
ACOUSTIC THERMOAGITATION BASED ON PIEZOELECTRIC
β-PVDF POLYMER FILMS
Potential Evaluation in Lab-on-a-Chip Applications
V. F. Cardoso,
G. Minas
University of Minho, Department of Industrial Electronics, Campus de Azurém, 4800-058, Guimarães, Portugal
P. Martins, J. Serrado Nunes,
L. Rebouta, S. Lanceros-Méndez
University of Minho, Department of Physics, Campus de Gualtar, 4710-057, Braga, Portugal
G. Botelho
University of Minho, Department of Chemistry, Campus de Gualtar, 4710-057, Braga, Portugal
Keywords: Lab-on-a-chip, PVDF, Acoustic thermoagitation.
Abstract: This paper describes a lab-on-a-chip device with acoustic thermoagitation based on a piezoelectric β-PVDF
polymer. The device is used for testing and monitoring biochemical parameters in biological fluids using
optical absorption spectrophotometry. Experimental results regarding the influence of the electrical signal
amplitude and frequency applied for the generation of acoustic thermoagitation is presented. The individual
contribution of the heating and the microagitation provided by the actuation of the piezoelectric film for the
fluids mixture was determined. The paper is completed with a study of the β-PVDF degradation with
transparent conductive electrodes, ITO and AZO, when placed in contact with uric acid fluids. The final
goal of using this technique is the improvement of mixing and reaction time without interfering with
biochemical reactions and analytical measurements.
1 INTRODUCTION
For the development of a lab-on-a-chip device for
fluidic analysis, the microfluidics technologies have
been a vital tool allowing the fabrication of precise
and small structures. The huge interest in these
technologies is inherent to the performance
achieved: reduction on the sample quantity, high
integration and consequently high potential for
fluids automation and control in sub-microliter
volumes, decrease of response time, reduction of
chemical quantities stored and a large reduction in
total costs (Auroux et. al, 2002).
Lab-on-a-chip devices can perform several
analyses simultaneously. In order to get in-loco,
quick and reliable results, they need an automatic
system for microfluids control, which can cover all
the steps of a chemical or biological process.
Usually, a bio(chemical) process needs the
mixture of fluids. For that, devices based in MEMS
(Micro Electro Mechanical Systems) such as
micropumps (Reyes et. al, 2002) and microvalves
(Rife et. al, 2000) are used, but they increase the
device cost, need complex control systems and their
integration is complex. The mixture of the fluids
using just diffusion avoids these disadvantages.
However, when large molecules with small
diffusivities take part in the reaction, it is needed
high transit times of the molecules on the channels
and consequently long channels (Ottino et. al, 2004).
To overcome this high transit times, it is necessary
to develop alternative methods for improving
mixture.
The use of acoustic waves is an interesting
solution and it is one of the main issues of this
present work. The acoustics waves traveling inside
the fluid create differential pressures and induce the
so called acoustic propagation. For occurring the
394
F. Cardoso V., Minas G., Martins P., Serrado Nunes J., Rebouta L., Lanceros-Méndez S. and Botelho G. (2009).
ACOUSTIC THERMOAGITATION BASED ON PIEZOELECTRIC β-PVDF POLYMER FILMS - Potential Evaluation in Lab-on-a-Chip Applications.
In Proceedings of the International Conference on Biomedical Electronics and Devices, pages 394-397
DOI: 10.5220/0001546903940397
Copyright
c
SciTePress
acoustic microagitation of the fluids, the reaction
chamber must be coated with an electroactive
polymer. One example is the β-PVDF (Lanceros-
Mendez et. al, 2006), which the use for fluid
microagitation purposes is the main innovation of
this work. Applying an a.c. voltage to the contacts of
the piezoelectric β-PVDF film, it is produce
mechanical oscillations promoting the movement,
mixture and reaction of the fluids, as well the heat
generated by this technique, the so called acoustic
thermoagitation.
2 DEVICE DESCRIPTION
This paper describes the incorporation and
validation of an acoustic thermoagitator based on a
piezoelectric β-PVDF polymer in a fully-integrated
disposable lab-on-a-chip for point of care testing and
monitoring of biochemical parameters in biological
fluids. This lab-on-a-chip has interesting
characteristics such as portability, low-cost and
disposability. Furthermore, it has a completely
automatic operation and uses optical absorption
spectrophotometry as analytical measurement
technique.
2.1 Biosystem Operation
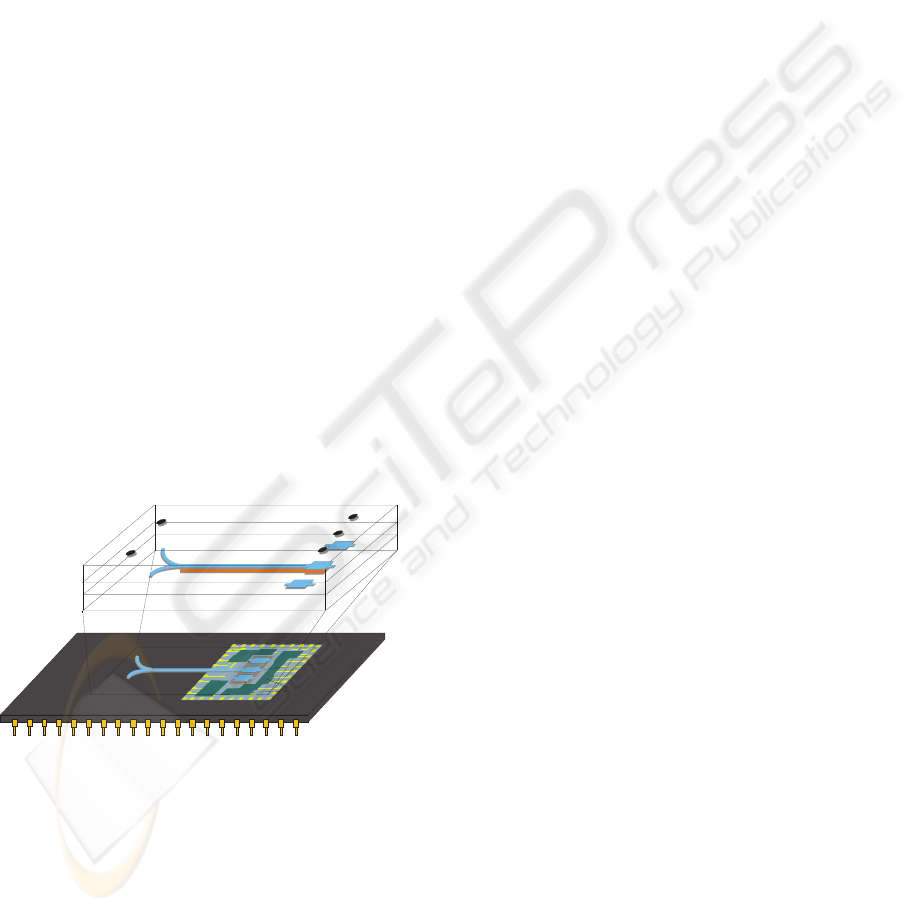
The lab-on-a-chip is composed by two dies: the
fluidic die and the detection die (Figure 1).
Figure 1: Schematic representation of the lab-on-a-chip
structure with the β-PVDF deposited underneath the
microfluidic structure.
The microfluidic die, fabricated in SU-8,
includes the microchannels and the reaction
chambers. Three reaction chambers are needed for
each analysis. One with a well-known concentration
sample of the biochemical parameter that is being
analysed, is used for the calibration of the device.
Other, for the mixture of the sample and the reagent
is needed, to perform the analysis of the coloured
mixed solution. Finally, a third one is used for the
chemical reagent in order to obtain the baseline
reference. Underneath the reaction chambers was
deposited the piezoelectric β-PVDF polymer with its
corresponding electrodes.
The detection die includes the photodetectors and
the electronic components for signal actuation and
detection, all fabricated in CMOS technology.
Above the photodetectors, there are several high-
selective band-pass optical filters, deposited by Ion
Beam Deposition, that select the wavelength
according to the several biomolecules into analysis.
This optical filtering system allows the use of non-
calibrated external polychromatic light source.
2.2 Analytical Measurement Technique
Among the several analytical techniques available in
laboratories of clinical analysis, the
spectrophotometry is the most used. However, this
technique cannot be used directly since a high
number of biomolecules for clinical analysis do not
have chromophores which absorb the light in the
visible spectra. In order to overcome this limitation,
several specific chemical reactions allow to
transform these biomolecules in colored products
which absorbency is within the visible light spectra -
colorimetric reactions.
In the ideal colorimetric analysis, the mixture
coloration intensity is proportional to the
concentration of the biochemical parameter and can
be quantified measuring the optical absorption of the
mixture (reagent + biomolecule) at a specific
wavelength (Thomas, 1999).
2.3 Acoustic Streaming
The acoustic streaming is a steady flow generated by
the propagation of acoustic waves in a viscous fluid.
It arises from the transfer of momentum and energy
of the acoustic field to the medium, through its
acoustic attenuation.
Acoustic streaming offers several distinct
advantages for application in microfluidic devices
(Frampton et. al, 2004). In this work, the acoustic
streaming is due to the absorption of the acoustic
energy in the fluid itself. This absorption results in a
radiation pressure in the direction of the acoustic
propagation and is termed by quartz wind.
Quartz wind velocity and effective pressure are
limited by the heating and cavitation tolerance. A
small fraction of the incident acoustic energy goes
into kinetic energy of the fluid and the rest is
transformed into heat. In this way, the acoustic
thermoagitation generated by this effect becomes
Chip
Optical filters and
Photodetectors
Electronics
β−PVDF
Reagent
Mixture
Calibrator
Reagent
Sample
Glass
Glass
SU-8
ACOUSTIC THERMOAGITATION BASED ON PIEZOELECTRIC ß-PVDF POLYMER FILMS - Potential Evaluation
in Lab-on-a-Chip Applications
395
advantageous, once in some applications or
reactions, the increase of temperature is beneficial in
order to reduce the mixture time (Rife et. al, 2000).
2.4 Poly(vinylidene fluoride)
Nowadays, with the rapid development of polymer
processing methods and synthetic technology, the
industrial materials used in various manufacturing
fields are being substituted by polymer substances,
which have better properties than those of materials
previously used in many industrial areas. One of the
polymer that has received increased attention is the
poly(vinylidene fluoride), also known as PVDF. It
shows an unusual polymorphism in this class of
materials, showing four different crystalline phases.
From the technology point of view, the β-phase is
the one which shows better properties to be applied
in sensors, actuators and transducers, due to its
higher piezo- piro- and ferroelectric properties.
Moreover, it shows an excellent combination of
processability, chemical agent resistance, lightness,
moldability and low-cost production. While ceramic
materials break easily and have hard and dense
structures, PVDF is flexible, has a low density and
can be easily produced into thin-films (Brown,
1992). This polymer also shows low acoustic and
mechanical impedance (Foster, 2000) crucial for
generating the acoustic waves that produce the
thermoagitation of the fluids, as is the purpose of
this study. Another interesting feature of the PVDF
is its transparency. Indeed, in the case of this
application, the analytical measurement by
spectrophotometry requires that the PVDF and the
corresponding conductive electrodes, deposited in
the reaction chamber, are transparent to visible light.
3 EXPERIMENTAL RESULTS
3.1 Acoustic Thermoagitation
The quantitative evaluation of the mixing process
was carried out using the Far Diagnostic kit and
standards of urine with 5 mg/dl of uric acid
concentration. The reagent reacts with the sample of
urine containing uric acid in a 40:1 ratio with a
maximum absorption at 550 nm.
The acoustic thermoagitation was studied for
various frequencies and amplitudes of the electrical
signal applied to the electrical contacts of a 110 μm
thick β-PVDF film. The system was calibrated and
the reaction was studied up to a maximum time of
20 minutes, which is the time of the complete
mixture without thermoagitation.
The thermoagitation was performed powering
the β-PVDF, with a sinusoidal signal of 10 V
amplitude at different frequencies. After, a 10 MHz
frequency sinusoidal signal with various amplitudes
was applied. They results were processed to obtain
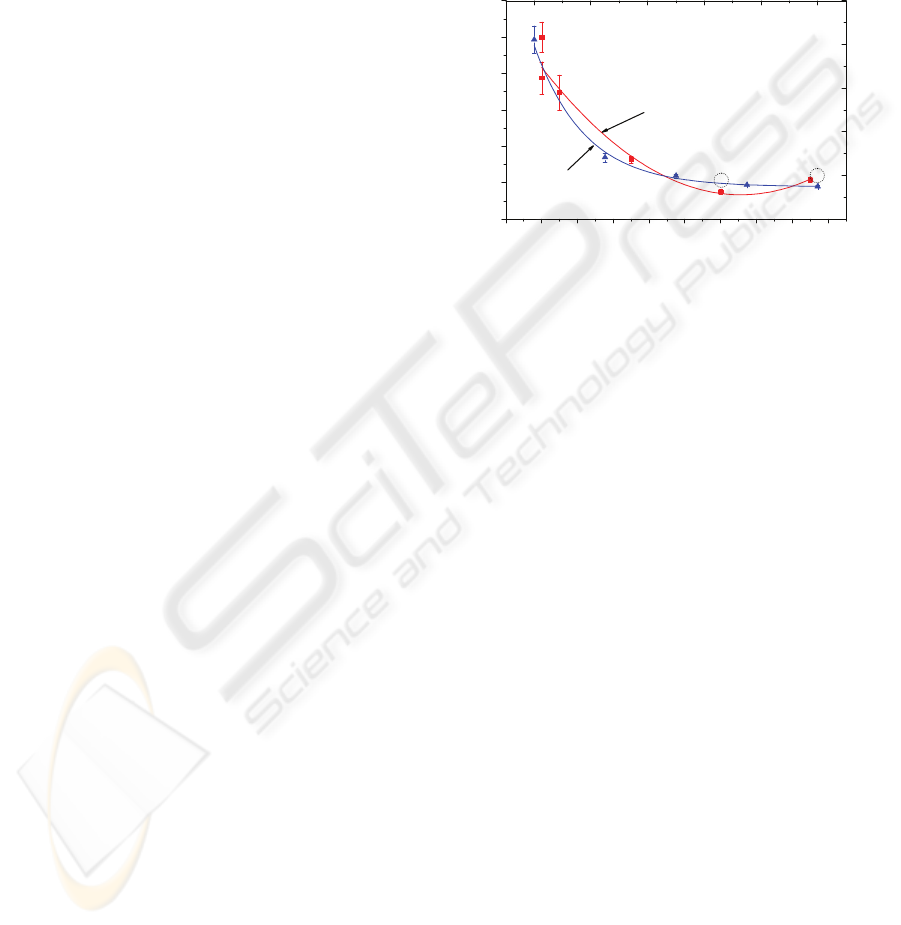
the results shown in Figure 2.
-2000 0 2000 4000 6000 8000 10000 12000 14000 16000
200
400
600
800
1000
1200
1400
Frequency signal
Mixture Time (s)
Frequency signal (kHz)
0246810
Amplitude signal
Amplitude signal (V)
Figure 2: Mixing time as a function of the frequency and
amplitude of the signal applied to the electrical contacts of
a 110 μm thick β-PVDF film.
It can be observed that with the application of
thermoagitation, the mixing occur in a faster way,
being the time necessary to obtain the complete
mixing, at 10 V amplitude and 10 MHz, only two
sevenths (351 s) of the complete mixing time
without thermoagitation (1200 s). In this way, the
resonance frequency of the used β-PVDF film
(10 MHz) is the most efficient for the generation of
thermoagitation. This result is consistent with the
theory (Eiras, 2007). On the other hand, the mixing
time decreases as the signal amplitude increases.
According to previous results, it was determined
the individual contribution of the agitation and the
heating generated by the use of this acoustic
thermoagitation technique. For that, the temperature
profile of the sample was measured applying an
electrical signal of 10 V amplitude and 10 MHz to
the β-PVDF film. Then, the sample was heated with
the same temperature profile obtained before with
the thermoagitation, using a temperature controller
(SHIMADZU TCC-260). It was observed that, for
the two sevenths in terms of gains achieved by the
application of the acoustic thermoagitation
technique, three-fifths are due to heating and two
fifths to agitation.
3.2 Transducer Degradation
As the first application of the present lab-on-a- chip
will be for measurement of uric acid concentrations,
BIODEVICES 2009 - International Conference on Biomedical Electronics and Devices
396
a degradation study (using a high-performance
liquid chromatograph model 875 from Jasco) of the
110 μm β-PVDF film with ITO (Indium Tin Oxide)
and AZO (Aluminium doped Zinc Oxide)
electrodes, when placed in contact with the
biological fluids, was performed. The study was
done with the Far Diagnostic kit of uric acid,
described previously. The chemical reaction was
performed with and without acoustic
thermoagitation during 20 minutes. The
thermoagitation was set using an electrical signal at
10 V amplitude and 10 MHz. The results shown in
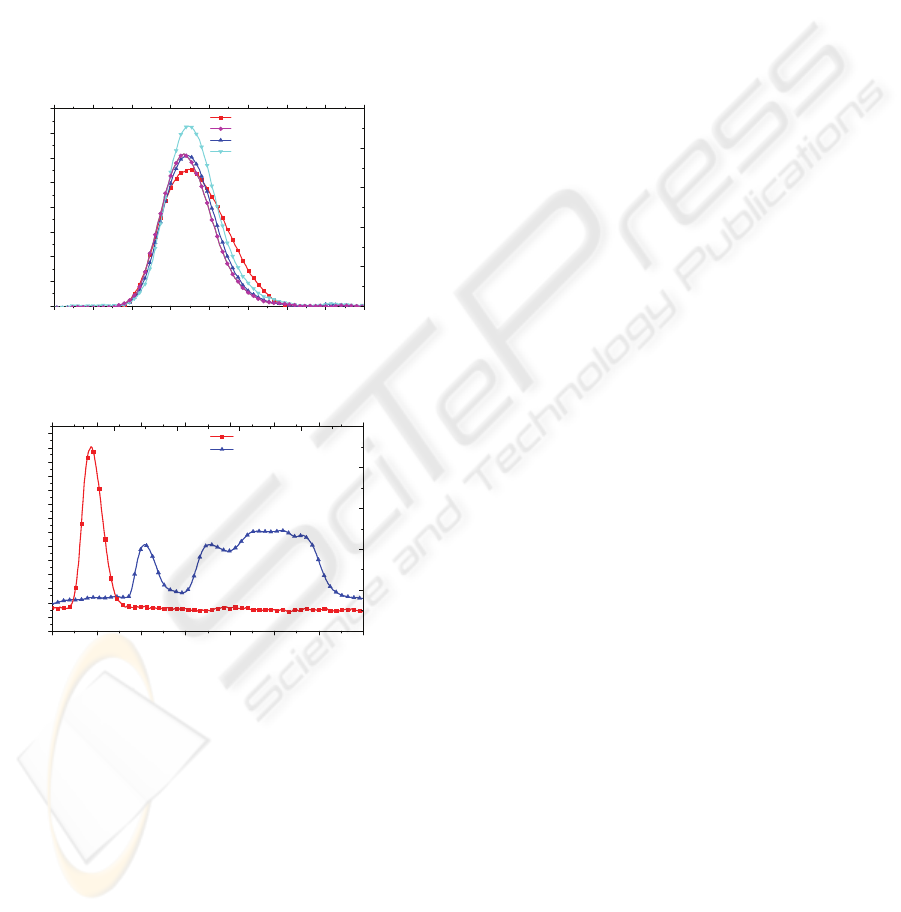
Figures 3 and 4 are representative for not degraded
and degraded electrodes, respectively.
2.00 2.05 2.10 2.15 2.20 2.25 2.30 2.35 2.40
0
2
4
6
8
10
12
14
16
Intensity (a.u.)
Time (s)
Allantoin
AZO without thermoagitation
ITO without thermoagitation
ITO with thermoagitation
Figure 3: Chromatogram of the samples that did not
suffered degradation with a reaction time of 20 min.
2.0 2.2 2.4 2.6 2.8 3.0 3.2 3.4
-2
-1
0
1
2
3
4
5
6
7
8
9
10
11
12
Allantoin
AZO with thermoagitation
Intensity (a.u.)
Time
(
s
)
Figure 4: Chromatogram of the samples that suffered
degradation with the reaction time of 20 min.
It can be observed that the β-PVDF films with
ITO electrodes did not influence the analyses results.
The opposite happens when the electrodes of the
β-PVDF film include aluminium, even in small
concentrations, like the transparent conductive AZO.
4 CONCLUSIONS
The application of acoustic thermoagitation through
the β-PVDF piezoelectric polymer is gainful when
fluids need to be mixed in a microfluidic device. It
reduces the mixing time resulting in a fast, complete
and homogeneous reaction of the reactants,
improving the global performance of the analysis
that is being performed.
With the optimization tests, it was proved that
better mixing results were obtained with the
electrical signal for actuation at the resonance
frequency of the film and with an amplitude of 10V.
Moreover, it was demonstrated that the improvement
of the mixture is mainly due to the heating of the
solution.
In the degradation study, it was verified that, for
all electrodes, the ITO is the one which does not
degrade in contact with the uric acid kit.
ACKNOWLEDGEMENTS
Work supported by the Portuguese Science
Foundation (grants PTDC/BIO/70017/2006,
PTDC/CTM/69362/2006).
REFERENCES
Auroux, P., Iossifidis, D., Reyes, D. R., Manz, A., 2002,
Micro Total Analysis Systems: Analytical Standard
Operations and Applications. Anal. Chem., 74, p.
2637-2652.
Reyes, D. R., Iossifidis, D., Auroux, P., Manz, A., 2002,
Micro Total Analysis Systems. 1. Introduction,
Theory, and Technology. Anal. Chem., 74, p. 2623-
2636.
Rife, J. C., et. al., 2000, Miniature Valveless Ultrasonic
Pumps and Mixers. Sensors and Actuators B., 86, p.
135-140.
Ottino, J. M., Wiggins, S., 2004, Introduction: Mixing in
Microfluidics. Phil. Trans. R. Soc. Lond. A. 362, p.
923-935.
Lanceros-Mendez, S., Sencadas, V., Gregorio Filho, R.,
Portuguese patent n.º 103318.
Thomas, M., 1999, Ultraviolet and visible spectroscopy.
Analytical Chemistry by Open Learning, p. 2-47.
Frampton, K. D., Minor, K., Martin, S., 2004, Acoustic
streaming in micro-scale cylindrical channels. Applied
Acoustic, Vol. 65, p. 1121-1129.
Brown, L. F., 1992, Ferroelectric Polymers: current and
future ultrasound applications. In IEEE Ultrason.
Symp. Proc. p. 539-550.
Foster, F. S., 2000, A history of medical and biological
imaging with polyvinylidene fluoride (PVDF
transducers. In IEEE Transactions on Ultrasonics,
Ferroelectrics and Frequency Control. Vol. 47 Nº 6.
Eiras, J. A., 2007, Piezoelectric Materials. Departamento
de Física, Universidade Federal de São Carlos, Brasil.
ACOUSTIC THERMOAGITATION BASED ON PIEZOELECTRIC ß-PVDF POLYMER FILMS - Potential Evaluation
in Lab-on-a-Chip Applications
397
