
Inertial-based Gait Analysis Applied to Patients with Parkinson Disease
Joana Sousa
1
, Joana Silva
2 a
, Ricardo Leonardo
2 b
, Hugo Gamboa
1,2 c
and Josefa Domingos
3 d
1
Faculdade de Ci
ˆ
encias e Tecnologia, FCT, Universidade Nova de Lisboa, 2829-516 Caparica, Portugal
2
Associac¸
˜
ao Fraunhofer Portugal Research, Rua Alfredo Allen 455/461, 4200-135 Porto, Portugal
3
Grupo de Patologia M
´
edica, Nutric¸
˜
ao e Exerc
´
ıcio Cl
´
ınico do Centro de Investigac¸
˜
ao Interdisciplinar Egas Moniz (CiiEM),
Caparica, Portugal
Keywords:
Gait Analysis, Parkinson’s Disease, Foot-mounted IMU, Spatio-temporal Gait Metrics, Complementary
Filter, ZUPT.
Abstract:
People with Parkinson’s disease have a high incidence of falls due to motor difficulties. Recent studies have
shown that PD patients can receive benefit from motor therapy based on cueing and feedback. This study
describes a system based on a foot-mounted IMU for the calculation of gait parameters applied to different
datasets of healthy elderly people, geriatric patients and patients with PD, in order to integrate it into a real-
time acquisition system with application for tactile cueing. This system is divided into different steps: the
identification of gait cycles and their events, the estimation of the path of the foot, which includes the esti-
mation of the orientation of the foot, the application of methods to correct the error derived from the double
integration of acceleration such as ZUPT, and finally the estimation of the different gait metrics. The results
show that the algorithm developed is an accurate method for stride segmentation and is considered adequate
to assess the gait metrics for gait evaluation of patients with motor difficulties.
1 INTRODUCTION
Parkinson’s disease (PD) is the second most common
neurodegenerative disease and it is increasing with the
aging of the world population. However its low inci-
dence, it affects about 1% of the population over 65
years old worldwide (Moore et al., 2005). PD causes
a high incidence of falls due to death dopaminergic
neurons in the substantia nigra that causes movement
disorders (Moore et al., 2005). In fact, ”it is esti-
mated that 60.5% of patients with PD have at least
one fall and that 39% have recurrent falls” (Kalilani
et al., 2016), which consequently contributes to the
increased risk of fractures in patients. The symptoms
of the disease are gradual and include problems with
gait, balance, posture, in addition to tremors, bradyki-
nesia and rigidity. The disease can’t be cured, but it
is usually treated with medication, such as levodopa,
and in some cases with neurosurgery, but the patients
benefit from physiotherapy since diagnose to improve
physical capacity, gait and balance (Tomlinson et al.,
a
https://orcid.org/0000-0002-6214-5868
b
https://orcid.org/0000-0003-2695-4462
c
https://orcid.org/0000-0002-4022-7424
d
https://orcid.org/0000-0001-9390-6183
2014).
With the increase in the number of Parkinson’s pa-
tients, the high incidence of falls and considering the
need for physical therapy to improve their quality of
life, it is increasingly important to develop solutions
that allow patients to improve their motor skills in
a common environment, outside the physical therapy
environment. In addition, access to physical therapy
and the frequency of sessions can often become in-
creasingly limited, which motivates the development
of a device that assesses the patient’s gait and pro-
vides feedback to correct certain positions, or even
stimulates a more controlled gait.
In fact, motor impairment of PD leads to specific
gait characteristic in PD, such as reduced step length,
longer step time and consequently reduced gait speed
and less regular gait cycle (Mariani et al., 2013).
Stance duration and double support duration were in-
creased for the Parkinson’s population, whereas sin-
gle support duration, mean cadence, and heel-to-heel
base of support were markedly reduced (Nelson et al.,
2002). Some of these parameters are difficult to as-
sess and vary significantly from patient to patient, so
an additional system that can estimate gait metrics is
beneficial in helping doctors confirm their observa-
Sousa, J., Silva, J., Leonardo, R., Gamboa, H. and Domingos, J.
Inertial-based Gait Analysis Applied to Patients with Parkinson Disease.
DOI: 10.5220/0010328403270334
In Proceedings of the 14th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2021) - Volume 4: BIOSIGNALS, pages 327-334
ISBN: 978-989-758-490-9
Copyright
c
2021 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
327

tions and diagnoses during long-term trails.
Gait analysis is usually performed in a laboratory
setting using a motion capture system or a pressure-
sensitive walkway. These systems are considered
gold standard in terms of accuracy. These systems al-
low a high degree of accuracy in measurement, how-
ever they are very complex to use on a daily ba-
sis and are only suitable for laboratory or hospital-
like environments such as physiotherapy clinics, due
to their high monetary value and little ease of use
(Tunca et al., 2017) and adaptation to any environ-
ment. Therefore, a portable system of good precision
that can estimate the parameters of gait is an added
advantage for the evaluation of motor disorders and
for monitoring motion performance in PD.
The evolution of wearable sensor technology en-
abled to obtain an acquisition system with a low cost
and size that allows gait dysfunction and motor symp-
toms to be assessed. The most used wearable sen-
sors are inertial sensors packed into inertial measure-
ment units (IMUs) (Tunca et al., 2020). IMU consists
of a unit that integrates tri-axial accelerometer, gyro-
scope and magnetometer. This unit also allows BLE
or Wireless communication with a device, being able
to acquire synchronized data in real time from several
IMUs, in order to obtain a complete information of
the gait (Tunca et al., 2017).
Currently, there are many studies of inertial sen-
sors in the scope of gait assessment and gait monitor-
ing performance, mainly for lower-body gait analy-
sis with foot-located IMUs. In fact, one of the major
focuses of research in inertial sensors is to develop a
system that extracts gait metrics, both spatial and tem-
poral, over short and long gait paths for accurate as-
sessment of dysfunctions. Methodologies with good
precision have been developed that combine step seg-
mentation methods by identifying different walking
moments and the calculation of spatial metrics (Mar-
iani et al., 2013; Tunca et al., 2017). These studies
vary by the segmentation method of the gait, (Rampp
et al., 2015) use multi-dimensional subsequence dy-
namic time warping (msDTW) for geriatric patients
and some patients with PD, (Ferrari et al., 2016;
Tunca et al., 2017) used algorithms that involve the
identification of the angular velocity peaks for Parkin-
son’s patients and healthy.
This study describes a foot-mounted IMU system
for the calculation of gait parameters applied to differ-
ent datasets of healthy people, geriatric patients and
patients with PD, in order to integrate it into a real-
time acquisition system and application of biofeed-
back. This system is divided into different steps: the
identification of gait cycles and their events, the esti-
mation of the path of the foot, which includes the esti-
mation of the orientation of the foot, the application of
methods to correct the error derived from the double
integration of acceleration such as zero-velocity up-
dates algorithms (ZUPT), and finally the estimation
of the different gait metrics.
2 METHODS
2.1 Acquisition Protocol and Datasets
The requirements of development of this system had
the partnership of the Portuguese Parkinson Disease
Patient Association (APDPk). However, due to the
pandemic situation that was established, the coopera-
tion was no longer possible. The pandemic prevented
the involvement of patients in the association, as they
are a group of potential risk. Patient participation
would undermine safety measures and increase risk
of contagion. For this reason, this section describes
the datasets used, which are mostly public.
In that sense, it was used 2 datasets and all have
certain requirements. The datasets include samples of
triaxial inertial data from sensors placed on both feet
of the patient while walking and data from an accu-
rate reference system to validate the methods. In the
initial phase of familiarization with the data and for
the definition of different gait metrics (such as num-
ber of steps, speed, stride length), the subjects did not
need to have a neurodegenerative disease.
The public eGaIT database (embedded Gait anal-
ysis using Intelligent Technologies) contains two
datasets which was used in this study, the Validation
Stride Segmentation and the Validation of Gait Pa-
rameters. These datasets were provided by Profes-
sor Ph.D Bjoern Eskofier of the Machine Learning
and Data Analytics laboratory at Friedrich-Alexander
University Erlangen-Nuernberg and they are accessi-
ble for collaborative research (FAU, 2015). The iner-
tial data of the both datasets were acquired using the
same material and set-up system (Barth et al., 2015;
Rampp et al., 2015).
The eGaIT database for Validating Stride Segmen-
tation consists of data from 30 individuals. Of the to-
tal participants, 10 are elderly controls, 5 male and 5
female, 10 individuals are patients with Parkinson’s
disease, 5 male and 5 female, and 10 are geriatric pa-
tients, 4 male and 6 female (Barth et al., 2015). The
mean age for the elderly controls is 64.0 ± 8.4 years,
the mean age for patients with PD is 63.8 ± 9.3 years
and for geriatric patients is 81.0 ± 4.1 years (mean ±
standard deviation). The patients with PD were eval-
uated to assess symptoms, severity and degree of dis-
ease. The ground truth of the start and end points of
BIOSIGNALS 2021 - 14th International Conference on Bio-inspired Systems and Signal Processing
328
each stride was provided with the inertial data.
The eGaIT database for Validating Gait Parame-
ters consists of data from 101 inpatient geriatric pa-
tients, where 55 are female and 46 are male, the mean
age is 82.1 ± 6.5 years and the mean height is 164.0 ±
10.0 cm (mean ± standard deviation) (Rampp et al.,
2015). The ground truth of the stride time, stride
length, swing time and stance time was also provided
with the inertial data. This data set does not contain
data from patients with Parkinson’s, but does contain
data from participants with motor difficulties. It con-
tains the ground truth of the start and end points of the
stride and the validation of different spatial and tem-
poral metrics (stride length, stride time, swing time
and stance time) from a GAITRite system, which is
useful to validate the algorithm of spatial gait param-
eters (Barth et al., 2015; Rampp et al., 2015).
2.2 Algorithm Architecture
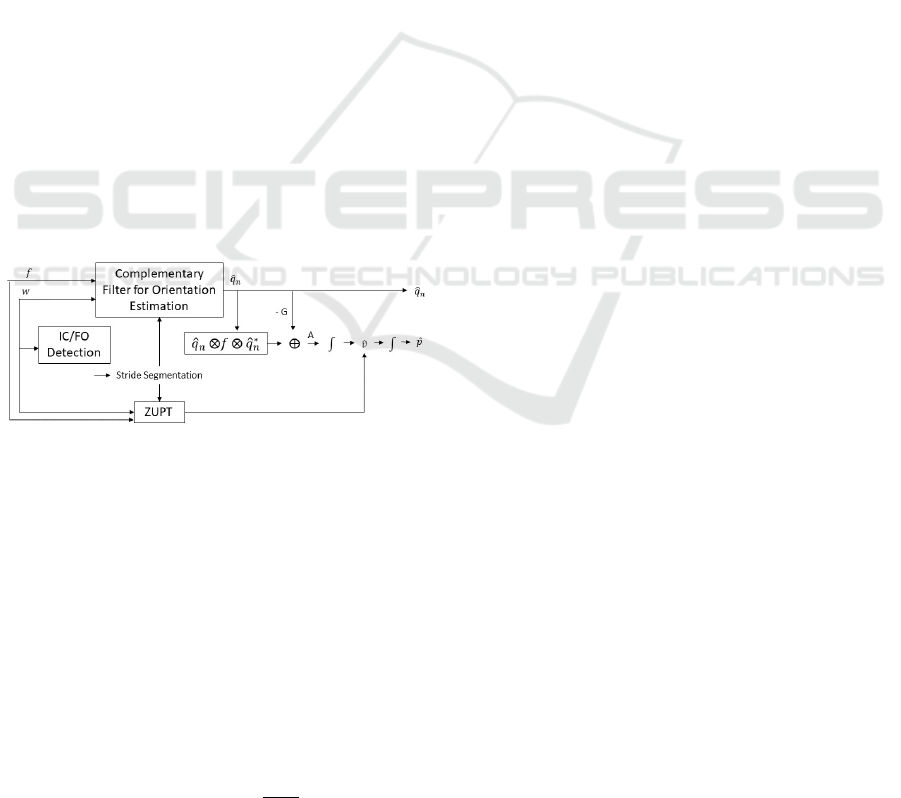
The algorithm architecture is represented in Fig.1 and
is based on two approaches. The first is the foot-
mounted complementary filter (CF) aided IMU ap-
proach for pedestrian tracking in indoor environment
(Fourati, 2015). However, the system proposed in this
study is not for estimating the trajectory, but for esti-
mating gait parameters. The second is the identifica-
tion of gait events to estimate the position for each
stride to calculate gait metrics (Rampp et al., 2015).
Figure 1: System Architecture (Barth et al., 2015).
In this sense, the complementary filter is used to
estimate the orientation of the foot along the gait, rep-
resented by the quaternion, ˆq
n
(t). The complemen-
tary filter has as inputs the raw acceleration ( f ) and
raw angular velocity given by the sensors (w). This
quaternion will be used to transform the acceleration
into the body’s reference, which is the raw accelera-
tion given by the sensor ( f ), in the Earth’s coordinate
system, a =
0 a
T
e
T
.
a = ˆq
n
(t) ⊗ f ⊗ ˆq
∗
n
(t) (1)
Gravitational acceleration G is also calculated
with the use of a quaternion, G
0
= q
n
(t) ⊗
0 0 −1 0
⊗q
∗
n
(t), G =
G
0
k
G
0
k
2
. Next, it is nec-
essary to remove the contribution of G from the ac-
celeration vector a
e
=
a
ex
a
ey
a
ez
T
, to obtain
the acceleration of movement of the foot in the earth
frame,
A(t) = a
e
(t) − G (2)
and thus it is possible to integrate the calculated ac-
celeration by obtaining the 3-D vector in the Earth co-
ordinate system. Theoretically, the resulting velocity
vector can be immediately integrated again to obtain
position. However, due to the presence of noise in the
inertial signal, which leads to changes and drifts in the
measured acceleration and errors in the orientation es-
timation by the quaternion, the immediate integration
of the acceleration would result in an estimation of
the wrong position, due to the referred accumulation
of errors. One method to reduce the error is the appli-
cation of the Zero Velocity Update (ZUPT) method,
which will be referred to in section 2.2.2. This method
is based on the correction of linear speed during gait.
Gait is a cyclic movement that results in the transfer
of weight from one foot to another alternately. When
a foot is in contact with the ground, its linear speed is
theoretically zero (Fourati, 2015). Due to the accumu-
lation of error during the referred process, the linear
speed during these support phases may not be zero,
and thus the ZUPT method identifies the moments of
zero speed and corrects the speed initially calculated.
The corrected speed is integrated to obtain position.
Previous studies (Ferrari et al., 2016; Tunca et al.,
2017) showed that this approach was not as accurate
as the Pedestrian Dead-Reckoning (PDR) system us-
ing the Kalman Filter. However, this method will be
used to calculate the position step by step, and in each
step the position will return to zero and a new orienta-
tion will be calculated by not increasing the accumu-
lation of the error throughout the acquisition period.
Thus, through position and speed it is possible to cal-
culate the spatial metrics of the gait. Also, it was used
the CF due to its easy implementation, understanding
and because it requires little computational power.
2.2.1 IC/FO Events Detection
As mentioned in the introduction, there are many lit-
erature that have studied and developed stride seg-
mentation methods and some which are based on the
identification of the initial contacts of the foot on the
floor (IC) and the foot-off (FO), the lifting of the foot
from the floor (Ferrari et al., 2016; Tunca et al., 2017),
but few are capable of running in closed loop for real
time acquisition or for real time gait events. The iden-
tification of these events is important because from
them it is possible to estimate all temporal metrics of
the gait, such as stride time, swing time, stance time,
cadence and double support. The evaluation of the
Inertial-based Gait Analysis Applied to Patients with Parkinson Disease
329
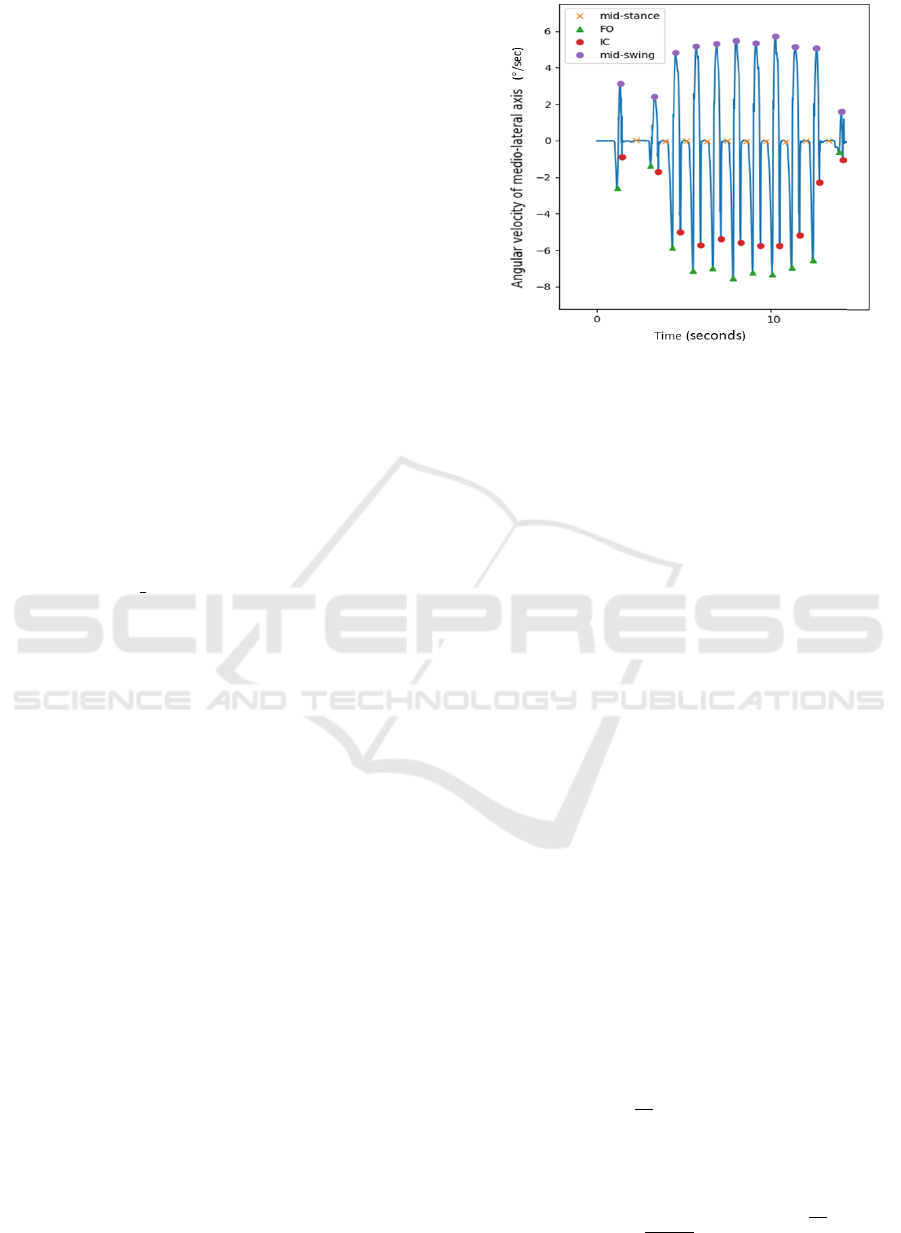
angular velocity signal according to the medio-lateral
axis is a method with good accuracy, ideal for auto-
matic detection of gait events. The method developed
is based in (Ferrari et al., 2016).
Unlike studies of (Ferrari et al., 2016) that cal-
culate in the first instance the positive peaks, which
represent the moments that the foot is rotating coun-
terclockwise considering the side view of a person
walking to the right. The developed algorithm first
identifies the negative peaks that may correspond to
the FO / IC events, moments of the gait when the foot
is rotating clockwise. Calculating these events first,
it is guaranteed that no steps are missed in this pro-
cessing. Next, the positive peaks, which can repre-
sent mid-swing events, are calculated and it is veri-
fied if this positive peak is between the first and the
second detected peak. If the previous fact occurs then
the identified peaks can be considered to correspond
in fact to the events of the gait. If not, that is, if a
first FO peak has been identified and does not follow
a mid swing, the FO is not considered, or if a negative
second peak, a possible IC, has not been identified af-
ter a positive peak, then it will be looked for a next
negative peak following the mid swing found.
The identification of positive and negative peaks is
based on the find peaks function of the Python Scipy
Signal library (Community, 2020) and takes into ac-
count limits and conditions that the peaks must meet
in order to be recognized as IC an FO events in all
patients. These limits are based on the algorithm used
by (Ferrari et al., 2016) and adapted so that events are
detected in all datasets for healthy, geriatric and pa-
tients with PD. The developed algorithm is executed
in soft real-time and has a maximum delay of half a
step, since it is necessary to evaluate the gait from step
to step to calculate the metrics. However, this delay is
compatible for mobile applications, such as feedback
tips while walking in loop mode (Ferrari et al., 2016).
The result of the algorithm is represented in Figure 2.
With the identification of these events it is pos-
sible to segment the stride, calculate the stride time,
identify moments such as the stance phase and the
swing phase and their respective duration. But for
the calculation of the position of each step is impor-
tant to determine also the mid-stance moments. The
mid-stance events were defined has the middle of the
stance phase, being the stance phase the period be-
tween a IC and a FO moments.
2.2.2 ZUPT Algorithm
The gait can be divided into two important phases,
which can be identified through the inertial signal of
the IMU fixed on the foot of a person walking. The
first is the swing phase which means that the foot and
Figure 2: Detection of IC, FO, mid-stance and mid-swing
events of a Patient with PD from a eGait dataset.
the IMU are in the air. The other is the stance phase,
which is the period that the foot is in contact with
the floor. According to the studies of (Fourati, 2015),
during the stance phase the angular and linear veloc-
ity must be very close to zero and the accelerometer
should measure only the earth gravitational accelera-
tion, because the foot is on the ground and theoretical
it is not moving. However, this phase includes the IC
event, the foot-flat period in which the foot is com-
pletely resting on the floor and the FO event, when the
foot is leaving the ground but is still in contact. Con-
sidering that the IMU is fixed only in one part of the
foot, it does not present speed only during the foot-
flat period (Wang et al., 2015). Thus, Multi-condition
ZUPT presented by (Guo et al., 2015) was adapted for
this study, aims to identify these moments, the periods
when the inertial sensor is not in motion, representing
them with a logical function assigning 1 when the lin-
ear velocity should be zero and 0 when the velocity
does not need correction. This algorithm uses data
from the accelerometer and gyroscope and presents
several conditions, as follows:
1. Since the acceleration in periods 1 must be just the
acceleration of gravity, the magnitude of the ac-
celeration (|a
k
|) must be between two limits. The
limits are defined around the value 9.8 m/s
2
.
C1 =
1 8 <
|
a
k
|
< 11
0 otherwise
(3)
2. The acceleration variance must be above a given
threshold where a
b
k
is a mean acceleration value at
time k, and s is the size of the averaging window
(s = 15). The variance is computed by:
σ
a
b
k
2
=
1
2s + 1
k+8
∑
j=k−8
a
j
− a
b
k
2
(4)
BIOSIGNALS 2021 - 14th International Conference on Bio-inspired Systems and Signal Processing
330

The second condition is defined by this way:
C2 =
1 σ
a
b
k
< 0.5
0 otherwise
(5)
3. The magnitude of the angular rate (|ω
k
|) must be
below a given threshold.
C3 =
1
|
ω
k
|
< 0.8
0 otherwise
(6)
The logical result of the ZUPT algorithm is ob-
tained when the 3 conditions are satisfied. The final
logical result is filtered out by a median filter with
neighboring window of 29 samples. The Figure 3
shows de result of this multi-condition algorithm from
data of a patient with parkinson disease.
Figure 3: ZUPT algorithm applied to all data acquisition of
a patient with PD from the eGait dataset.
This method will work for each stride and if the
zero speed zones identified by the ZUPT are outside
the IC-FO period, the system does not consider the
ZUPT phases and considers the stance phase identi-
fied by the event detection algorithm 2.2.1.
2.2.3 Gait Parameters
Temporal gait parameters were calculated based on
the gait events identified from each dataset, and were
defined from two consecutive strides. The stance time
is defined in seconds as the duration of stance phase,
starting with initial-contact (IC) or heel strike (HS)
and ending with foot-off (FO) or toe-off (TO) of the
same foot. The swing time is the duration of swing
phase, starting with FO and ending with IC of the
same foot. The stride time is the duration of a gait cy-
cle. Considering the instant when the event occurred
of the stride i as t
IC
(i) and t
FO
(i), it is possible to de-
fine each time parameter (Rampp et al., 2015).
Stride Time (i) = t
IC
(i + 1)−t
IC
(i) (7)
Swing Time (i) = t
IC
(i) − t
FO
(i) (8)
Stance Time (i) = t
FO
(i + 1) −t
IC
(i) (9)
After calculating the 3D position based on the de-
veloped system, it is easy to calculate the spatial met-
rics. The stride length is the distance between two
successive placements of the same foot, defined as the
two norm of a 2-D vector containing the distance in
x- (posterior-anterior axis) and z- (medio-lateral axis)
direction at the end of the stride, since the position in
the start of the stride is zero (Rampp et al., 2015).
Stride length =
p
x
(n)
p
z
(n)
2
=
q
p
x
(n)
2
+ p
z
(n)
2
(10)
3 RESULTS
As in this study, different datasets are used with differ-
ent validation metrics. This section is divided into two
parts: stride segmentation results of the two datasets
of the eGait database and gait metrics results of the
second eGait dataset.
3.1 Stride Segmentation Results
The IC / FO event detection algorithm, described in
2.2.1, is the method used in this study to segment the
stride. The accuracy of this algorithm was tested in
the eGait datasets. Validation of this algorithm was
achieved using the annotated strides of the dataset.
The annotation of the samples referring to the begin-
ning and to the end of each stride, for each subject,
was performed by manual identification an labeling
of gyroscope peaks. In these studies, the stride was
considered from the moment when the foot leave the
ground (FO) of one cycle to the FO of the next cycle.
In Table 1 the results related to gait segmenta-
tion for the different patients in the Validation of
Stride Segmentation dataset of the eGait database
are provided. The table quantifies the number of
strides, more specifically, the number of strides an-
notated in the study (Annotated Strides), the number
of strides detected by the developed algorithm (De-
tected Strides) and the number of strides that were
detected by the developed algorithm and that coincide
with the annotated strides (True Detected Strides). In
the study, patients walked in a straight line for 10 me-
ters, four times, and at the end of each straight line
the subject rotates 180 degrees to walk in the opposite
direction. The strides annotated in the dataset do not
refer of the moments when the patient is changing di-
rection. With this information it is possible to verify
that all the annotated strides are correctly identified
Inertial-based Gait Analysis Applied to Patients with Parkinson Disease
331

Table 1: Results of Stride Segmentation of the eGait Dataset - Validation of Stride Segmentation. Mean of the stride period
annotated, the mean of the stride period calculated, the mean error of the start of each stride and end of each stride are given
as mean ± standard deviation.
Patients Foot
Annotated
Nr. Strides
Detected
Nr. Strides
True Detected
Nr. Strides
Mean Strides
per Patient
Annotated
Stride Period
(sec.)
Detected
Stride Period
(sec.)
Mean Error
Start Stride
(samples)
Mean Error
End Stride
(samples)
PD
Right 248 248 312 24.8 ± 1.8 0.94 ± 0.01 0.94 ± 0.04 -0.98 ± 0.04 0.98 ± 0.04
Left 248 248 313 24.8 ± 1.6 1.04 ± 0.02 1.04 ± 0.02 -1.01 ± 0.02 1.01 ± 0.02
Geriatric
Right 384 384 491 38.4 ±n11.0 1.35 ± 0.09 1.35 ± 0.09 -0.99 ± 0.02 0.99 ± 0.02
Left 381 381 490 38.1 ± 10.9 1.34 ± 0.08 1,34 ± 0,08 -0,99 ± 0.00 0,993 ± 0.00
Control
Right 242 242 306 24.2 ± 2.7 1.09 ± 0.03 1.09 ± 0.03 -0.99 ± 0.01 1.00 ± 0.01
Left 220 220 310 24.4 ± 2.2 1.10 ± 0.03 1.10 ± 0,03 -1.00 ± 0.00 1.00 ± 0.00
by the algorithm for all patients. However, the algo-
rithm has identified more strides, that are those that
correspond to moments of changing of direction.
The results table 1 also shows the average num-
ber of strides taken by the patients during walking in
straight line, that is, over a distance of 40 meters, the
average period, in seconds, of the stride annotated in
the study, and the average period of the true detected
strides. This period was obtained by dividing the du-
ration of the strides (in samples) by the sampling rate,
which is 102.4 Hz. The average number of steps taken
by geriatric patients is much higher than the average
number of other patients (PD or controls). This group
also has a much higher average age, as can be seen
in 2.1, which implies more limited mobility, leading
to a slower walking cycle (Stride Period) and since
all groups walked exactly 40 meters, it is possible to
deduce that they have a shorter gait length. Patients
with Parkinson have in average the same number of
steps and a relatively longer gait period than the con-
trol group, composed by elderly people. The differ-
ence is also not very clear, since the staging of Hoehn
and Yahr is on average 1.7 ± 0.9, which is a not very
advanced stage in the disease where changes in gait
begin to exist (Keus et al., 2009).
To evaluate the quality of the segmentation, the
average of the differences between the samples at
the beginning of each stride annotated in the dataset
and the ones calculated by the algorithm were calcu-
lated and is presented in the Table 1 (mean error start
stride). The same was retrieved for the end of the
stride, the average of the differences between the sam-
ples referring to the end of each stride annotated and
the ones calculated by the algorithm is also presented
in the Table 1 (mean error end stride). These aver-
age errors are very low and have the same modulus
value for detecting the beginning and the end of each
stride, which means that there is only a lag of about
1 sample leading to an equal stride time between the
annotated values and the ones retrieved by the algo-
rithm developed. These results show a great accuracy
of the algorithm to identify gait events, more specif-
ically FO, compared to other methods of gait events
identification (Barth et al., 2015).
Table 2 presents the results of gait segmentation
for the right and left foot of 97 subjects, inpatient geri-
atric patients, from the Validation of Gait Parameters
dataset of the eGait database. Four patients were ex-
cluded from the dataset because there were inconsis-
tencies in the data provided. The validation of the
stride annotations was carried out in the same way as
mentioned for the previous dataset. The table shows
the total number of strides annotated by the dataset in
the 10-meter path, the number of strides detected by
the algorithm, and the average number of strides per
subject during the acquisition period. To check the
accuracy of the algorithm, the average period of the
strides and the average error were also calculated, as
presented in the previous table 1. The results for this
dataset proved the accuracy of stride detection, since
all strides have been correctly identified with a small
average error leading to a stride period’s mean differ-
ence of milliseconds . In the next section, the results
of the temporal gait metrics and the stride length for
this dataset will be evaluated.
3.2 Gait Parameters Results
This section presents the results for the gait metrics
retrieved for the second dataset. The metrics calcu-
lated were stride length, stride time, stance time and
swing time, as shown in the Table 3. The results were
from 97 subjects of 101 patients of the dataset. The
Table shows the average errors (Mean error), defined
by the average of the differences between the metrics
calculated by the developed system and the metrics
provided in the annotated dataset, the absolute error
(Abs. Error), which is the average of the absolute er-
rors and the correlation that is defined by the Spear-
man correlation coefficient.
Compared to the study of (Rampp et al., 2015),
which uses the same dataset to analyze the same pa-
rameters of the gait, it is possible to verify that the
mean and absolute error of the stride time calculated
in our study for the right foot are smaller (Mean er-
ror=0.002 ± 0.043, Abs.error=0.023 ± 0.044) than
in the previous study (Mean error = 0.002 ± 0.068,
Abs error = 0.029 ± 0.062) and for the left foot
BIOSIGNALS 2021 - 14th International Conference on Bio-inspired Systems and Signal Processing
332

Table 2: Results of Stride Segmentation of the eGait Dataset - Validation of Gait Parameters. mean number of strides per
patient, mean stride period annotated in the dataset, mean stride period calculated by the algorithm, mean error of the initial
of each stride, and mean error of end of each stride are given as mean ± standard deviation.
Foot
Annotated
Nr. Strides
Detected
Nr Strides
Mean Strides
per Patient
Annotated
Stride Period
(samples)
Detected
Stride Period
(sec.)
Mean error
Start Stride
(sec.)
Mean error
End Stride
(Samples)
Left 663 663 6.84 ± 1.98 1.22 ± 0.19 1.22 ± 0.18 -0.09 ± 1.05 0.44 ± 1.81
Right 659 659 6.79 ± 1.96 1.23 ± 0.19 1.23 ± 0.18 -0.15 ± 1.07 0.25 ± 0.17
Table 3: Results of Gait Parameters of the eGait Dataset - Validation of Gait Parameters. Mean error and abs. error are given
as mean ± standard deviation. Time in sec and length in cm.
Foot Parameter
Mean
(Inertial-Sensors)
Mean
(GAITRite)
Mean Error Abs. Error Correlation Total
Right
Stride Time
1.22 ± 0.18 1.22 ± 0.18 0.002 ± 0.043 0.023 ± 0.044 0.97
756 Strides
97 Subj.
Stance Time
0.78 ± 0.15 0.85 ± 0.15 -0.07 ± 0.05 0.07 ± 0.04 0.96
Swing Time
0.44 ± 0.08 0.37 ± 0.08 0.07 ± 0.04 0.07 ± 0.04 0,91
Stride Length
83.0± 23.7 83.0 ± 21.3 -0.90 ± 9.76 7.45 ± 6.03 0.90
Left
Stride Time
1.23 ± 0.19 1.23 ± 0.19 0.003 ± 0.039 0.023 ± 0.045 0.98
759 Strides
97 Subj.
Stance Time
0.78 ± 0.15 0.84 ± 0.16 -0.07 ± 0.04 0.068 ± 0.039 0.96
Swing Time
0.45 ± 0.07 0.38 ± 0.07 -0.068 ± 0.03 0.068 ± 0.038 0.86
Stride Length
82.0 ± 25.4 83.0 ± 22.0 -0.98 ± 9.76 8.61 + 9.71 0.89
besides being higher (Mean error=0.003 ± 0.039,
Abs.error=0.002 ± 0.045), the values are very simi-
lar. The correlation coefficient of the stride time met-
ric is higher in this study, 0.97, since the correlation
achieved in the previous study was 0.95. With this in-
formation it is possible to verify that the segmentation
algorithm developed in our study is quite accurate.
However, for the remaining metrics, stance time,
swing time and stride length, the results are not so
comparable. The average errors obtained in the study
of (Rampp et al., 2015) were -0.008 ± 0.045, 0.009
± 0.069, -0.26 ± 8.37, for the respective metrics. The
average errors calculated by the developed algorithm,
for the time metrics, are higher, with an order of one
decimal place of difference. The reason for the val-
ues being so different is related to the approach used
to calculate the IC and FO events. There are differ-
ences in the literature in the method used to identify
the events at the beginning and at the end of the gait.
According to (Ferrari et al., 2016), the IC and FO
events are considered the negative peaks of angular
velocity in the medio-lateral axis, whereas according
to the study of (Rampp et al., 2015), the FO events
are considered to be the instants when the gyroscope
signal in to the same axis crosses the zero.
The stride length presents a mean error of less than
1 cm, both for the right foot and for the left foot,
which is a very small error considering the use of this
system for rehabilitation purposes. However, the av-
erage and absolute errors are much higher than the
one calculated in the study of (Rampp et al., 2015)
(Mean error = -0.26 ± 8.37, Abs error = 6.26 ±
5.56). The authors calculated the drift of the gyro-
scope that is contained in the linear acceleration, with-
drew its contribution, and after that the integration
for the speed was accomplished. In this study, the
only way of correcting the inertial errors was applying
the ZUPT method, which corrects the linear velocity
during the stride considering that in the moments of
stance the velocity must be zero. This method only
corrects the drift of the result of the integration of ac-
celeration and does not correct the error associated
with the gyroscope. However, in addition to the fact
that this correction does not exist, the results do not
differ significantly from the annotated values.
4 CONCLUSIONS
This study describes a foot-mounted IMU system for
the calculation of gait parameters applied to differ-
ent datasets of healthy people, geriatric patients and
patients with PD, in order to integrate it into a real-
time acquisition system and application of biofeed-
back. This system was divided into different steps:
the identification of gait cycles and their events, the
estimation of the path of the foot, which includes the
estimation of the orientation of the foot, the applica-
tion of methods to correct the error derived from the
double integration of acceleration such as ZUPT, and
finally the estimation of the different gait metrics.
There are different walking segmentation methods
(Haji Ghassemi et al., 2018) more robust methods like
msDTW, eDTW, sDTW and hHMM and more con-
ventional methods like peak detection algorithms. A
recent study of (Barth et al., 2015) showed that the
peak detection algorithm used has, in the best of sce-
narios, around 84% accuracy and 90% F-Score, for
geriatric patients and that when using the msDTW
method the accuracy is between 88% and 90% and
the F-measure is between 96% and 98%. Thus, the
algorithm developed in the study, in addition to be-
ing based on conventional peak detection methods,
Inertial-based Gait Analysis Applied to Patients with Parkinson Disease
333

can be compared to robust methods at the level of
precision, given the results obtained in the identifica-
tion of all strides for the datasets used. This method,
also presents an ideal configuration for a possible in-
tegration in a real-time system, which is a prospective
breakthrough in this work.
It was presented a complete algorithm that allows
gait metrics to be calculated using data from iner-
tial sensors in patients with motor difficulties, geri-
atric patients. This system presents adequate results
to make the specific gait evaluation for the right and
left foot. Although this system present less accurate
results than the analogue study (Barth et al., 2015), is
considered adequate for the scope of gait physiother-
apy. In the future, we intend to calculate other gait
metrics such as double support period, stride width,
swing width and gait speed, which allow a more de-
tailed analysis for people with Parkinson’s, and also
integrate it in a real-time system that allows feedback
to patient whenever the algorithm evaluates a risky
gait pattern, based on spatial parameters.
ACKNOWLEDGEMENTS
Supported by project Indoor Activity Notification for
Vigilance Services (AAL-2018-5-116), funded under
the AAL JP and co-funded by the European Commis-
sion and the National Funding Authorities of Portu-
gal, Belgium, and Switzerland.
REFERENCES
Barth, J., Oberndorfer, C., Pasluosta, C., Sch
¨
ulein, S.,
Gassner, H., Reinfelder, S., Kugler, P., Schuld-
haus, D., Winkler, J., Klucken, J., and Eskofier,
B. M. (2015). Stride segmentation during free walk
movements using multi-dimensional subsequence dy-
namic time warping on inertial sensor data. Sensors,
15(3):6419–6440.
Community, T. S. (2020). scipy.signal.find peaks — SciPy
v1.5.3 Reference Guide.
FAU (2015). Digital Biobank - eGaIT (embedded
Gait analysis using Intelligent Technologies).
https://www.mad.tf.fau.de/research/activitynet/digital-
biobank/.
Ferrari, A., Ginis, P., Hardegger, M., Casamassima, F., Roc-
chi, L., and Chiari, L. (2016). A mobile Kalman-filter
based solution for the real-time estimation of spatio-
temporal gait parameters. IEEE Trans. Neural Syst.
Rehabil. Eng., 24(7):764–773.
Fourati, H. (2015). Heterogeneous data fusion algorithm for
pedestrian navigation via foot-mounted inertial mea-
surement unit and complementary filter. IEEE Trans.
Instrum. Meas., 64(1):221–229.
Guo, H., Uradzinski, M., Yin, H., and Yu, M. (2015). Indoor
positioning based on foot-mounted IMU. Bulletin of
the Polish Academy of Sciences: Technical Sciences,
63(3):629–634.
Haji Ghassemi, N., Hannink, J., Martindale, C. F., Gaßner,
H., M
¨
uller, M., Klucken, J., and Eskofier, B. M.
(2018). Segmentation of gait sequences in sensor-
based movement analysis: A comparison of methods
in Parkinson’s disease. Sensors, 18(1):1–15.
Kalilani, L., Asgharnejad, M., Palokangas, T., and Durgin,
T. (2016). Comparing the incidence of falls/fractures
in Parkinson’s disease patients in the US population.
PLoS ONE, 11(9):1–11.
Keus, S. H., Munneke, M., Nijkrake, M. J., Kwakkel, G.,
and Bloem, B. R. (2009). Physical therapy in Parkin-
son’s disease: Evolution and future challenges. Move-
ment Disorders, 24(1):1–14.
Mariani, B., Jim
´
enez, M. C., Vingerhoets, F. J., and
Aminian, K. (2013). On-shoe wearable sensors for
gait and turning assessment of patients with parkin-
son’s disease. IEEE. Trans. Biomed. Eng., 60(1):155–
158.
Moore, D. J., West, A. B., Dawson, V. L., and Dawson,
T. M. (2005). Molecular Pathophysiology of Parkin-
son ’ s Disease. Annual Review of Neuroscience,
26(1):57–87.
Nelson, A. J., Zwick, D., Brody, S., Doran, C., Pulver,
L., Rooz, G., Sadownick, M., Nelson, R., and Roth-
man, J. (2002). The validity of the GaitRite and the
functional ambulation performance scoring system in
the analysis of Parkinson gait. NeuroRehabilitation,
17(3):255–262.
Rampp, A., Barth, J., Sch
¨
ulein, S., Gaßmann, K. G.,
Klucken, J., and Eskofier, B. M. (2015). Inertial
Sensor-Based Stride Parameter Calculation From Gait
Sequences in Geriatric Patients. IEEE. Trans. Biomed.
Eng., 62(4):1089–1097.
Tomlinson, C. L., Patel, S., Meek, C., Herd, C. P., Clarke,
C. E., Stowe, R., Shah, L., Sackley, C. M., Deane,
K. H., Wheatley, K., and Ives, N. (2014). Physio-
therapy for Parkinson’s disease: a comparison of tech-
niques. CDSR, pages 1–108.
Tunca, C., Pehlivan, N., Ak, N., Arnrich, B., Salur, G.,
and Ersoy, C. (2017). Inertial sensor-based robust gait
analysis in non-hospital settings for neurological dis-
orders. Sensors, 17(4):1–29.
Tunca, C., Salur, G., and Ersoy, C. (2020). Deep Learn-
ing for Fall Risk Assessment with Inertial Sensors:
Utilizing Domain Knowledge in Spatio-Temporal
Gait Parameters. IEEE J. Biomed. Health Inform.,
24(7):1994–2005.
Wang, Z., Zhao, H., Qiu, S., and Gao, Q. (2015). Stance-
Phase Detection for ZUPT-Aided Foot-Mounted
Pedestrian Navigation System. IEEE/ASME Trans.
Mechatronics, 20(6):3170–3181.
BIOSIGNALS 2021 - 14th International Conference on Bio-inspired Systems and Signal Processing
334
