
Architecture and Low Power Management of a Deep-tissue Medical
Implant System Powered by Human Body Energy Harvesting
Elisabeth Benke
1a
, Adrian Fehrle
1b
, Johannes Ollech
2c
, Simon Schrampfer
2d
and Jörg Franke
1e
1
Institute for Factory Automation and Production Systems, Friedrich-Alexander-Universität Erlangen-Nürnberg, Germany
2
Sentinum UG, Nürnberg, Germany
Keywords: Implantable Medical Devices, Energy Harvesting, Power Management, Hybrid Energy Storage Systems.
Abstract: Active mechatronic implants applied to provide therapy of insufficient bodily functions and acquisition of
biomedical data are an emerging field in the context of Medicine 4.0. Wireless data transmission between the
implant and out-body devices enables patients and health care professionals to access physiological data as
well as take technical control and also allows for home monitoring solutions. Due to the limitations associated
with primary batteries or conventional wireless power transferring methods in deep-tissue layers, human body
energy harvesting is a promising alternative or complement for power supply. A high efficient power
management in order to reduce the implanted device’s energy consumption is not only requested to effectively
use the limited amounts of energy harvested but also contributes to extend implantation times and thus avoid
invasive surgical procedures. This paper presents solution approaches for both software- and hardware-based
low power management and storage options for active deep-tissue implants using hybrid energy storage
systems and considering miniaturisation requirements of devices powered by energy harvesting.
1 INTRODUCTION
While battery-powered medical implant devices such
as pacemakers, neurostimulators or cochlear implants
are commonly applied and widely accepted, the trend
towards energy harvesting from the human body not
only opens a completely new field of novel energy
self-sufficient implants with new actuator principles,
but also addresses the disadvantages associated with
primary battery technologies, such as dominating the
device’s size as well as the frequent need for
replacement or recharging. Devices powered by
harvested energy have a longer lifetime and are
considered to provide more comfort and safety than
battery-powered implants (Hannan et al., 2014).
Conventional approaches for the energy supply of
implantable medical devices (IMDs) such as
transferring energy from outside the body using wires
or inductive coupling are associated with several
a
https://orcid.org/0000-0002-6610-4430
b
https://orcid.org/0000-0001-9803-5620
c
https://orcid.org/0000-0002-4917-8964
d
https://orcid.org/0000-0002-3594-8286
e
https://orcid.org/0000-0003-0700-2028
limitations, especially regarding size, bio-
compatibility and the implantation depth power can
be transferred into. Particularly the powering of deep-
tissue devices with implantation depths >10 cm
presents a challenge, making body energy harvesting
an interesting alternative or complement.
According to Paulo J. & Gaspar P.D. (2010) the
produced power of the human body ranges between
81 W during sleep and 1630 W while sprint walking.
As this is potentially sufficient energy to power
microelectronic devices, several approaches to
harvest energy from the human body have been
proposed. However, the usable amount of harvested
energy to power medical devices underlies several
limitations, requesting a low power architecture
design as well as an efficient power management. The
required miniaturisation of the harvesting device and
low efficiency factors restrict the total amount of
energy that can be harvested. Dependent on the
harvesting principle, the used energy source may not
Benke, E., Fehrle, A., Ollech, J., Schrampfer, S. and Franke, J.
Architecture and Low Power Management of a Deep-tissue Medical Implant System Powered by Human Body Energy Harvesting.
DOI: 10.5220/0009107302270234
In Proceedings of the 13th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2020) - Volume 1: BIODEVICES, pages 227-234
ISBN: 978-989-758-398-8; ISSN: 2184-4305
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
227

be permanently available. Furthermore, the human
body presents a perfectly balanced energy system – the
consequences of a sudden energy removal from its
inner workings have not been well studied so far.
The design of active deep-tissue IMDs presents
numerous challenges, not only concerning their energy
supply but also regarding a secure wireless
communication between the implant situated in deep-
tissue body layers and an out-body device.
Conventional communication principles have various
limitations, especially regarding their size and
transmission range in human body tissue.
This paper features specific challenges regarding
the system architecture of active deep-tissue IMDs
powered by human body energy harvesting. A possible
architecture of a system powering a radio module for
wireless communication and an actuator or sensor unit
as well as intended low power management options are
presented. In order to optimise the energy storage
efficiency, the combination of two different storage
technologies seems suitable since characteristic
disadvantages of one technology can be absorbed by
the other (Böhm et al., 2018). Thus hybrid systems
combining the advantages of lithium-ion batteries
(LIB) and ultracapacitors (UC) are used in the
presented system.
2 HUMAN BODY ENERGY
HARVESTING PRINCIPLES
Mateu et al. (2014) classify two ways to harvest energy
from the human body: active and passive power.
Accordingly, passive power is harvested from the
patient’s or user’s everyday actions, such as breathing
or walking motions, whereas active power is harvested
from actions the person especially executes for
harvesting reasons. This paper focuses on the former
harvesting form only.
Regarding the underlying physical principle,
harvesting methods can be divided into three types
according to Figure 1.
2.1 Mechanical Energy
Various approaches to harvest mechanical energy from
human body motions or body fluidic flows have been
presented in literature. As an example, mechanical
energy can be harvested through piezoelectric elements
in various in- and out-body positions. Approaches to
drive piezoelectric elements by the rotation of joints
(Cheng et al., 2015; Hannan et al., 2014), the vibrations
of
human breathing motions (Saida et al., 2018), the
Figure 1: Human body energy harvesting methods and
examples.
pulsating energy of the aorta (Zhang et al., 2015) or the
pressure generated in shoe soles by human walking
motions (Hannan et al., 2014; Johari & Rashid, 2017)
have been shown. Furthermore, concepts to power
harvesters by running turbine generators are proposed.
Micromechanical turbines can be driven by footstep-
induced airflow (Fu et al., 2015) or the cardiac output
blood flow in a peripheral artery (Pfenniger et al.,
2014). Niroomand & Foroughi (2016) designed a
rotary magnetic generator to harvest energy from
walking motions.
2.2 Thermal Energy
Thermal energy can be harvested from temperature
differences due to the Seebeck Effect, making this
principle an option to harvest energy from human body
heat differences. As shown in several publications
presenting thermoelectric modules, usually
comparatively low output powers in the range of µW
are generated as the associated temperature gradients
typically do not exceed a few K. In the early work of
Strasser et al. (2003) a CMOS thermoelectric generator
harvesting electrical energy from waste heat is
presented, achieving a power output of 1 µW with
generators in the size of 1 cm² and a temperature
gradient of 5 K making this sufficient to power a
human wrist watch by body heat. In (Wang Z. et al.,
2009) a thermoelectric generator creating an output
power of 0,3 nW when worn on the human body is
presented. Shi et al. (2018) conduct wrist wearing
experiments with a copper-foam-based wearable
thermoelectric generator to power a miniaturised
accelerometer.
2.3 Biochemical Energy
Bio-fuel cells are an alternative approach to power
IMDs by using bodily fluids surrounding the implant
as a potential energy source. Hereby biochemical
energy is transformed into electrical energy based on
BIODEVICES 2020 - 13th International Conference on Biomedical Electronics and Devices
228

electrochemical reactions processing glucose or
oxygen (Ben Amar et al., 2015). Different approaches
to harvest energy using body fluids such as urine,
saliva (Göbel et al., 2016) or human perspiration (Jia et
al., 2013) have been proposed.
3 SPECIFIC CHALLENGES
REGARDING DEEP-TISSUE
IMPLANTS
The development towards in-body systems that are in
a more miniaturised scale enables implanting surgery
procedures to be less invasive and makes the implant
less impairing for the patient. According to Cadei et al.
(2014) the size limit typically is in the range of 1 cm³.
Active medical implants in deep-tissue body layers
present specific challenges regarding their design and
functionality in comparison to established implants
closer to the body surface, such as cardiac pacemakers
or neurostimulators. Since wires implanted in the
human body present a potential infection risk, wireless
communication and energy supply is desired.
3.1 Communication
In order to enable patients and health care professionals
to access physiological data or take technical control,
e.g. to activate the system’s actuators, a secure
communication path between the implant and a
corresponding out-body device needs to be established,
using the human body tissue as a transmission medium.
Teshome et al. (2019) present a comparison of
different wireless communication principles regarding
their transmission range. Accordingly, antenna based
radiofrequency (RF) signal communication enables by
far the widest in-body transmission range of up to 2 m,
while electric-field based and ultrasonic
communication enable much smaller ranges of up to 10
cm. Other presented technologies are too limited in the
transmission range to be taken into consideration for
communication with deep-tissue implants.
The transmission of tissue with RF signals has been
investigated in various works. Living body tissue
generally absorbs RF signals and is an inapplicable
channel to high-frequency electromagnetic waves.
(Teshome et al., 2019) Short- wave signals, such as the
in wireless body area networks (WBAN) widely used
2,36 – 2,4 GHz band appear to be not suitable to
communicate with deep-tissue implants as attenuation
values increase with frequency (Alomainy et al., 2006).
Therefore, longer-wave electromagnetic signals such
as the 400 MHz frequency band set aside in the
Medical Implant Communication Service (MICS)
specification especially for medical implants and
devices are proposed. Since the size of the RF-enabling
antenna is proportional to the wave length of the used
signal, shorter frequency signals, however, have the
downside of requiring larger antennas potentially using
more space of the implanted unit.
3.2 Energy Supply
Active implants not only feature a communication unit
but also a logic unit providing adequate computing
power as well as an actuator or sensor module, all of
which require a sufficient wireless energy supply.
Inductive coupling is a commonly used principle
enabling mutual inductance between a primary coil
integrated in an out-body station and a secondary coil
implanted in the human body. This method is limited
to short implantation depths as with transcutaneous
IMDs or implants closer to the body surface since the
power absorbed by human tissue has to be minimised
in order to prevent tissue heating or other side effects.
The size of the coils usually is in the range of several
cm as the coupled energy is dependent on the coil size,
which counteracts the requirement of a high
miniaturisation (Ben Amar et al., 2015).
Regarding the limitations associated with
conventional methods, energy harvesting is a
promising alternative to power deep-tissue medical
implants, provided that the required energy to power
the device is harvested in the body region the implant
is situated in.
To efficiently generate electrical energy from
thermal harvesting, there needs to be a sufficient
temperature difference between the hot and cold end of
the applied thermocouple since the output power
depends on the dimension of this temperature gradient
(Ben Amar et al., 2015; Cadei et al., 2014). Between
the human body and the environment there is only a
small gradient in the range of 3 – 5°C (Mateu et al.,
2014), but in deep-tissue layers body temperature
differences are even smaller making them too
insignificant to effectively harvest thermal energy, thus
only the harvesting of mechanical energy is taken into
consideration by the authors.
4 SYSTEM STRUCTURE AND
OPERATION
Due to the characteristics of human body energy
harvesting regarding the time periods power is
Architecture and Low Power Management of a Deep-tissue Medical Implant System Powered by Human Body Energy Harvesting
229
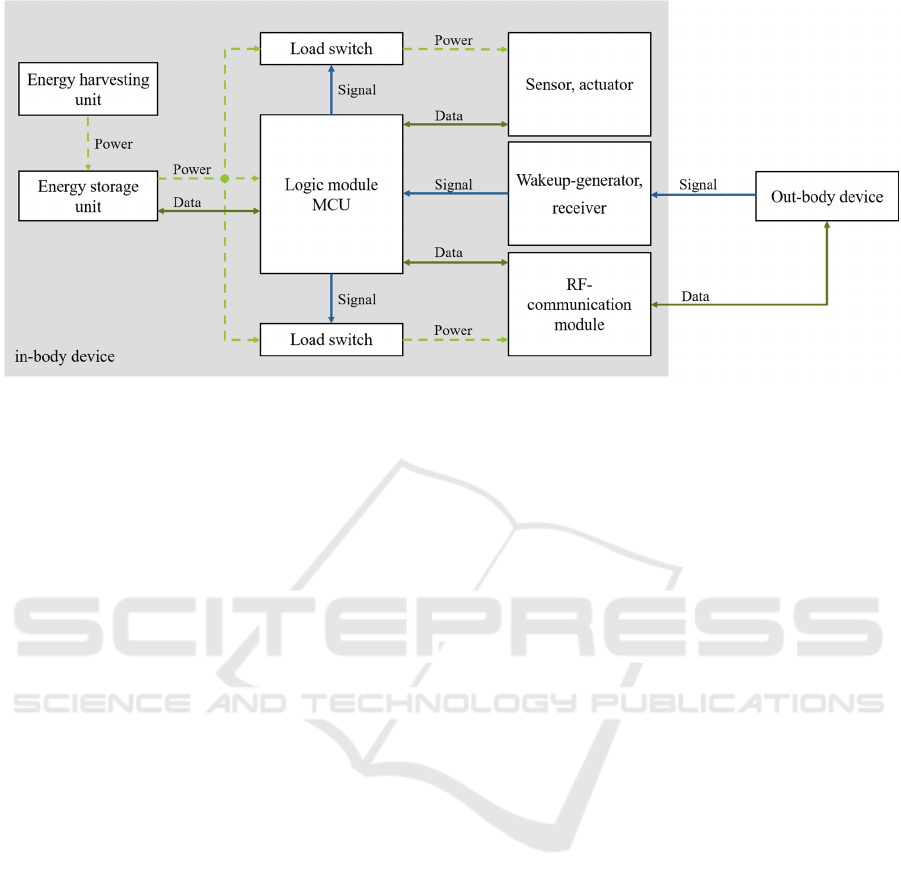
Figure 2: Architecture of the proposed system showing data, signal and power flow of the in-body and out-body device.
generated and the therefore required efficient power
management as well as the presented challenges of
communicating with devices situated in deep tissue,
suitable system architectures are required addressing
these issues.
The aim of the presented concept is to set up a
general system which can be applied for various
technical principles of energy harvesting, enable an
efficient power management of the limited amounts
of harvested energy and provide a communication
unit in order to establish a communication path
between the IMD and an out-body device when
requested by the patient or operator. Since the energy
source may only be temporarily available, the
architecture is designed to provide flexibility
regarding irregular time periods of the energy
harvesting process and the therefore strongly varying
load peaks.
As presented in Figure 2, the proposed system
consists of an energy harvesting unit, an accumulator
unit for energy storing and a logical unit regulating
the subsequent sensor or actuator and RF-
communication module. In order to reduce the
system’s energy consumption, load switches
regulated by the MCU are applied to turn on and turn
off the powering of the sensor or actuator and
communication unit. A wakeup-receiver is provided
to receive signals from an out-body device to wake up
the MCU from a power saving mode when system
operation is required by the patient or health care
professional. In the following the intended energy
storing unit and power management methods are
described in detail.
4.1 Energy Storage Unit
The accumulator unit of the circuit fulfils two
essential tasks: The storage of the electrical energy
generated by the harvesting module and the
subsequent supply of the required energy for the logic
module and actuator or sensor unit. The type of the
energy storage unit depends on the amount of energy
to be stored, the required power drain, the storage
duration and the target number of charge cycles.
The most commonly used type of energy storage
in IMDs are lithium ion batteries (LIB). They are
characterized by a high energy density and low self-
discharge which enables a long storage duration.
Since LIBs are solid state batteries, the mobility of the
charge carriers within the batteries is limited. This
also results in a limitation of the available power
density. Another disadvantage is the limited number
of charge cycles (Yu et al., 2018). Furthermore, the
degradation of the LIB capacity will be accelerated
when there are power peaks during the charging (Zou
et al., 2015).
As stated before, this approach also considers
energy generating methods harvesting from body
motions that are only temporarily executed by the
patient. In these cases, lithium-ion batteries are not
suitable as due to their low power density they cannot
absorb all energy provided and can even be damaged
by occuring power peaks (Zou et al., 2015). A
potential alternative to LIBs are ultracapacitors (UC).
Due to their very high power density compared to
LIBs, UCs can absorb almost the entire energy
generated during a short-term power peak (Burke,
2000). The better power drain compared to the solid
state LIB is based on a higher mobility of the charge
BIODEVICES 2020 - 13th International Conference on Biomedical Electronics and Devices
230
carriers. However, since the free movement of the
charge carriers results in a higher self-discharge, the
absorbed energy’s storing duration in the UC is
comparatively short. In the presented circuit concept,
the storage duration is desired to be as long as
possible, so using a UC alone is no ideal alternative
to a LIB.
One possibility to achieve all necessary
requirements for the energy storing unit is the use of
a hybrid energy storage system (HESS). A HESS
combines a classic LIB with a UC. The UC takes over
the short-term absorption of high power amounts and
protects the battery, while the latter can store the
energy for a longer period of time so that it can be
used later to power the downstream modules. HESSs
are already being used in electric vehicles with larger
scaling. For example, they make it possible to utilize
the short-term high power generated during
regenerative braking recuperation with a high degree
of efficiency and at the same time protect the
expensive lithium batteries in the car from peak
power. (Hochgraf et al., 2014; Hu et al., 2018)
There are two possible options for the realisation
of a HESS within the framework of the presented
circuit concept. In a simpler design, the HESS
consists of only one LIB and a parallel-connected UC.
This enables an implementation which is easy to
realise and allows for a high degree of
miniaturisation. (Lukic et al., 2007) Due to a lack of
interconnected control electronics, however, the
energy stored in the UC cannot be completely utilised
for power supply of the downstream units. (Gao et al.,
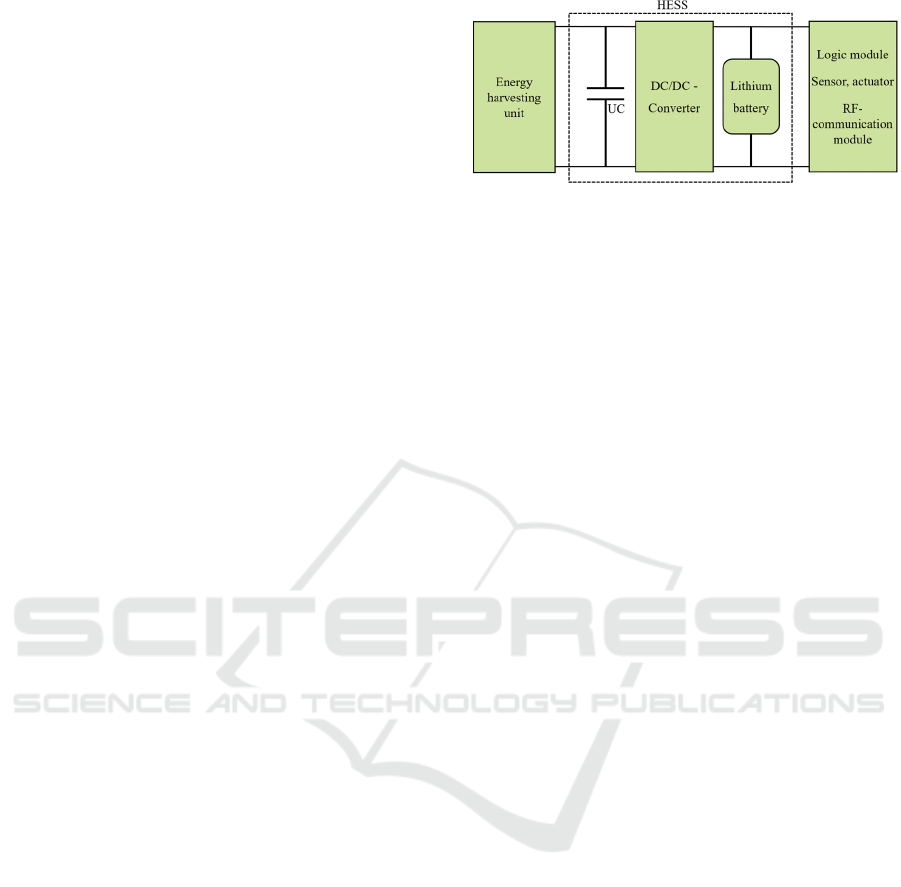
2005) Figure 3 shows a schematic of the second
option. A unidirectional DC/DC converter is
interconnected between the UC and a LIB. This
enables a higher degree of utilization of the energy
stored in the UC, but results in a larger space
requirement of the HESS, which must be taken into
account when designing and using the circuit.
(Lhomme et al., 2005)
The energy generated in the harvesting module is
temporarily stored in the UC and transferred to the
lithium battery via the DC/DC converter. From there,
the remaining circuit including the actuator module is
supplied with the previously generated energy.
Combined with a UC, the LIB is exposed to less stress
in terms of varying load peaks during the loading
process. As a result, the lifecycle of the LIB is
significantly prolonged which enables longer
implantation times in the body and thus leads to a
minimisation of invasive surgical interventions. (Cao
& Emadi, 2012)
Figure 3: Hybrid energy storage system with integrated
DC/DC-converter.
4.2 Power Management Methods
Since the amounts of output power are limited due to
the stated reasons, an efficient management of the
harvested energy is required to be able to unfailingly
power the logic module and actuator or sensor unit
according to the patient’s needs.
4.2.1 Hardware-enabled Management
Providing that the patient only temporarily
communicates with the IMD in order to operate the
actuator or access biological data stored, it is
determined which of the intended system’s modules
need to be operated only temporarily. As it is desired
to reduce leakage currents caused by passive
components, the sensor or actuator and
communication modules are only to be powered when
their operation is required. This is ensured by an
appropriate design of the hardware architecture as
presented in Figure 2. Load switches are applied as a
basic solution to turn on and turn off the powering of
the downstream modules, i.e. the sensor or actuator as
well as the RF-communication module.
4.2.2 MCU Wake-up Options
For many applications MCUs are designed in such a
way that they are able to enter different low-power or
sleep modes in order to reduce their energy
consumption. In these modes typically the
functionality of different components, such as the
CPU, memory or peripheral components, is reduced
at different levels or completely turned off.
In order to establish a communication path
between the out-body device and the RF-
communication module, the system is listening for
occurring external connecting requests (Figure 4). In
order to reduce the energy consumption, the MCU
can be set to turn into a low-power mode between
these high-energy consuming listening events. This
software-controlling of frequent listening events,
however, is connected to the downside that the CPU
Architecture and Low Power Management of a Deep-tissue Medical Implant System Powered by Human Body Energy Harvesting
231
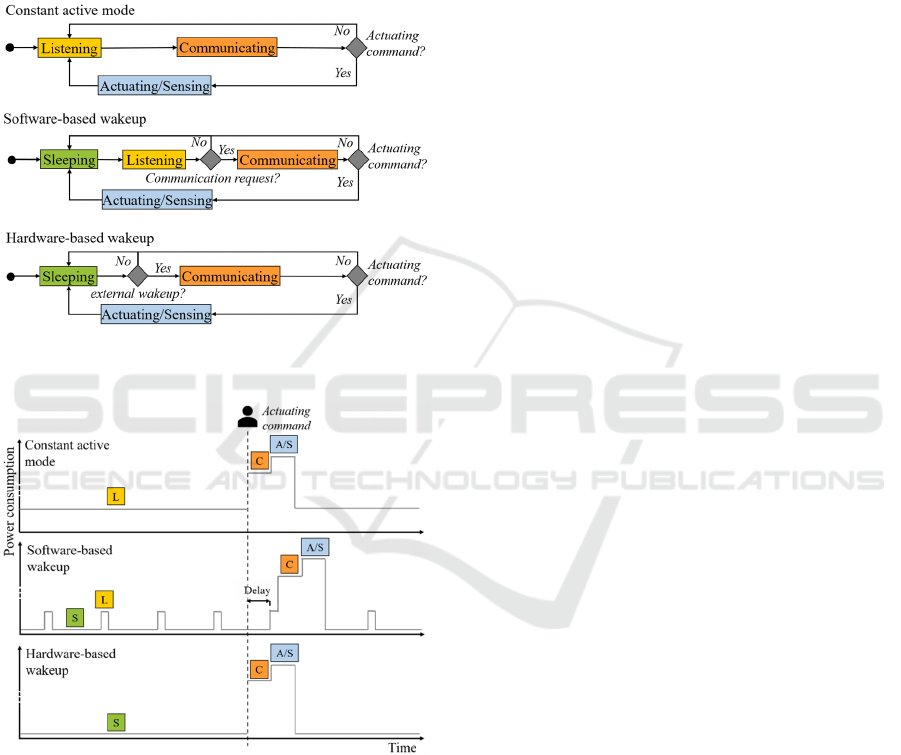
cannot be turned off which causes an increased
energy consumption even in low-power mode, as
shown in Figure 5.
In deep-sleep mode, however, the CPU is disabled
from executing commands. In order to return to active
mode in this case an interrupt needs to be set to be
triggered by an external energy pulse received by the
wakeup receiver (Figure 4). While this option is the
most power-saving as the system is only woken up
Figure 4: Hardware- and software-based wakeup operation
compared to constant active mode.
Figure 5: Power consumption levels of hardware- and
software-based wakeup operation compared to constant
active mode. (S) Sleeping, (L) Listening, (C) Communi-
cating, (A/S) Actuating/Sensing.
from sleep-mode when requested by the patient via
the out-body device (Figure 5), it presents the
challenge of wirelessly transmitting the interrupt
impulse through human body tissue. To solve this
issue, alternative concepts of power transfer in deep-
tissue layers, such as ultrasound wakeup, have to be
taken into consideration.
5 CONCLUSIONS
Since external approaches to wirelessly power IMDs
situated in deep-tissue layers underlay several
limitations, human body energy harvesting is a
promising powering approach. In this paper therefore
a system architecture for an IMD powered by
harvesting of mechanical energy is proposed. The
presented system provides great flexibility regarding
the used harvesting principle and enables the patient
to wirelessly communicate with the IMD according to
his needs. Since the harvested energy may not be
permanently available, hybrid storage systems
combining the advantages of conventional LIB and
UC are intended to be used for energy storage.
Research approaches have shown that these systems
not only are able to efficiently process load peaks, but
also offer great potential regarding the
miniaturisation of the system and prolonged
implantation times due to an extended number of
possible load cycles. Furthermore, suitable power
management options to be able to power an RF-
communication module for data exchange or
technical control by the patient as well as an actuator
or sensor unit are proposed. As a result, the energy
consumption of the IMD potentially can be
considerably reduced.
ACKNOWLEDGEMENTS
The research on deep-tissue implants presented in this
paper has received funding from the Bavarian
Ministry of Economic Affairs, Regional
Development and Energy within the framework of the
research transfer initiative “Medical Valley Award”.
REFERENCES
Alomainy, A., Hao, Y., Yuan, Y., & Liu, Y. (2006).
Modelling and Characterisation of Radio Propagation
from Wireless Implants at Different Frequencies. In
Proceedings of the 9th European Conference on
Wireless Technology: Manchester, UK, 1-12 September
2006 (pp. 119–122). IEEE. https://doi.org/10.1109/
ECWT.2006.280449
Ben Amar, A., Kouki, A. B., & Cao, H. (2015). Power
Approaches for Implantable Medical Devices. Sensors
(Basel, Switzerland), 15 (11),
28889–28914. https://doi.org/10.3390/s151128889
Böhm, R., Weindl, C., & Franke, J. (2018). Control of a
hybrid storage system (HSS) comprising a RedOx-
Flow Battery and a High Speed Flywheel for a hybrid
compensation system. In 2018 IEEE PES Innovative
BIODEVICES 2020 - 13th International Conference on Biomedical Electronics and Devices
232

Smart Grid Technologies Conference Europe (ISGT-
Europe): Sarajevo, Bosnia and Herzegovina, October
21-25, 2018 : conference proceedings (pp. 1–6). IEEE.
https://doi.org/10.1109/ISGTEurope.2018.8571631
Burke, A. (2000). Ultracapacitors: why, how, and where is
the technology. Journal of Power Sources, 91 (1), 37–
50. https://doi.org/10.1016/S0378-7753(00)00485-7
Cadei, A., Dionisi, A., Sardini, E., & Serpelloni, M. (2014).
Kinetic and thermal energy harvesters for implantable
medical devices and biomedical autonomous sensors.
Measurement Science and Technology, 25 (1), 12003.
https://doi.org/10.1088/0957-0233/25/1/012003
Cao, J., & Emadi, A. (2012). A New Battery/UltraCapacitor
Hybrid Energy Storage System for Electric, Hybrid,
and Plug-In Hybrid Electric Vehicles. IEEE
Transactions on Power Electronics, 27 (1), 122–132.
https://doi.org/10.1109/TPEL.2011.2151206
Cheng, Q., Peng, Z., Lin, J., Li, S., & Wang, F. (2015).
Energy harvesting from human motion for wearable
devices, 409–412.
https://doi.org/10.1109/NEMS.2015.7147455
Fu, H., Xu, R., Seto K., Yeatman E.M., & Kim S.G. (2015).
Energy Harvesting from Human Motion Using
Footstep-Induced Airflow. Journal of Physics:
Conference Series. Advance online publication.
https://doi.org/10.1088/1742-6596/660/1/012060
Gao, L., Dougal, R. A., & Liu, S. (2005). Power
Enhancement of an Actively Controlled
Battery/Ultracapacitor Hybrid. IEEE Transactions on
Power Electronics, 20 (1), 236–243. https://doi.org/
10.1109/TPEL.2004.839784
Göbel, G., Beltran, M. L., Mundhenk, J., Heinlein, T.,
Schneider, J., & Lisdat, F. (2016). Operation of a
carbon nanotube-based glucose/oxygen biofuel cell in
human body liquids—Performance factors and
characteristics. Electrochimica Acta, 218, 278–284.
https://doi.org/10.1016/j.electacta.2016.09.128
Hannan, M. A., Mutashar, S., Samad, S. A., & Hussain, A.
(2014). Energy harvesting for the implantable
biomedical devices: Issues and challenges. Biomedical
Engineering Online, 13, 79. https://doi.org/10.1186/
1475-925X-13-79
Hochgraf, C. G., Basco, J. K., Bohn, T. P., & Bloom, I.
(2014). Effect of ultracapacitor-modified PHEV
protocol on performance degradation in lithium-ion
cells. Journal of Power Sources, 246, 965–969.
https://doi.org/10.1016/j.jpowsour.2012.09.038
Hu, J., Jiang, X., Jia, M., & Zheng, Y. (2018). Energy
Management Strategy for the Hybrid Energy Storage
System of Pure Electric Vehicle Considering Traffic
Information. Applied Sciences, 8 (8), 1266.
https://doi.org/10.3390/app8081266
Jia, W., Valdés ‐ Ramírez, G., Bandodkar, A. J.,
Windmiller, J. R., & Wang, J. (2013). Epidermal
Biofuel Cells: Energy Harvesting from Human
Perspiration.
Angewandte Chemie International
Edition, 52 (28), 7233–7236.
https://doi.org/10.1002/anie.201302922
Johari, J., & Rashid, T. M. A. T. (2017). Optimization of
piezoelectric transducer placement in shoe insole for
energy harvesting. In E. a. S. E. International
Conference on Electrical (Ed.), 2017 International
Conference on Electrical, Electronics and System
Engineering (ICEESE): 9-10 Nov. 2017 (pp. 61–66).
IEEE. https://doi.org/10.1109/ICEESE.2017.8298406
Lhomme, W., Delarue, P., Barrade, P., Bouscayrol, A., &
Rufer, A. (2005). Design and control of a
supercapacitor storage system for traction applications.
In Conference record of the 2005 IEEE Industry
Applications Conference: Fortieth IAS Annual
Meeting, 2-6 October, 2005, Kowloon, Hong Kong
(pp. 2013–2020). IEEE.
https://doi.org/10.1109/IAS.2005.1518724
Lukic, S. M., Wirasingha, S. G., Rodriguez, F., Cao, J., &
Emadi, A. (2007). Power Management of an
Ultracapacitor/Battery Hybrid Energy Storage System
in an HEV. In O. Wilde (Ed.), 2006 ieee vehicle power
and propulsion conference (pp. 1–6). John Wiley.
https://doi.org/10.1109/VPPC.2006.364357
Mateu, L., Dräger, T., Mayordomo, I., & Pollak, M. (2014).
Energy Harvesting at the Human Body. In Wearable
sensors: Fundamentals, implementation and
applications (pp. 235–298). Academic Press.
https://doi.org/10.1016/B978-0-12-418662-0.00004-0
Niroomand, M., & Foroughi, H. R. (2016). A rotary
electromagnetic microgenerator for energy harvesting
from human motions. Journal of Applied Research and
Technology, 14 (4), 259–267.
https://doi.org/10.1016/j.jart.2016.06.002
Paulo J., & Gaspar P.D. (2010). Review and Future Trend
of Energy Harvesting Methods for Portable Medical
Devices. Proceedings of the World Congress on
Engineering 2010 Vol II.
Pfenniger, A., Vogel, R., Koch, V. M., & Jonsson, M.
(2014). Performance analysis of a miniature turbine
generator for intracorporeal energy harvesting.
Artificial Organs, 38 (5), E68-81.
https://doi.org/10.1111/aor.12279
Saida, M., Zaibi, G., Samet, M., & Kachouri, A. (2018).
Design and Study of Piezoelectric Energy Harvesting
Cantilever from Human Body. In SSD '18: The 15th
International Multi-Conference on Systems, Signals &
Devices : program of the Multi-Conference on Systems,
Signals & Devices : SSD 2018 : March 19-22, 2018,
Hammamet, Tunisia (pp. 164–168). IEEE.
https://doi.org/10.1109/SSD.2018.8570616
Shi, Y., Wang, Y., Mei, D., & Chen, Z. (2018). Wearable
Thermoelectric Generator With Copper Foam as the
Heat Sink for Body Heat Harvesting. IEEE Access, 6,
43602–43611.
https://doi.org/10.1109/ACCESS.2018.2863018
Strasser, M., Aigner, R., Lauterbach, C., Sturm, T. F.,
Franosh, M., & Wachutka, G. (2003). Micromachined
CMOS thermoelectric generators as on-chip power
supply. In Transducers'03: The 12th International
Conference on Solid-State Sensors, Actuators and
Microsystems : digest of technical papers : [June 9-12,
2003], Boston
(pp. 45–48). IEEE. https://doi.org/
10.1109/SENSOR.2003.1215249
Architecture and Low Power Management of a Deep-tissue Medical Implant System Powered by Human Body Energy Harvesting
233

Teshome, A. K., Kibret, B., & Lai, D. T. H. (2019). A
Review of Implant Communication Technology in
WBAN: Progress and Challenges. IEEE Reviews in
Biomedical Engineering, 12, 88–99.
https://doi.org/10.1109/RBME.2018.2848228
Wang Z., Leonov V., Fiorini P., & Hoof C.V. (2009).
Realization of a wearable miniaturized thermoelectric
generator for human body applications. Sensors and
Actuators A: Physical, 156 (1), 95–102.
https://doi.org/10.1016/j.sna.2009.02.028
Yu, H., Castelli-Dezza, F., & Cheli, F. (2018, January 19).
Multi-objective Optimal Sizing and Energy
Management of Hybrid Energy Storage System for
Electric Vehicles. http://arxiv.org/pdf/1801.07183v2
Zhang, H., Zhang, X.-S., Cheng, X., Liu, Y., Han, M., Xue,
X., Wang, S., Yang, F., A S, S., Zhang, H., & Xu, Z.
(2015). A flexible and implantable piezoelectric
generator harvesting energy from the pulsation of
ascending aorta: in vitro and in vivo studies. Nano
Energy, 12, 296–304. https://doi.org/10.1016/
j.nanoen.2014.12.038
Zou, Y., Hu, X., Ma, H., & Li, S. E. (2015). Combined State
of Charge and State of Health estimation over lithium-
ion battery cell cycle lifespan for electric vehicles.
Journal of Power Sources, 273, 793–803.
https://doi.org/10.1016/j.jpowsour.2014.09.146
BIODEVICES 2020 - 13th International Conference on Biomedical Electronics and Devices
234
