
Spontaneous Pupillary Oscillation Signal Analysis Applying Hilbert
Huang Transform
Fabiola M. Villalobos-Castaldi
1
, José Ruiz-Pinales
2
, Nicolás C. Kemper-Valverde
1
,
Mercedes Flores-Flores
3
, Laura G. Ramírez-Sánchez
1
and Metztli G. Ortiz-Hernández
1
1
Centro de Ciencias Aplicadas y Desarrollo Tecnológico, UNAM, Expert Systems Lab,
Circuito Exterior S/N C.P. 04510, Ciudad Universitaria, Ciudad de México, D.F., Mexico
2
Electronics Engineering Department, Engineering Division, Universidad de Guanajuato
Carretera Salamanca - Valle de Santiago Km. 3.5 + 1.8. Comunidad de Palo Blanco Salamanca,
Leon, Gto., C.P. 36885, Mexico
3
Tecnológico de Estudios Superiores de Ecatepec, Av. Tecnológico S/N, Valle de Anahuac, C.P. 55210,
Ecatepec de Morelos, Mexico
Keywords: Time-frequency Analysis, Hilbert Huang Transform, Spontaneous Pupillary Oscillation, Non-traditional
Time-series Characterization Scheme.
Abstract: This paper proposes a new application of the Hilbert-Huang transform (HHT). Pupillogram recordings were
analyzed through the non-traditional HHT to investigate patterns in the time-frequency parameters of
Spontaneous Pupillary Oscillation (SPO) signals. The traditional Fourier transform is only useful for linear
stationary signals analysis, but the HHT was designed for the analysis of non-linear and non-stationary
signals. However, the HHT is a more suitable tool to study SPO signals which are fundamentally non-
stationary. The intrinsic properties of the Spontaneous Pupillary Oscillation signals were highlighted by the
HHT scheme and the results showed that SPO waves present local and intermittent variations through the
time span. The numerical parameters demonstrated that it is a wide inter-subject variation in the intrinsic time-
frequency parameters contribution from each yielding mode to the total signal content.
1 INTRODUCTION
This study proves the assumption that spontaneous
pupillary fluctuation is stationary can be a serious
misconception. Complex interactions exist between
the sympathetic and para-sympathetic systems which
are two components of the ANS autonomic nervous
system acting as a balance between competing neural
mechanisms. Using selected stimulations, the
dynamics of this balance mechanism could be
significantly altered. Such modifications can be
studied using relevant autonomic indices (De Souza,
et al., 2007).
The iris is a vascular structure and changes in
pupil size are controlled by two smooth muscles in the
iris (Sylvain and Brisson, 2014). The sphincter
pupillae located in the stromal layer is under
cholinergic control, mediated via para-sympathetic
nerves from the Edinger-Westphal nucleus. The
dilated pupillae situated posterior to the constrictor
muscle is innervated by adrenergic fibers originating
in the superior sympathetic ganglion. This set of
opposing muscles exercise a fine but extensive
control over the pupil (Newman, 2008).
The constriction and dilatation of the pupillary
aperture is produced mainly through ANS control
exerted on the muscles of the iris. More specifically,
neurons of the PSN innervate circular fibers of the
iris, causing pupillary constriction, whereas
excitation by SNS neurons causes the radial fibers of
the iris to produce dilation of the pupil (Lowenstein,
1950). Pupil diameter can range from 1.5 to more than
9 millimeters in humans, and a little time, just about
0.2 seconds, it is required by the muscles of the iris to
react to stimulation (Goldwater, 1972). The ANS in
its role of maintaining homeostasis is in a state of
constant fluctuation (Malpas, 2010), (Hreidarsson
and Gundersen, 1988). This fluctuation, expressed by
spontaneous variations in the rhythmic changes in
pupil-size of around 1% occurs with heart beats and
Villalobos-Castaldi, F., Ruiz-Pinales, J., Kemper-Valverde, N., Flores-Flores, M., Ramírez-Sánchez, L. and Ortiz-Hernández, M.
Spontaneous Pupillary Oscillation Signal Analysis Applying Hilbert Huang Transform.
DOI: 10.5220/0005697400670077
In Proceedings of the 9th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2016) - Volume 4: BIOSIGNALS, pages 67-77
ISBN: 978-989-758-170-0
Copyright
c
2016 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
67

breathing due to fluctuations in blood pressure
(Warga, et al, 2009). The variance of normal pupil
size is very large. When a constant light level is
maintained, the pupil is seen to exhibit continual
fluctuations called Spontaneous pupillary
fluctuations (SPF) or pupil unrest or physiological
hippus. These variations in pupil size is a
characteristic of the regulatory mechanism and it is
often said that it shows a state of pupillary unrest.
This is a physiologic phenomenon that represents a
dynamic equilibrium of pupil size of the sympathetic
and para-sympathetic activity modulated by the
central nervous system is responsible (Newman,
2008), (Nowak et al., 2008). A typical recording of
pupil size in a constant light source shows marked
spontaneous activity with an irregular pattern of a
periodic oscillation. Unrest amplitudes are highest for
medium pupil sizes (Rosenberg and Kroll, 1999).
2 RELATED WORKS
In the nighties (McLaren et al., 1992) the authors
developed digital filtering techniques and Fourier
analysis to calculate several parameters designed to
report hippus and miosis. These techniques provided
a quantitative manner to evaluate pupillograms to be
used in the assessment of alertness. The first
parameter designed to report hippus was derived from
the Fourier-transformed pupillogram and was the area
of the frequency spectrum in a selected band of
frequencies. A second parameter to report hippus was
also investigated. Spectra differ the most at low
frequencies. At higher frequencies, differences were
seen to diminish.
A fast Fourier transformation was carried out as a
vigilance objective test for frequencies from 0.0 to 0.8
Hz in (Lüdtke et al., 1998) with the purpose of
detecting fatigued waves, i.e., slow pupillary
oscillations. For temporal changes analysis in the
frequency domain of pupillary oscillation two
parameters were extracted. One parameter based on
the FFT regarded only frequencies below 0.8 Hz,
neglecting fast pupillary changes (>1.5625 Hz). An
additional parameter referring to the tendency for
pupil instability, the pupillary unrest index (PUI), was
defined by cumulative changes in pupil size based on
mean values of consecutive data sequences. The
power and PUI were compared using the Mann–
Whitney U-test. Both parameters showed significant
differences between the two groups. The main
differences between an alert and a sleepy group of
people in power and PUI demonstrated the usefulness
of this method to objectively detect and quantify
sleepiness.
The study purpose reported in (Calcagnini et al,
2000
) was to assess whether and the extent LF and
HF rhythms contribute to spontaneous pupil diameter
fluctuations in rest and during sympathetic activation.
To investigate the statistical properties of the SPDF,
a parametric spectral and cross-spectral estimation
was used. The spectral coherences were used to
quantify the statistical link among rhythms in
different signals. A rhythmic respiratory component
(HF) was clearly found at 0.25 Hz in the pupillogram
spectrum in all the subjects. Cross spectral analysis
showed a significant coherence in this band between
pupil and respiration, pupil and tachogram, and pupil
and systogram (Cerutti, 2000).
In conclusion the analysis of the SPF showed the
contribution of two specific harmonic components,
which have been found to correspond to the well-
known LF and HF rhythms of the Heart Rate and
Blood Pressure variability signals (Cerutti, 1997).
Additionally, the authors concluded that the
apparently stochastic behavior of the spontaneous
PDF hides specific harmonic components reflecting
autonomic activity (Cerutti, 1999).
In the two thousands (Nowak et al, 2008) the
authors developed a new method of variability
description for the Spontaneous Pupillary Fluctuation
(SPF) signals based on the time-frequency analysis by
studying the variability of the SPF signal spectrum.
The application of fast pupillometry for recording the
SPF permitted expanding the analyzed frequency
band to 20 Hz. The proposed method of analysis and
the introduced measures of SPF variability helped
them in the detection and quantitative description of
short-lasting time-frequency and time-amplitude
variations that were obscured by the overall spectral
analysis (Elsenbruch, 2000). The authors mentioned
that the Fourier Transform (which requires a
stationary signal and is commonly used in the spectral
analysis of SPF), has limitations in signal testing. It
was concluded that the SPF signal is non-stationary,
i.e. its spectrum is varying in time. Thus, it was
clearly demonstrated that the previous assumptions
are no longer valid (Longtin, 1989).
The previous belief was that fluctuations in pupil
diameter arose from random processes, but new
results from chaotic time series has shown that they
may also arise from deterministic systems
(Rosenberg and Kroll, 1999). The accumulated
evidence supports the notion that the dynamics of
pupil size are governed by deterministic chaos rather
than a linear or stochastic process, and this has been
demonstrated by analyzing the pupil size versus time
from six subjects using pupillography and nonlinear
BIOSIGNALS 2016 - 9th International Conference on Bio-inspired Systems and Signal Processing
68

techniques (Rosenberg and Kroll, 1999). The
examined values of the correlation dimension, the
Hurst exponent, the flat power spectra, the values of
the Lyapunov exponent, and phase plane analyses
indicate a chaotic system as the origin of the
phenomenon. Pupil activity reported showed
repetitive complex patterns which could be explained
by a chaos rather than a random system (Rosenberg
and Kroll, 1999).
As previously mentioned, the SPF signal analysis
has been used for monitoring the level of alertness in
clinical conditions, diagnosing sleep disorders,
assessing how the different rhythms of the SPS
contribute to spontaneous pupil diameter fluctuations
at rest and during sympathetic activation, and also for
assessing the efficacy of therapeutic interventions
(Boyina et al., 2012), (Naber et al., 2013), (Jain et al.,
2011), (Heaver, 2012), (Calcagnini et al., 2000
),
(Bouma and Baghuis, 1971), (Pedrotti et al., 2014),
(Regen et al., 2013) and (Lüdtke et al., 1998). These
analyses have generally been performed by spectral
indices computed from standard spectral analysis
techniques (as the Fast Fourier Transform). Such
methods have generated comparable results but
cannot account for the unavoidable inter-individual
variability that naturally occurs in the pupillary
fluctuation signals. There are some crucial
restrictions using these techniques: the system must
be linear and the data must be strictly stationary;
otherwise, the features extracted via the Fourier
Transform do not have any physiological significance
(Boyina et al., 2012), (Cong, Z. 2009).
Ephemeral nature of many physiological events
indicates associated data are non-linear and non-
stationary waves. Even so, data being non-stationary
has not received the proper attention and its effects
are frequently disregarded. Serious consequences of
such assumptions are inaccurate results or incorrect
interpretation of the underlying physics.
Even if under exceptionally general conditions,
Fourier transform may be used by those methods, it
exists some basic limitations: indeed the system has
to be linear and the data must be strictly periodic or
stationary. Hence, if conditions are not keep within
such limits, then the resulting spectrum will not make
any sense physically at all, and Fourier spectral
analysis based methods will be considered of limited
use. Having no alternatives, current works are still
using Fourier spectral analysis to process such data
(Huang, N., 2005). (Faust and Bairy, 2012). At the
end, methods which do not assume a stationary status
(Cong , Z., 2009) are hardly desirable to have.
In the light of such conditions, it is clear that
computer methods using Fourier spectral analysis are
of limited use. Shortly, both the indiscriminate use of
Fourier spectral analysis and the acceptance of the
stationary and linearity sates may give wrong results,
some of them are described below. (Huang, N., 2005).
(Faust and Bairy, 2012). It is desirable to employ
quantitative methods which do not assume a
stationary status (Cong, Z., 2009). The Hilbert Huang
Transform (HHT) initially developed for natural and
engineering sciences and now is applied to financial
data (Huang et al., 2003). The HHT method is
specially developed for analyzing non-linear and non-
stationary data.
Nowadays, several methods are available for the
time-frequency analysis of non-stationary signals.
For instance, the short-time Fourier transformation
(STFT) can be used when the signal is piece-wise
stationary, whereas the wavelet transform can be used
for linear non-stationary signals (Wu and Huang,
(2005). One drawback of the wavelet transform is that
a priori knowledge about the signal to analyze is
needed in order to choose a suitable wavelet. Another
drawback is that its time-frequency resolution is
limited by the Heisenberg-Gabor uncertainty
principle. In contrast, the HHT can be used to analyze
non-linear and non-stationary signals with excellent
resolution in both time and frequency (Barnhart and
Eichinger 2011) and (Barnhart, 2011).
3 HILBERT HUANG
TRANSFORM
HHT’s development was motivated by the need to
describe non-linear and non-stationary distorted
waves in detail (Huang and Long, 2006). It was
designated by the National Aeronautics and Space
Administration (NASA) Goddard Space Flight
Center (GSFC), and pioneered by (Huang, Long and
Shen, 1996), (Huang et al., 1999), (Huang et al.,
1998), (Huang et al., 2003), (Huang and Shen, 2005)
and (Huang et al., 2007). Since this new analysis
technique was developed, it has shown the ability to
analyze non-linear and non-stationary data in many
areas of research (bio-signal, chemistry, chemical
engineering, financial applications and others).
Different from many of the previous transform
methods, like Fourier-based techniques [wavelet and
fast Fourier Transform], this remarkable method is
intuitive, direct, a posteriori, and adaptive; and
consequently, highly efficient. Mainly because its
origin rise from the decomposition based on and
derived from the physical signal. The bases so derived
have no close analytic expressions, and they can only
Spontaneous Pupillary Oscillation Signal Analysis Applying Hilbert Huang Transform
69

be numerically approximated. (Huang, N. 2005).
This decomposition method is adaptive, and
therefore, highly efficient. As indicated by (Flandrin
et al., 2004), one of the advantages of the HHT is that
its data driven criteria is not fully dependent on
theoretical input or formula. Also, the HHT analyzes
non-stationary signals locally.
There are two processes involved in the HHT: the
Empirical Mode Decomposition (EMD) and the
Hilbert Spectral Analysis (HSA). The key part of the
method is the pre-processing step, the EMD, in which
any complicated data set can be decomposed into a
finite and often small number of Intrinsic Mode
Functions (IMF). With the Hilbert transform, the IMF
yields instantaneous frequencies as functions of time
that give sharp identifications of imbedded structures
(Huang et al., 2003). The final presentation of the
results is a time-frequency-energy distribution, which
was designated as the Hilbert Spectrum. Comparisons
with the Wavelet and Fourier analyses showing that
the HHT method offers much better temporal and
frequency resolutions.
The Hilbert transform, , of any function or
signal is given by:
≡
1
(1)
where PV denotes the Cauchy’s principle value
integral.
An analytic representation of the signal can be
formed with the Hilbert transform pair as:
(2)
where
/
,
(3)
,
and
√
1
and
are the instantaneous amplitudes and
phase functions, respectively (Huang et al., 2001).
The instantaneous frequency can be computed by
means of:
(4)
In order to apply the Hilbert transform to a signal, it
must first be converted to a symmetric signal with
mean zero and no negative maxima and no positive
minima. The Empirical Mode Decomposition (EMD)
is a method to reduce a signal into a collection of
intrinsic mode functions with “well-behaved” Hilbert
transforms. Unlike the Fourier series or transform,
this representation allows a simultaneous
understanding of the signal in both frequency and
time.
3.1 Empirical Mode Decomposition
Empirical Mode Decomposition (EMD) is a tool
which provides a signal-adaptive decomposition
useful for the analysis of non-stationary and non-
linear data and shows a strong capability to precisely
adjust to the spectral content of the analyzed data. It
is based on the concept that any complicated set of
data can be decomposed into a finite and often small
number of mono-component signals called the
Intrinsic Mode Functions (IMF) which are associated
with different spectral contributions and then
applicable to compute the physical meaning of the
complex signal. Decomposition of a signal is made
through an iterative process to cancel out the local
means from the signal. The sifting process is as
follows:
1. Compute all the local extrema (maxima and
minima).
2. Obtain the upper envelope by connecting all the
local maxima with a cubic spline.
3. Repeat for all the minima to obtain the lower
envelope.
Then, the mean of the splines is subtracted from the
original signal. The sifting process is repeated with
the resulting residual signal until the mean and
standard deviation of the average spline is near zero.
The resulting residual signal meets the definition of
an IMF. An IMF is therefore defined as a signal with
zero-mean, and number of extrema and zero-
crossings differing by at most one (Huang et al.,
1998), (Huang et al., 1999) and (Huang et al., 2003).
This first IMF is subtracted from the original signal
and the whole sifting process is repeated with this
new residual signal. The decomposition process stops
when the residue is a monotonic function which not
an IMF. The last extracted IMF corresponds to the
trend which is the lowest frequency component of the
signal.
As a result, the original signal can be written in
terms of the IMFs and the final residue as:
(5)
BIOSIGNALS 2016 - 9th International Conference on Bio-inspired Systems and Signal Processing
70

Using equations 2 and 3, the analytic function can be
written as:
(6)
This function has a similar form to the Fourier
decomposition of a signal
:
(7)
Thus, the EMD decomposition can be regarded as a
generalized Fourier decomposition. Unlike the
Fourier transform, which is predicated on a priori
selection of basic functions that are either of infinite
length or have fixed finite widths, this empirical
decomposition method is adaptive, and, therefore,
highly efficient (Cong, Z., 2009). Since the
decomposition is based on the local characteristic
data time scale, it is applicable to non-linear and non-
stationary processes. The EMD is also useful as a
filter to extract variability of different scales. At a
signal time instance, only a single frequency
component exists. The EMD method is a necessary
data pre-processing before the Hilbert transform can
be applied (Huang, N.E., 2003).
4 DATA PROCESSING AND
ANALYSIS
A data set of 44 documented pupillary records of
healthy university students has been analyzed. Length
of signals are one minute at a rate of 30 fps. Details
of the collected data are: an IR video camera was used
for recording one eye only, typically the left eye, and
they were done under constant luminance level (40
cd/m
2
). Frame by frame, pupil images were recorded
and transferred by means of a video capture board
providing real-time digitizing besides the video
sequences. The video-capture board was fitted by
USB on a Pentium® Dual-Core T4200 @ 2 GHz and
4 GB of internal memory; MATLAB 7.10 (R2013a)
was used to obtain real time recordings of the movie.
The video sequences were collected from subjects in
a resting condition, that is, they were seated on a
comfortable chair. Experiment facilities were located
in a quiet usability laboratory. For readers going into
details about the infrared video Pupillography system
and the image processing method, please refer to
(Villalobos and Suaste, 2013).
Following the procedure described above (EMD
algorithm) the SPO signals were decomposed into
their IMF components. To exemplify the results
obtained from EMD, in Figure 1 a segment of the IMF
components obtained from a selected SPO signal
(black line) are shown. In this example, the algorithm
yielded 8 components (blue lines) plus a residue R
(red line). Each IMF has a distinctive amplitude and
frequency content. For the whole data set (44
pupillograms), the number of IMFs needed to
decompose the signals fluctuates from 4 to 10 modes
(plus the non-linear trend or residue R) Huang et al.,
indicated that the broad number of modes extracted
from a signal is approximately equal to log
2
(N),
where N is the number of points per signal (Huang et
al., 2003). The values obtained in this study fall
within the limits set by that expression.
The intra signals variations could be due to the
informative nature of the IMFs, and is evidence of the
numerous and different factors that direct each
subject’s response. A remarkable advantage of the
decomposition is that the local variation of the pupil
area can be observed directly, the intrinsic
oscillations emerge naturally and, based on
knowledge of the monitored phenomena, solid
conclusions can be obtained about the meaning of
each mode and its relationship to particular behaviors.
Even the residue has a sounded meaning; it keeps
the mean pupil diameter without fast and short lasting
pupil oscillations. The non-linear trend, hallmark of a
non-stationary process, is negative because it slowly
decays through the time scale (Papoulis and
Saunders, 1989). Concerning the frequency and
energy levels involved in the residual component,
from now on, in the IMFs analysis they will be
excluded.
Once the signals were decomposed into their
IMFs, a numerical proof of completeness is
necessary. The numerical reconstruction of the
selected example is as follow:
Spontaneous Pupillary Oscillation Signal Analysis Applying Hilbert Huang Transform
71
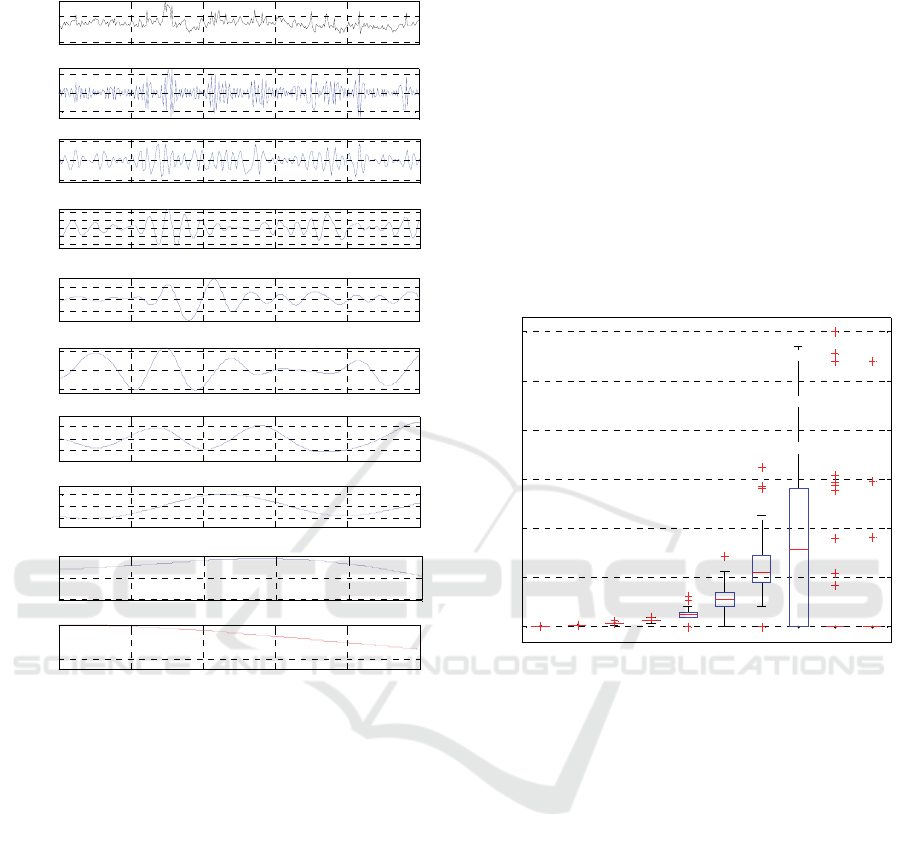
Figure 1: EMD processing of a typical SPF signal. The top
panes is the original signal, imf1 to imf8 are the modes and
res is the residue.
Adding R to each component, starting from the
longest period component to the shortest (IMF1), the
original signal is recreated. When the procedure has
reached the IMF2 the original series has been
practically recovered (total energy contained in the
original SPO data). The latest component (IMF1)
does not contribute significantly to reassemble the
signal, actually it can be seen as noise. The
completeness was numerically verified for the whole
data set. The IMFs pertinence was proved through
null differences between the original signals and the
sum of the components.
4.1 IMF Statistics
In order to clarify the meaning of the IMFs and to use
them adequately to describe the studied phenomena,
the modes are characterized by well-known statistics
metrics.
4.1.1 Period
Based on the definition of the IMF function which
established that the frequency of an IMF can change
continuously with time, the signal period is assumed
as not constant (Huang et al., 2003). The IMF period
can be calculated by dividing the number of data
points by the number of peaks (local maxima). So, if
T is the length of the IMF component and s the
number of peaks, then, the IMF period is equal to T/s.
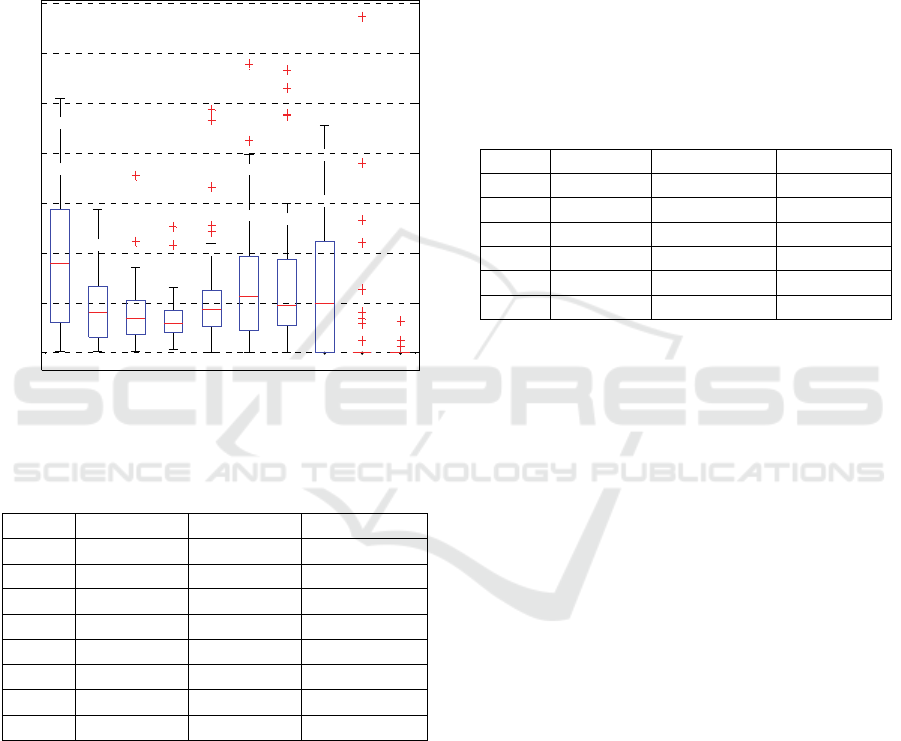
A boxplot for the mean period versus IMFs is
depicted in Figure 2.
Figure 2: Boxplot for the period content according to the
IMF index.
The first mode has the smallest mean period (~3), and
for the successive components the mean period
doubling is followed. For the last mode, the period
variability is evident going from values near zero to
the maximum that almost reaches 600 which is
indicative of very slow pupillary oscillations. About
the period doubling, it is more evident in the first
modes and not clearly verified for the last 2 modes;
this may be due to the increasingly noticeable
presence of noisy components as the decomposition
process approaches the residue.
As observed in the graph, there are not significant
period differences in the whole data set when
analyzing each mode separately. It is important to
note that the period significantly changes in the 8
th
IMF.
4.1.2 Variance
The mean variance contribution from each IMF was
0 2 4 6 8 10
6
8
O riginal
signal
Empirical Mode Decomposition
time [seconds]
0 2 4 6 8 10
-0.5
0
0.5
imf1
time [seconds]
0 2 4 6 8 10
-0.5
0
0.5
imf2
time [seconds]
0 2 4 6 8 10
-0.4
-0.2
0
0.2
0.4
imf3
time [seconds]
0 2 4 6 8 10
-0.2
0
0.2
imf4
time [seconds]
0 2 4 6 8 10
-0.2
0
0.2
imf5
time [seconds]
0 2 4 6 8 10
-0.1
0
0.1
imf6
time [seconds]
0 2 4 6 8 10
-0.1
0
0.1
im f7
time [seconds]
0 2 4 6 8 10
-0.2
0
0.2
imf8
time [seconds]
0 2 4 6 8 10
7
7.5
res.
time [seconds]
0
200
400
600
800
1000
1200
12345678910
IMF
Period
BIOSIGNALS 2016 - 9th International Conference on Bio-inspired Systems and Signal Processing
72
also computed. The variance is used as a simple and
intuitively benchmark to determine the significance
of each component (IMF) of the original signal. Thus,
components with larger variance are more significant.
It was found that the contribution of each IMF to
the total variance also changes from signal to signal
Figure 3 illustrates the boxplot for the variance
contribution (%) depending on the IMF component
number.
Figure 3: Boxplot for the variance content according to the
IMF index.
Table 1: Variance content for a selected subject.
IMF VAR VAR (%) ∑ (%)
1 0.0479 31.7013 31.7013
2 0.0305 20.1545 51.8559
3 0.0219 14.4560 66.3119
4 0.0124 8.2111 74.5230
5 0.0089 5.9113 80.4344
6 0.0068 4.4731 84.9074
7 0.0061 4.0455 88.9529
8 0.0167 11.0471 100.0000
Table 1 gives the contribution of each IMF to the total
sum of variances for a selected subject in absolute
terms, % of total variance and cumulative % variance
contribution.
In this table it is possible to see the large
differences in the variance contribution from each
IMF to the total variance within the selected signals.
Based on the variance content reported in Table 1, it
is concluded for this specific subject, that the most
important components are the highest frequency
modes (IMF1, 2 and 3); the sum of variances for these
important components contribute 66.31% of the total
variance, but the low frequency components have less
effect on the whole change of this SPO signal. The
variance intervals of the first components are wide
and grow as the mode index increases.
Conversely to the last case, in the SPO wave
presented in Table 2, the most important IMF
components are the lowest frequency modes (IMF4,
5 and 6), and their variance sum contributes
86.5874% of the total variance. Therefore, the high
frequency components have less effect on the total
variance of the signal. The interval of the higher
frequency components is narrow while the interval of
the lower frequency modes becomes wider.
Table 2: Variance content of another selected subject.
IMF VAR VAR (%) ∑ (%)
1 0.0007 3.7769 3.7769
2 0.0004 1.9960 5.7729
3 0.0014 7.6397 13.4126
4 0.0038 21.4697 34.8822
5 0.0083 46.6456 81.5279
6 0.0033 18.4721 100.000
As can be concluded, the frequency at which the
highest variance contribution occurs changes from
signal to signal, so the highest variance content for
each signal is present on different IMFs.
4.2 Instant Amplitude and Frequency
Analysis
Having obtained the IMF components, it is not
difficult to apply the Hilbert transform to each IMF
component in order to compute the instantaneous
amplitude and frequency according to equation 3 and
4. The combination of the EMD and the Hilbert
Spectral Analysis is designated as the Hilbert–Huang
Transform (HHT).
4.2.1 Instant Amplitude
Figure 4 shows the boxplot of the amplitude
contribution according to the number of IMFs. As can
be seen, there is a large variation between
components and they are highly intermittent through
the time span. The amplitude variation between
modes leads to the conclusion that for the studied
phenomenon and analyzed population a particular
IMF cannot be selected as the one that contains the
higher energy levels. The mean amplitude
contribution of all components is in the 0.1-0.2 mm
interval.
The amplitude has a slight tendency to decrease as
0
10
20
30
40
50
60
70
12345678910
IMF
Variance (%)
Spontaneous Pupillary Oscillation Signal Analysis Applying Hilbert Huang Transform
73
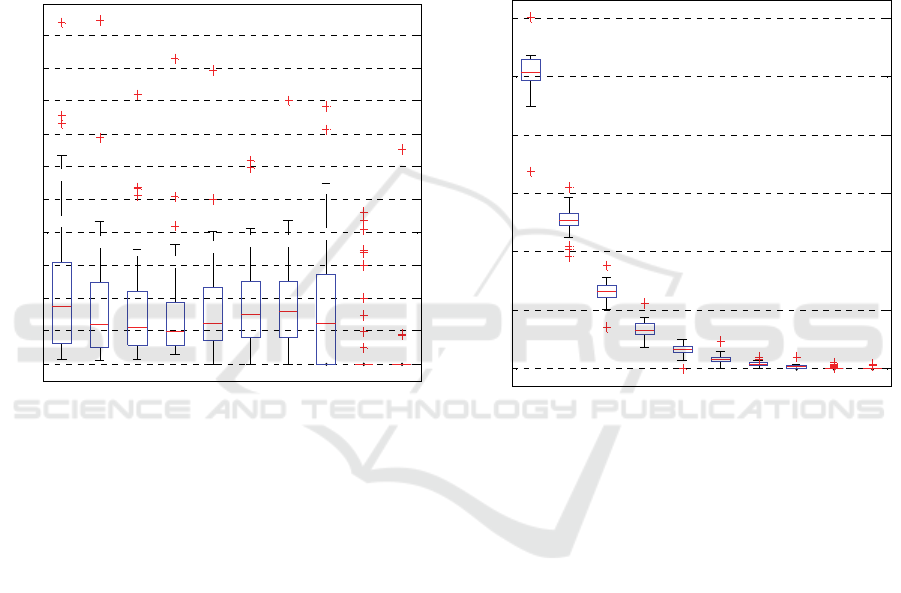
the number of IMFs increases. However, in the first
and last modes (8
th
IMF, which is the most
representative before reaching R) the fluctuations of
the pupil area are wider (~0 - 0.3mm). The amplitude
intervals of in-between modes become high and are
mainly concentrated below the ~0.25 mm range. It is
evident that there is a wide inter-subject variation and
no major modes are detected, i.e. the highest
amplitude values for each signal (subject) are on
different IMFs. While for some subjects the
amplitude is higher at the first modes, for others the
maximum values are present at superior.
Figure 4: Boxplot for the amplitude content according to the
IMF index.
There are interesting responses to be noted:
a) Where amplitude levels are almost equally
distributed in all modes,
b) Where amplitude values different from zero are
only present in the first modes, 1
st
to 4
th
IMFs and,
c) Where the amplitude concentration cannot be
referred to trends a) nor b).
4.2.2 Frequency
Figure 5 illustrates the boxplot of the frequency
variation according to number of IMFs. As can be
observed, the frequency value progressively falls as
the mode number increases. The frequency range of
each IMF is well defined and independent of others.
There are marked differences between the limits of
the frequency characteristic of each mode. It is
verified that the highest frequencies are contained in
the first components and, consequently, the latest
modes have the lower frequency levels. The variation
intervals become narrower as the IMF index is higher.
The first mode has the highest frequency content
(~ 9 Hz to ~11 Hz), the 2
nd
IMF frequency is around
5 Hz while from the 3
rd
to 8
th
modes the activated
frequencies go from ~3 Hz to ~ 0.01 Hz. Observing
the response trends defined concerning amplitude
behavior, interesting relationships can be
pronounced.
There are subjects with strong pupillary
oscillations at the first modes; at the highest
frequency levels, others present them at the higher
modes and at the lower frequency levels.
Figure 5: Boxplot for the frequency content according to the
IMF index.
With regard to the highlighted responses:
a) Some subjects respond with similar amplitude
levels at every frequency detected,
b) The behavior of the subjects having zero
amplitudes in the higher modes must be
interpreted as responses only in the highest
frequency levels and,
c) Some individuals have reactions that activate
frequencies below 1 Hz.
The differences in the frequency content and their
association with the amplitude has an clear effect on
the conclusions about the studied phenomenon, the
stationary statement (signals) and even on the
classification system that generates the data as
nonlinear.
4.3 Spectral Analysis
Having obtained the IMF components, the next step
0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1
12345678910
IMF
Am plitude (mm )
0
2
4
6
8
10
12
12345678910
IMF
Frequency (Hz)
BIOSIGNALS 2016 - 9th International Conference on Bio-inspired Systems and Signal Processing
74
in the proposed method is to apply the Hilbert
Transform to each component and generate the
Hilbert Spectrum (HS). Having the Hilbert Spectrum
defined, we can compute the Marginal Spectrum
(MS) that offers a measure of total amplitude (or
energy) contribution from each frequency value. In
other words, the MS represents the cumulated
amplitude over the entire data spam. So the MS is the
HS that was integrated through all time. In this
simplification, the time coordination is lost as in the
Fourier spectral analysis, which leave a summary of
the frequency content of the event.
The frequency in either HS or MS has a
completely different meaning from results generated
by applying Fourier spectral analysis. The existence
of energy at a particular frequency in the Fourier
representation, means a component of a sine or a
cosine wave persisted through the time span of the
data. So the existence of energy at the particular
frequency, means only that, in the whole time span of
the data, that there is a higher like hood for such a
wave to have appeared locally.
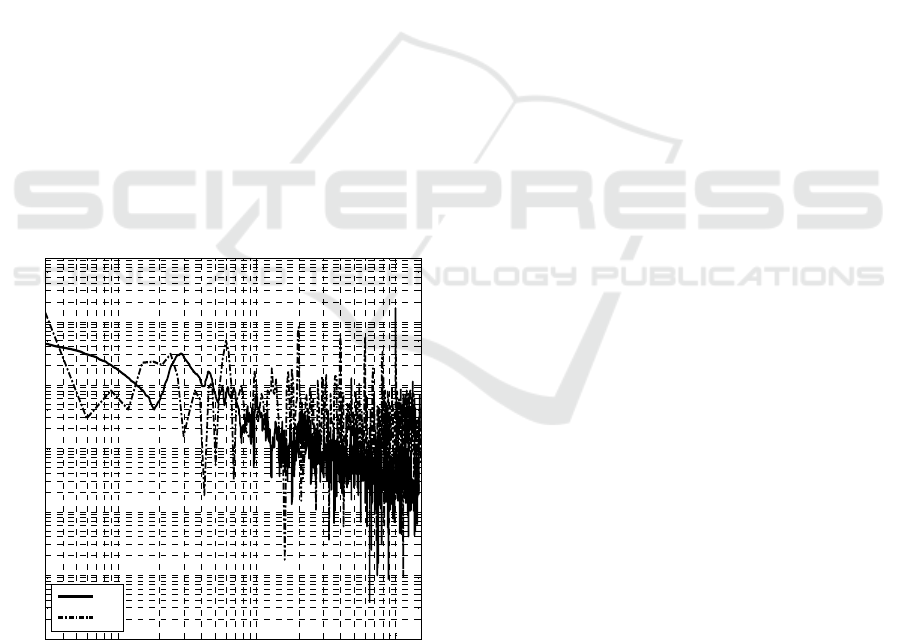
In order to try to compare the proposed spectral
representation method against Fourier conventional
method, we performed a standard FT analysis. In
Figure 6 we directly compare the obtained MS (solid
line) against the conventional Fourier technique
(dotted line) of the SPO data from the Figure 1.
Figure 6: Power spectra of SPF data.
It can be seen that the Marginal spectrum differs from
the Fourier based spectrum in two main issues.
Although the spectral density at the low-frequency
portion is higher in the Hilbert spectra, the Fourier
spectrum is dominated by the DC term due to the non-
zero mean pupil area, this is also meaningless because
of the non-stationary of the data. The spectral density
is lower than the Fourier analysis at higher
frequencies, which was expected because of the
different interpretation of the HHT’s and the Fourier-
based method of data non-linearity. Most of the
Fourier-based techniques always decompose a
nonlinear signal into its base frequency and higher
harmonics; because of this some spectral energy in
the higher frequencies is leaked from their lower
frequency sub harmonics. The HHT interprets signal
non-linearity in terms of frequency modulation, and
the spectral energy of a nonlinear signal remains at
the neighborhood of the base frequency (see Figure
6).
These findings coincided with the ones reported
by (Nowak et al.2008), where by the application of
fast pupillometry for recording and extended spectral
analysis in examining SPO signals, they found
interesting frequency components in some subjects in
the range above 1 Hz. They also reported the
existence of several harmonics even up to 20 Hz
frequency range. The analysis they carried out
showed that the distribution of the PSD amplitude
peaks was different for different frequencies.
Contrary to their proposed method where they
considered the computed numerical parameters as
global, we represent the pupil area fluctuation tacking
advantage of the instantaneous frequency approach,
which even under the worst conditions, is still
consistent with the physics of the system being
studied and could represent it much more accurately
than previous techniques based on Fourier analysis.
5 CONCLUSIONS
This paper has addressed to the possibility of
characterizing the natural behavior of Spontaneous
Pupillary Oscillation SPO records by the time-
frequency signal variations. The objective of
describing data from non-traditional perspectives
(time-frequency-intensity domain) and for finding
specific and indicative behavior patterns is addressed
by applying the HHT to the SPO waves. Non-linear
and non-stationary intrinsic characteristics of these
signals are discovered and reported in this paper using
the advantageous non-traditional signal processing
technique.
The EMD was implemented to decompose the
original SPO signals; the obtained IMF modes
varying from 4-10 plus a non-linear trend which also
10
-1
10
0
10
1
10
-9
10
-8
10
-7
10
-6
10
-5
10
-4
10
-3
Frequency (Hz)
Spectral density
Marginal vs Fourier analysis
HHT
FFT
Spontaneous Pupillary Oscillation Signal Analysis Applying Hilbert Huang Transform
75

showed a clear non-stationary behavior. The fact that
the EMD analysis decomposed the spatially
distributed SPO data into a set of natural oscillations
(Khademul, 2006), showed the IMFs are more
effective in isolating physical processes of various
time scales and are also statistically significant.
The obtained results, lead us to observe that the
SPO signals present local and intermittent pupil area
variations in time. The EMD successively extracts the
IMFs starting with the highest local spatial
frequencies in a recursively way, which is effectively
a set of successive low-pass spatial filters based
entirely on the properties exhibited by the data
(Khademul, 2006). It is also observed that there are
wide inter-subject differences in the variance, period,
amplitude, and frequency contribution from each
mode to the total signal. These inter-mode variations,
lead us to the conclusion that for the studied
phenomenon and analyzed population, a particular
IMF cannot be selected as the one that contains the
higher amplitude level or dominant frequencies.
Our characterized analysis is of a preliminary
nature and many issues have to be addressed and
investigated rigorously, and from the obtained results,
the HHT seems to have much more potential for this
initial approach. Applying non-traditional
alternatives to the study of the pupillograms presents
a great opportunity to understand behaviors and to
mitigate diseases or specific medical conditions, for
example: discern between well and diseased states,
explore if SPF records could provide information for
the evaluation of the psychophysiological response of
ANS to affective triggering events or as a quantitative
way in the assessment of alertness.
As the SPO signals are not stationary, the Fourier
spectrum is meaningless physically, in contrast, we
have demonstrated that with the HHT as analytic
method, the resulting frequency-energy spectrum
provides a physical meaningful interpretation of the
signal.
REFERENCES
Barnhart, B. and Eichinger, W., (2011). Empirical Mode
Decomposition applied to solar irradiance, global
temperature, sunspot number, and CO2 concentration
data. Journal of Atmospheric and Solar-Terrestrial
Physics, 73(13), pp.1771-1779.
Barnhart, B.L., (2011). The Hilbert-Huang Transform:
theory, applications, development, PhD thesis,
University of Iowa.
Bouma, H., and Baghuis, L., (1971). Hippus of the pupil:
Periods of slow oscillations of unknown origin. Vision
Research, 11(11), 1345-1351.
Boyina,S.R, Anu.H, Moneca.K, Mahalakshmi Malini.G,
Priyadharshini.R, (2012). Pupil Recognition Using
Wavelet And Fuzzy Linear Discriminant Analysis For
Authentication, International Journal Of Advanced
Engineering Technology, IJAET/Vol.III/ Issue II.
Calcagnini, G., Censi, F., Lino, S., and Cerutti, S., (2000).
Spontaneous fluctuations of human pupil reflect central
autonomic rhythms. Methods Inf Med, 39(2), 142-145.
Cerutti, S., (1997). Cardiovascular autonomic rhythms in
spontaneous pupil fluctuations, Computers in
Cardiology 1997 CIC-97.
Cerutti, S., (1999). Baroceptor-sensitive fluctuations of
human pupil diameter", Computers in Cardiology,Vol
26 (Cat No 99CH37004) CIC-99.
Cerutti, S., (2000). Pupil diameter variability in humans,
Proceedings of the 22nd Annual
InternationalConference of the IEEE Engineering in
Medicine and Biology Society (Cat No 00CH37143)
IEMBS-00.
Cong, Z., (2009). Hilbert-Huang transform based
physiological signals analysis for emotion recognition,
2009 IEEE International Symposium on Signal
Processing and Information Technology (ISSPIT), 12.
De Souza Neto, E., Abry, P., Loiseau, P., Cejka, J., Custaud,
M., Frutoso, J., Gharib, C. and Flandrin, P. (2007).
Empirical mode decomposition to assess cardiovascular
autonomic control in rats. Fundam Clin Pharmacol,
21(5), pp.481-496.
Elsenbruch, S., (2000). Physiological Measurement, 05.
Faust, O. and Bairy, M., (2012). Nonlinear analysis of
physiological signals: a review. J. Mech. Med. Biol.,
12(04), p.1240015.
Flandrin, P., Rilling, G., and Goncalves, P., (2004).
Empirical mode decomposition as a filter bank. Signal
Processing Letters, IEEE, 11(2), 112-114.
Goldwater, B., (1972). Psychological significance of
pupillary movements. Psychological Bulletin, 77(5),
pp.340-355.
Heaver, B., (2012). Psychophysiological indices of
recognition memory (Doctoral dissertation, University
of Sussex).
Hreidarsson, A. B., and Gundersen, H. J. G.,
(1988).Reduced Pupillary Unrest: Autonomic Nervous
System Abnormality in Diabetes Mellitus, Diabetes.
Huang, N. (2005). Empirical mode decomposition for
analyzing acoustical signals. The Journal of the
Acoustical Society Of America, 118(2), 593.
http://dx.doi.org/10.1121/1.2040268.
Huang, N. E., and Shen, S. S., (2005). Hilbert-Huang
transform and its applications (Vol. 5). World
Scientific.
Huang, N. E., Long, S. R., and Shen, Z., (1996). The
mechanism for frequency downshift in nonlinear wave
evolution. Advances in applied mechanics, 32, 59-
117C.
Huang, N. E., Shen, Z., and Long, S. R., (1999). A new view
of nonlinear water waves: The Hilbert Spectrum 1.
Annual review of fluid mechanics, 31(1), 417-457.
Huang, N. E., Shen, Z., Long, S. R., Wu, M. C., Shih, H.
H., Zheng, Q. and Liu, H. H.,(1998). The
BIOSIGNALS 2016 - 9th International Conference on Bio-inspired Systems and Signal Processing
76

empirical mode decomposition and the Hilbert
spectrum for nonlinear and non-stationary time series
analysis. Proceedings of the Royal Society of London
Mathematical, Physical and Engineering Sciences (Vol.
454, No. 1971, pp. 903-995). The Royal Society.
Huang, N., Wu, M., Qu, W., Long, S., and Shen, S., (2003).
Applications of Hilbert-Huang transform to non-
stationary financial time series analysis. Appl.
Stochastic Models Bus. Ind., 19(3), 245-268.
Huang, N.E., and Long, S., (2006). On the Normalized
Hilbert Transform and Its Applications in Remote
Sensing, Signal and Image Processing for Remote
Sensing.
Huang, N.E., (2003). Applications of Hilbert-Huang
transform to non-stationary financial time series
analysis, Applied Stochastic Models in Business and
Industry, 7.
Huang, Y., Schmitt, F. G., Lu, Z., and Liu, Y.,(2007).
Empirical mode decomposition analysis of
experimental homogeneous turbulence time series. In
21° Colloque GRETSI, Troyes, FRA, 11-14 septembre
2007. GRETSI, Groupe d’Etudes du Traitement du
Signal et des Images.
Jain, S., Siegle, G., Gu, C., Moore, C., Ivanco, L.,
Studenski, S., Greenamyre, J. and Steinhauer, S.,
(2011).Pupillary unrest correlates with arousal
symptoms and motor signs in Parkinson disease.
Movement Disorders, 26(7), pp.1344-1347.
Khademul, I.M., (2006)."Empirical mode decomposition
analysis of climate changes with special reference to
rainfall data", Discrete Dynamics in Nature and
Society.
Longtin, A., (1989). Nonlinear Oscillations, Noise and
Chaos in Neural Delayed Feedback department of
Piysics, McGill University, Montréal, Canada.
Lowenstein, O., (1950). Role of Sympathetic and
Parasympathetic Systems in Reflex Dilatation of the
Pupil. Archives of Neurology and Psychiatry, 64(3),
313.
Lüdtke, H., Wilhelm, B., Adler, M., Schaeffel, F., and
Wilhelm, H., (1998). Mathematical procedures in data
recording and processing of pupillary fatigue waves.
Vision Research, 38(19), 2889-2896.
Malpas, S., (2010). Sympathetic Nervous System
Overactivity and Its Role in the Development of
Cardiovascular Disease. Physiological Reviews, 90(2),
pp.513-557.
McLaren, J. W., Erie, J. C., and Brubaker, R. F., (1992).
Computerized analysis of pupillograms in studies of
alertness. Invest. Ophthalmol. Vis. Sci, 33(3), 671-676.
Naber, M., Alvarez, G., and Nakayama, K., (2013).
Tracking the allocation of attention using human
pupillary oscillations. Frontiers in Psychology, 4.
Newman, S., (2008). Clinical Neuro-Ophthalmology. A
Practical Guide. Journal of Neuro-Ophthalmology,
28(4), p.362.
Nowak, W., Hachol, A. and Kasprzak, H., (2008). Time-
frequency analysis of spontaneous fluctuation of the
pupil size of the human eye. Optica Applicata,
XXXVIII, No. 2, pp.269-280.
Papoulis, A., and Saunders, H., (1989). Probability,
Random Variables and Stochastic Processes (2nd
Edition). J. Vib. Acoust. 111(1), 123.
Pedrotti, M., Mirzaei, M., Tedesco, A., Chardonnet, J.,
Mérienne, F., Benedetto, S., and Baccino, T.,
(2014).Automatic Stress Classification with Pupil
Diameter Analysis. International Journal of Human-
Computer Interaction, 30(3), 220-236.
Regen, F., Dorn, H., and Danker-Hopfe, H., (2013).
Association between pupillary unrest index and waking
electroencephalogram activity in sleep-deprived
healthy adults. Sleep Medicine, 14(9), 902-912.
Rosenberg, M. L., and Kroll, M. H., (1999). Pupillary
hippus: an unrecognized example of biologic chaos.
Journal of Biological Systems, 7(01), 85-94.
Sylvain, S., and Brisson,J., (2014). "Pupillometry:
Pupillometry", Wiley Interdisciplinary Reviews
Cognitive Science.
Villalobos-Castaldi, F., and Suaste-Gómez, E. (2013). A
new spontaneous pupillary oscillation-based
verification system. Expert Systems with Applications,
40(13), 5352-5362. http://dx.doi.org/10.1016/
j.eswa.2013.03.042.
Warga, M., Lüdtke, H., Wilhelm, H., and Wilhelm, B.,
(2009). How do spontaneous pupillary oscillations in
light relate to light intensity? Vision research, 49(3),
295-300.
Wu, Z. and Huang, N.E., (2005).Statistical Significance
Test of Intrinsic Mode Functions", Interdisciplinary
Mathematical Sciences.
Spontaneous Pupillary Oscillation Signal Analysis Applying Hilbert Huang Transform
77
