tract (LUT), which en-
tail different degrees of invasiveness for the patient
(Abrams, 2005). A urological study of a patient con-
sists of carrying out various costly neurological as
well as physical tests like flowmetry and cystometry
examinations with high degrees of complexity and in-
vasiveness. This project is intended to aid the special-
ist in obtaining a reliable diagnosis with the small-
est possible number of tests. To this end we propose
the use of artificial neural networks (ANNs) since
these present good results for classification problems
(Begg, 2006). The reason why we decided to ap-
ply ANNs instead of other artificial intelligence (AI)
methods for the support of medical diagnosis is due
to the fact that ANNs can be trained with appropri-
ate data learning in order to improve their knowledge
of the system. In comparison to other techniques or
other (more classical) approaches such as rules based
systems or probabilities, ANNs are more suitable for
medical diagnosis. On one hand, rules based systems
(MYCIN system (Mycyn, 1976)) contain ”if-then”
rules where the ”if” side of any rule is a collection
of one or more conditions connected by logical oper-
ators such as ”AND”, ”OR” and ”NOT”. On the other
hand, other systems such as probability systems cal-
culate measures of confidence without the theoretical
underpinnings of probability theory. These formal ap-
proaches based on probability theories are precise but
can be awkward and non-intuitive to use.
Therefore, in medical diagnosis, we use the advan-
tages of ANNs, which are considered as a method
of disease classification. This classification has two
divergent meanings. We can have a set of registers,
vectors or data with the aim of establishing the exis-
tence of classes or clusters. In contrast, we can know
with certainty that there exist some classes, and the
aim is to establish a rule able to classify a new regis-
ter into one of these classes. The first type is known
as Supervised Learning and the second one is known
as Unsupervised Learning (or Clustering). We believe
that the accuracy of the diagnosis in medicine and, in
particular, in urology will be improved by using these
types of architectures (one supervised and two unsu-
191
Gil Mendez D., Johnsson M., Soriano Paya A. and Ruiz Fernandez D. (2008).
ARTIFICIAL NEURAL NETWORKS FOR DIAGNOSES OF DYSFUNCTIONS IN UROLOGY.
In Proceedings of the First International Conference on Health Informatics, pages 191-196
Copyright
c
SciTePress
pervised).
With the system of aid to the diagnosis major ben-
efits are obtained both for the patient, by avoiding
painful tests, and for the medical centres by avoid-
ing expensive urodynamical tests and reducing wait-
ing lists.
Although the use of ANNs in medicine is a rather
recent phenomenon, there are many applications de-
ployed as in the field of diagnosis, imaging, pharma-
cology, pathology and of course prognosis. ANNs
have been used in the diagnosis of appendicitis, back
pain, dementia, myocardial infraction (Green, 2006),
psychiatric disorders (Peled, 2005)(Politi, 1999),
acute pulmonary embolism (Suzuki, 2005), and tem-
poral arteries.
In Urology,prostate cancer serves as a good exam-
ple of the usability of ANNs (Remzi, 2001)(Batuello,
2001)(Remzi, 2004). However our work is more re-
lated with the neurological part which is less explored
(Gil, 2006)(Gil, 2005)(Ruiz, 2005).
In this paper we describe the implementations of
ANN based systems aiming at support diagnoses of
dysfunctions of the LUT. The remaining part of the
paper is organized as follows: first, we give a brief
description of the employed neural network architec-
tures. Next we describe the design of our proposal
and the training of the ANNs by the available data.
Then we describe the subsequent testing carried out
in order to analyze the results. Finally we draw the
relevant conclusions.
2 NEURAL NETWORK
ARCHITECTURES
We have tested three different neural network archi-
tectures, two unsupervised ANNs and one supervised
ANN. The goal is to obtain a system that supports the
diagnoses of the dysfunctions of the LUT. The clas-
sification in the maps of the unsupervised ANNs and
the output from the supervised ANN will assist the
urologist in their medical decisions.
2.1 Supervised ANN - Multilayer
Perceptron
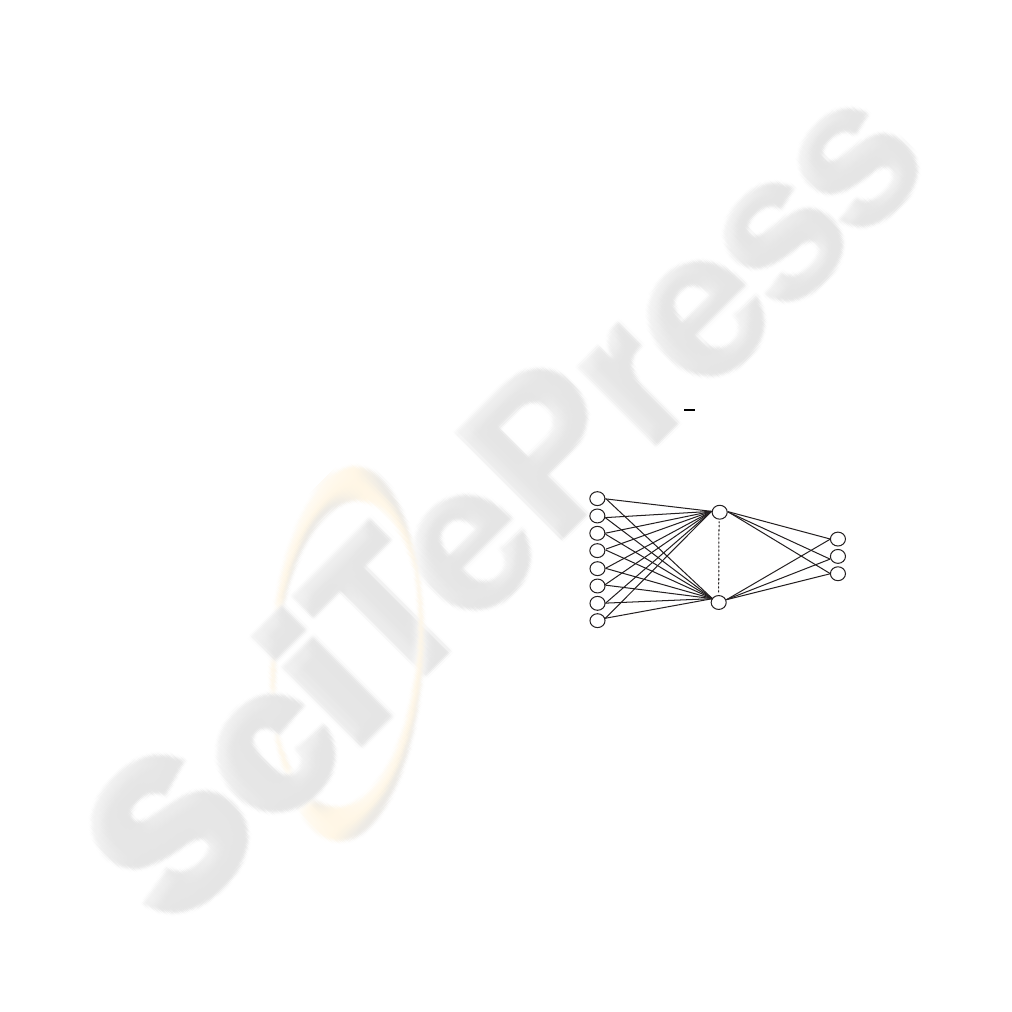
A typical Multilayer perceptron (MLP) network con-
sists of three or more layers of neurons: an input layer
that receives external inputs, one or more hidden lay-
ers, and an output layer which generates the classifi-
cation results (Jiang, 2006)(Fig. 2). Note that unlike
other layers, no computation is involved in the input
layer. The principle of the network is that when data
are presented at the input layer, the network neurons
run calculations in the consecutive layers until an out-
put value is obtained at each of the output neurons.
This output will indicate the appropriate class for the
input data.
The architecture of the ANN (MLP) consists of
layer 1, with the inputs that correspond to the input
vector, the layer 2, with the hidden layer and the layer
3 which are the outputs (the 3 diagnoses of the LUT)
and the learning used is backpropagation and the al-
gorithm runs as follows:
All the weight vectors m are initialized with small
random values from a pseudorandom sequence gen-
erator. The following steps are repeated until conver-
gence (i.e. when the error E is below a preset value):
Update the weight vectors m
i
by
m(t + 1) = m(t) + ∆m(t) (1)
where
∆m(t) = −h∂E(t)/∂m (2)
Compute the error E(t+1).
where t is the iteration number,m is the weight vector,
and h is the learning rate. The error E can be chosen
as the mean square error function between the actual
output y
j
and the desired output d
j
:
E =
1
2
n
j
∑
j= 1
(d
j
− y
j
)
2
(3)
Input
Hidden
Output
Figure 1: The architecture of the MLP.
2.2 Unsupervised ANN - Kohonen’s
Self-Organizing Maps
Kohonen’s Self-Organizing Map (SOM) is com-
posed of neurons located in a two-dimensional matrix
(Kohonen, 1988)(Kohonen, 1990)(Kohonen, 2000).
There is a weight vector, m
i
= (m
i1 i2
... m
in
) asso-
ciated with every neuron in the SOM, where n is the
dimension of the input vectors. In our case they are
the n fields of each observation of a pattern or a pa-
tient in the register.
The SOM is used as a classifier and is organized
as indicated in figure 1.
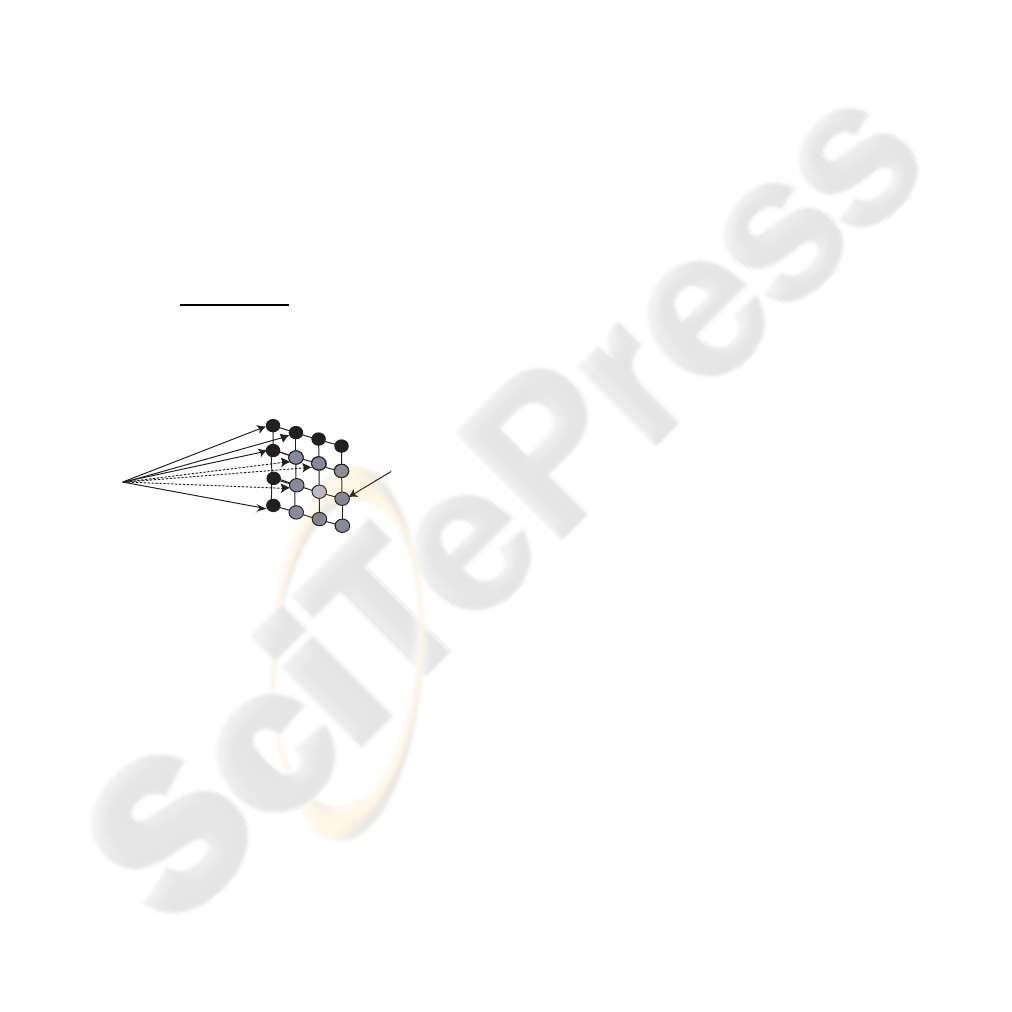
In the fully trained network each neuron is asso-
ciated with a vector in the input space. The SOM is
a soft competitive neural network, which means the
winner neuron, i.e. the neuron with the weight vec-
tor that is closest to the current input vector according
to some measure (dot product in our implementation),
gets its weight vector updated so that it becomes more
similar to the input vector. The neurons in the vicin-
ity of the winner neuron also get their weight vec-
tors updated but to a lesser extent. Usually a Gaus-
sian function of the distance to the winner neuron is
used to modify the updating of the weight vectors.
The trained SOM is a projection of the input space,
which preserves the topological relationships of the
input space. The training of the SOM works as fol-
lows:
At time step t an input signal x ∈ R
n
activates a
winner neuron c for which the following is valid:
∀i : x
T
(t)m
c
(t) ≥ x
T
(t)m
i
(t) (4)
The weights are updated according to:
m
i
(t + 1) =
(
[m
i
(t) +α(t)x(t)]
||m
i
(t) +α(t)x(t)||
if i ∈ N
c
(t)
m
i
(t) if i /∈ N
c
(t)
(5)
where N
c
(t) is the neighbourhood of the neuron c at
time t and 0 < α(t) < ∞.
Input Vector
{x1...xn}
Reference Vector
{mi1...min}
Figure 2: The Kohonen SOM.
2.3 Unsupervised ANN - Growing Cell
Structures
It has been pointed out that the predefined structure
and size of Kohonen’s SOM bring limitations to the
resulting mapping. One attempt to solve this prob-
lem is the growing cell structures (GCS) (Fritzke,
1993)(Fritzke, 1997), which has a flexible and com-
pact structure, a variable number of elements and a k-
dimensional topology where k can be arbitrarily cho-
sen.
In principle, the adaptation of a weight vector in
the GCS is done as described in the previous sec-
tion (SOM), i.e. determine the best matching neu-
ron, adjust its weight vector and the weight vectors of
its topological neighbours. However, there are two
important differences when compared to the SOM,
namely that the adaptation strength is constant over
time, and that only the best matching neuron and its
direct topological neighbours are adapted.
The GCS estimates the probability density func-
tion P(x) of the input space by the aid of local signal
counters that keep track of the relative frequency of
input signals gathered by each neuron. These esti-
mates are used to indicate proper locations to insert
new neurons. The insertion of new neurons by this
method will result in a smoothing out of the relative
frequency between different neurons. The advantages
of this approach is that also the topology of the net-
work will self-organize to fit the input space and the
proper number of neurons for the network will be au-
tomatically determined, i.e. the algorithm stops when
a certain performance criterion is met. Another ad-
vantage is that the parameters are constant over time.
The basic building block and also the initial con-
figuration of the GCS is a k-dimensional simplex.
Such a simplex is for k = 1 a line, for k = 2 a triangle,
and for k = 3 a tetrahedron. In our implementation
k = 2.
The self-organizing process of the GCS works as
follows:
The network is initialized to contain k + 1 neurons
with weight vectors m
i
∈ R
n
randomly chosen. The
neurons are connected so that a k-dimensional sim-
plex is formed.
At time step t an input signal x ∈ R
n
activates a
winner neuron c for which the following is valid:
∀i : ||x − m
c
|| ≥ ||x − m
i
|| , (6)
and the squared distance between the input signal and
the winner neuron c is added to a local error variable
E
c
:
∆E
c
= ||x − m
c
||
2
. (7)
The weight vectors are updated by fractions ε
b
and ε
n
respectively according to:
∆m
c
= ε
b
(x− m
c
) (8)
∀i ∈ N
c
: ∆m
i
= ε
n
(x− m
i
), (9)
where N
c
is the set of direct topological neighbours of
c. A neuron is inserted if the number of input signals
that have been generated so far is an integer multi-
ple of a parameter λ. This is done by dividing the
longest edge between the neuron q with the largest
accumulated error and its connected neighbour f, and
then removing the earlier connection (q, f) and con-
nect r to all neighbours that are common for q and
f. The weight vector for r is interpolated from the
weight vectors for q and f:

m
r
= (m
q
+ m
f
)/2. (10)
The error variables for all neighbours to r are de-
creased by a fraction α that depends on the number
of neighbours of r:
∀i ∈ N
r
: ∆E
i
= (−α/|N
r
|) · E
i
, (11)
The error variable for r is set to the average of its
neighbours:
E
r
= (1/|N
r
|) ·
∑
ι∈Nr
E
i
, (12)
and then the error variables of all neurons are de-
creased:
∀i : ∆E
i
= −βE
i
(13)
3 EXPERIMENTATION
An exhaustive urological exploration with 21 differ-
ent measurements has been carried out with 200 pa-
tients with dysfunctions of the lower urinary tract in
order to create a database. The data has been analyzed
and processed before entered into the network to en-
sure that it is homogenized. These 200 registers con-
tribute to the full knowledge adding different values
to delimit the ranks of each measure. Each of these
registers contains the information measured in the 21
fields showed in Table 1. For this reason, this database
plays a crucial role in order to obtain the knowledge
base of our system.
The table 1 shows the fields (every variable) of the
urological database (their physical units and types of
data). This table helps us to understand the dimension
of the problem to deal with (different types of data,
ranges and incomplete fields). The column direction
of the table indicates the meaning for the ANN: all the
fields are input for the system except the field diagno-
sis, which is the dysfunction of each register. There
are three dysfunction and one output more for the di-
agnosis free of dysfunction.
Our database presents a diversity of ranks, values
and types. For this reason it is better to start with a
process of discretization in order to find a way to guar-
antee the homogeneity as a first step in the process
of training of the ANN. It was adjusted and weighted
with the help of the urologists, following their instruc-
tions and suggestions.
For example, some of the fields of the database are
age, volume of urine and micturition time (as can be
seen in table 1). As the differences among all these
fields are huge, we created ranks in the values of the
Table 1: The fields of the urological database.
Variable Type of data Values Direction
Age Numerical 3 85 years Input
Sex Categorical Male, female Input
Neurological Physical Examination
Perineal and peri-
anal sensitivity
Categorical Partial, absence,
normal, weak
Input
Voluntary control
anal sphincter
Categorical Partial, absence,
normal, weak
Input
Anal tone Categorical Normal, relaxed Input
Free Flowmetry
Volume of urine Numerical 7 682 ml Input
Maximum flow
rate
Numerical 4 58 ml/s Input
Medium flow Numerical 1 43 ml Input
Post void residual Numerical 0 550 ml Input
Micturition time Numerical 13 160 s Input
Cystometry
Bladder storage Numerical 50 461 ml Input
First sensation of
bladder filling
Numerical 50 300 ml Input
Detrusor pressure
during filling
Numerical 2 30 cm H20 Input
Test pressure / flow
Detrusor contrac-
tion
Categorical Invol., Vol.,
Invol.-Vol.
Input
Volume of urine
in micturition
Numerical 0 556 ml Input
Maximum
pressure Detrusor
Numerical 2 200 cm H20 Input
Average flow rate Numerical 0 10 ml/s Input
Abdominal pres-
sure
Categorical Yes, no Input
Post void residual Numerical 0 350 ml Input
Maximum flow
rate
Numerical 0 31ml/s Input
Micturition time Numerical 2 318 s Input
Diagnosis
Areflexia, Hyperreflexia, Obstruction of the LUT, No
Dysfunction
Output
fields dividing them into subclasses in order to adjust
the data input.
For instance:
Age: 0-20 (1), 21-50 (2), 51-65 (3), >65 (4)
Volume of urine: 0-150 (1), 151-300 (2), 301-500
(3), >500 (4)
The numbers between the parentheses are the dis-
cretized representations. As we can observe the diffi-
culties are not only in the data ranks, but also in the
types of data, which complicates the process of data
discretization.
The next step is to run the experimentation.
This has been performed by using cross-validation
method. The data has been divided in five sets (S1,
S2, S3, S4, S5) and the five experiments performed
were:
Experiment 1-Training: T1, T2, T3, T4; Test: V (S5)
Experiment 2-Training: T1, T2, T3, T5; Test: V (S4)
Experiment 3-Training: T1, T2, T4, T5; Test: V (S3)
Experiment 4-Training: T1, T3, T4, T5; Test: V (S2)
Experiment 5-Training: T2, T3, T4, T5; Test: V (S1)
T1
T2
T3
T4
V
T2
T3
T4
V
T1
Experiment 1
Experiment 5
Figure 3: Cross Validation method.
This method is represented in figure 3. There are
four data sets used for the process of constructing the
model (the training data). The other set of data used
to validate the model (the test data). The test data are
chosen randomly from the initial data and the remain-
ing data form the training data. The method is called
5-fold cross validationsince this process has been per-
formed five times.
The results of the Multilayer Perceptron with
the backpropagation algorithm are of around 80%
of accuracy. For the other networks, the unsuper-
vised ones, the results are of 76% for the Grow-
ing Cell Structures and 74% for Kohonen’s Self-
Organizing Map. The MLP offers a slightly better
performance than GCS and SOM. However the unsu-
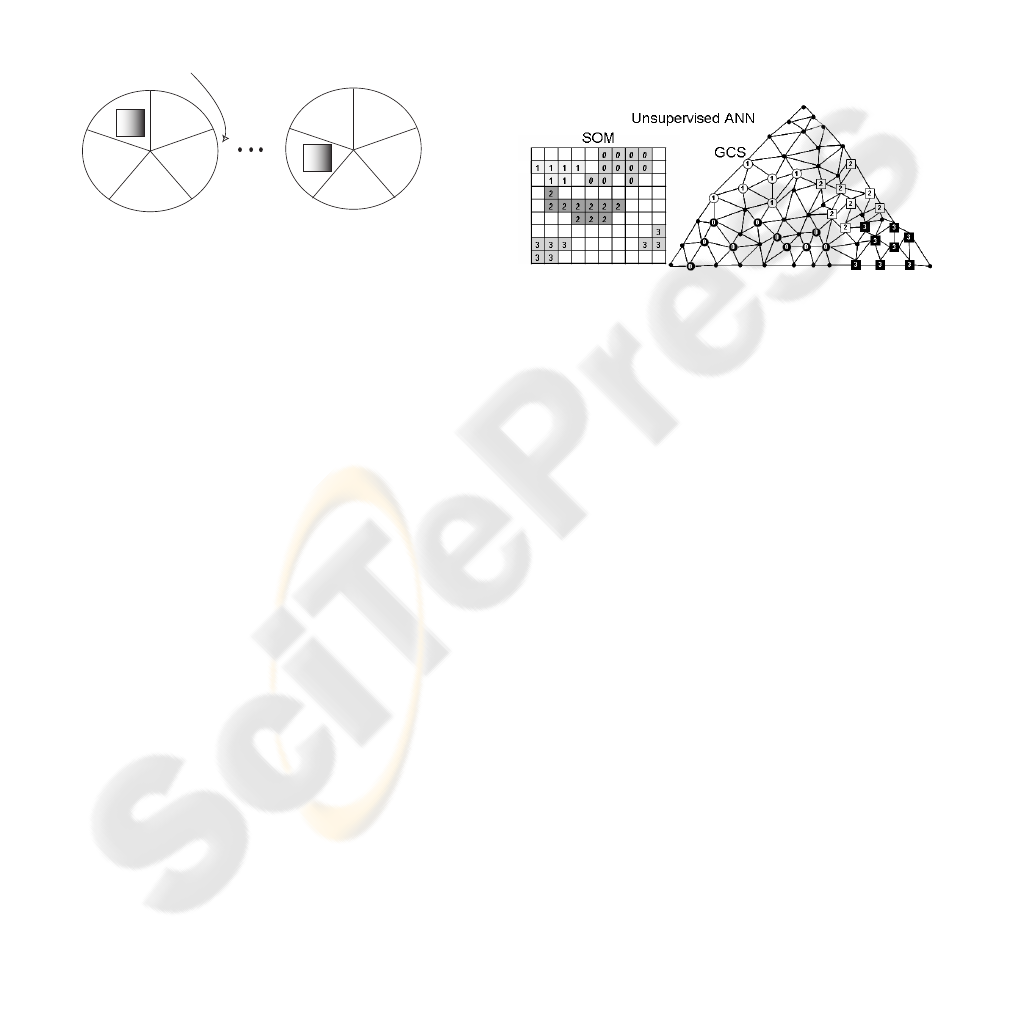
pervised ANNs give a useful visual information. This
is the reason why we believe a hybrid system with
supervised and unsupervised ANNs works properly
incorporating the advantages of each ANN.
This comparativeand its results with the three dif-
ferent types of ANNs not only has the goal of discov-
ering which is the best neural network but also and
mainly to find out relations between the dysfunctions
(output of the network). As shown in figure 4 it will
help the urologist to pay attention in some tests and
its measures to increase the accuracy of the system.
In this regard, it will be possible to eliminate some of
the tests in order to save money, time and sometimes
pain for the patients.
To measure the performance of the ANNs we pro-
ceed as follow: Once the ANNs have been trained
there are some membership functions for each area,
we take a test and we see in which area it is cate-
gorized. If the neuron has only input vectors of the
same dysfunction the accuracy is 100% otherwise it
depends on the mix of dysfunctions and it is another
percentage (it incorporates the fuzzy logic idea since
it measures the degree of membership to the catego-
rized area). Moreover, a big advantage of using unsu-
pervised ANNs , as we can see in figure 4, is the visual
perception of the proximities of the different dysfunc-
tions (their membership functions) categorized in ar-
eas to find out relations between similar inputs. The
system does not produce negative false. This fact
makes it highly reliable amongst the urologists.
Figure 4: Evaluation of the unsupervised ANNs.
4 CONCLUSIONS AND FUTURE
WORK
In this paper we have evaluated the performance of
three different kinds of artificial neural networks,
the Kohonen Self-Organizing Map, the Growing Cell
Structures and the Multilayer Perceptron with the
backpropagation algorithm, when applied to the cat-
egorization of urological dysfunctions. The ANNs
were trained with data from a database with registers
of different patients with urological dysfunctions.
The experiment starts with a stage of discretiza-
tion of the urodynamical measures from a patient in
order to provide them to the ANN and to determine
if there are any of the three dysfunctions of the LUT
or not. In case of finding a dysfunction it would de-
termine what type of dysfunction or dysfunctions the
patient could have.
The human expert is able to generalize by using
his experience. This big advantage is compensated for
ANNs by using graphs offering a visual perception.
The frontiers between two dysfunctions help the urol-
ogist to discover resemblances. They can also help
detecting similarities between fields or urodynamical
samples.
In this work we obtained comments valuable from
the urologists after using the system. They remarked

its advantages to give a more precise diagnosis and,
therefore, to save time and money to the public health.
Their comments encourage us to continue our work to
develop a system that uses the diagnosis obtained as
a result of the combined use of different neural net-
works. This gives an even more accurate diagnosis.
Next step is the use of data mining involving
several steps such as pre-processing with sampling,
cleaning and others learning methods as bayesian net-
works, decision trees, etc.
ACKNOWLEDGEMENTS
We want to express our acknowledgement to
Christian Balkenius for helpful comments on the
manuscript. The data used in the development of this
system is the result of several years of collaboration
with urologists of the Hospital of San Juan (Alicante-
Spain). The work has been supported by the Office of
Science and Technology as part of the research project
“Cooperative diagnosis systems for Urological dys-
functions (2005-2006)”.
REFERENCES
Abrams, P. Urodynamics. 3rd edn., Springer, 2005,20-39.
Begg, R., Kamruzzaman, J., Sarkar R.: Neural Networks in
Healthcare: Potential and Challenges, 2006.
E.H. Shortliffe, Computer-Based Medical Consultations:
MYCIN, Elsevier/North Holland, New York, 1976.
Green, M., Bjork, J., Forberg, J., Ekelund, U., Edenbrandt,
L. and Ohlsson, M.: Comparison between neural net-
works and multiple logistic regression to predict acute
coronary syndrome in the emergency room. Artificial
Intelligence in Medicine, Vol. 38, (2006) 305-318.
Peled, A.: Plasticity imbalance in mental disorders the neu-
roscience of psychiatry: Implications for diagnosis
and research. Medical Hypotheses, Vol. 65, (2005)
947-952.
Politi, E., Balduzzi, C., Bussi, R. and Bellodi, L. Artificial
neural networks: a study in clinical psychopharmacol-
ogy, Psychiatry Research, Vol. 87, (1999) 203-215.
Suzuki, K., Shiraishi, J., Abe, H. MacMahon, H. and Doi,
K.: False-positive reduction in computer-aided diag-
nostic scheme for detecting nodules in chest radio-
graphs by means of massive training artificial neural
network. Academic Radiology, Vol. 12, (2005) 191-
201.
Remzi M., Djavan B., Seitz C., Hruby S., Marberger M.:
Artificial Neural Networks to predict the outcome of
repeat prostate biopsies. J Urol (2001).
Batuello JT., Gamito EJ, Crawford ED., Han M., Partin
AW., McLeod DG., et al.: Artificial Neural Network
Model for the assessment of lymph node spread in pa-
tients with clinically localized prostate cancer. Urol-
ogy, Vol. 57, (2001) 481–485.
Remzi M., Djavan B.: Artificial neural networks in urology.
European Urology, (2004) 33-38.
Gil, D., Soriano, A., Ruiz, D., Montejo, C.A.: Develop-
ment of an artificial neural network for helping to di-
agnose diseases in urology. Proceedings of the Bio-
Inspired mOdels of NEtwork, Information and Com-
puting Systems (BIONETICS’06), (2006).
Gil, D., Soriano, A., Ruiz, D., Garc´ıa, J., Fl´orez, F.; Deci-
sion support system for diagnosing dysfunctions inthe
lower urinary tract. 3rd European Medical and Biolog-
ical Engineering Conference. IFMBE European Con-
ference on Biomedical Engineering. (2005).
Ruiz, D., Garc´ıa, J.M., Mac´ıa, F., Soriano, A.: Robust Mod-
elling of Biological Neuroregulators. IEEE Engineer-
ing in Medicine and Biology Society, (2005).
Jiang, Y., Zheng, J., Peng, C. and Li, Q.: A multilayer
perceptron-based medical decision support system for
heart disease diagnosis. Elsevier, Expert Systems with
Applications, Vol. 30, (2006) 272-281.
Kohonen, T.: Self-Organization and Associative Memory,
Berlin Heidelberg, Springer-Verlag, (1988).
Kohonen, T.: The self-organizing map. Proceedings of the
IEEE, 78, 9, (1990) 1464-1480.
Kohonen, T.: Self-Organizing Maps, Berlin Heidelberg,
Springer-Verlag, (2001).
Fritzke, B.: Growing Cell Structures - A Self-organizing
Network for Unsupervised and Supervised Learning,
(1993) 93-126.
Fritzke, B.: Unsupervised ontogenic networks. In Fiesler
and Beale (eds.). Handbook of Neural Computation,
IOP Publishing Ltd. and Oxford University Press,
(1997).
